Abstract
Background
Chlorhexidine gluconate (CHG) is a topical antiseptic solution recommended for skin preparation before central venous catheter placement and maintenance in adults and children. Although CHG is not recommended for use in children aged <2 months owing to limited safety data, it is commonly used in neonatal intensive care units worldwide. We used zebrafish model to verify the effects of early-life exposure to CHG on the developing nervous system, highlighting its impact on oligodendrocyte development and myelination.
Methods
Zebrafish embryos were exposed to different concentrations of CHG from 4 h post fertilization to examine developmental toxicity. The hatching rate, mortality, and malformation of the embryos/larvae were monitored. Oligodendrocyte lineage in transgenic zebrafish embryos was used to investigate defects in oligodendrocytes and myelin. Myelin structure, locomotor behavior, and expression levels of genes involved in myelination were investigated.
Results
Exposure to CHG significantly induced oligodendrocyte defects in the central nervous system, delayed myelination, and locomotor alterations. Ultra-microstructural changes with splitting and fluid-accumulated vacuoles between the myelin sheaths were found. Embryonic exposure to CHG decreased myelination, in association with downregulated mbpa, plp1b, and scrt2 gene expression.
Conclusion
Our results suggest that CHG has a potential for myelin toxicity in the developing brain.
Impact
-
To date, the neurodevelopmental toxicity of chlorhexidine gluconate (CHG) exposure on the developing brains of infants remains unknown.
-
We demonstrated that CHG exposure to zebrafish larvae resulted in significant defects in oligodendrocytes and myelin sheaths.
-
These CHG-exposed zebrafish larvae exhibited structural changes and locomotor alterations.
-
Given the increased CHG use in neonates, this study is the first to identify the risk of early-life CHG exposure on the developing nervous system.
Similar content being viewed by others
Introduction
Chlorhexidine gluconate (CHG) is a commonly used topical antiseptic with broad-spectrum antimicrobial activity against gram-positive and gram-negative bacteria, fungi, and certain types of viruses. It is a cationic biguanide with bacteriostatic and bactericidal mechanisms of action, depending on its concentration.1 CHG has been used to prevent healthcare-associated infections, particularly in patients in the intensive care unit (ICU), including the neonatal ICU (NICU), and 86% of NICUs in the United States report CHG use for antisepsis.2,3,4,5 Despite the current increase in CHG use in neonates, due to limited safety data, no recommendation can be made for the safety or efficacy of chlorhexidine in infants aged <2 months.6 Preterm infants have immature skin with poor skin integrity and vulnerable developing neurological systems, and several studies have raised safety concerns regarding the use of CHG.7,8,9,10 A previous study showed detectable serum levels of systemic absorption after exposure to topical CHG in preterm infants.9
Concerns about CHG-related neurodevelopmental toxicity arise from hexachlorophene (HCP), a phenol derivative similar to CHG, which was withdrawn in the 1970s following cases of vacuolar encephalopathy in children, including preterm infants.11,12 HCP induces myelin vacuolation corresponding to intramyelinic edema of the white matter in the central and peripheral nervous systems.13,14 The changing pattern of the distribution of the vacuolation of the reticular formation at different ages suggests that the susceptibility of myelin is determined by neural maturation.12
The axonal bundles, which form the basis of white matter, mainly develop before the third trimester of pregnancy. However, myelination of the first axons starts after 30 weeks of gestational age and primarily occurs postnatally.15,16 Most myelination occurs during the first year of life, and the number of oligodendrocytes in the human white matter increases by the age of 5 years.16 One report showed that the use of CHG in vitro decreased neuronal growth at concentrations similar to those found in preterm infants exposed to CHG.10 However, early-life exposure of CHG to myelination during brain development and the subsequent behavioral changes have not been studied widely to date.
As an in vivo developmental model organism for pediatric research, zebrafish (Danio rerio) has numerous advantages, including high genetic similarity with humans, rapid embryonic development, and high fecundity, compared to rodents and non-human primates. Furthermore, transparent eggs undergoing external fertilization allow researchers to observe the embryos easily through a microscope from the very beginning of development.17 Because of these strengths, zebrafish has been widely used in the field of neurotoxicity to study the neurological effects of early-life exposure to various chemicals.18 Compared to myelination in rodents, which occurs during the first postnatal month, it occurs within the first week of life in zebrafish. These properties dramatically shorten the time required to investigate the molecular aspects of myelination or to assess the positive and negative effects of molecules on glial development.19
Herein, we used zebrafish to demonstrate the effects of early-life exposure to CHG on the development of the nervous system and emphasize its impact on oligodendrocyte development and myelination. Therefore, we examined the changes in the following aspects: (1) oligodendrocyte formation, (2) myelin synthesis, (3) myelin structure, (4) locomotor behavior, and (5) expression levels of genes involved in myelination to access the potential effects of CHG on zebrafish (Danio rerio) larvae.
Materials and methods
Zebrafish lines
Wild-type zebrafish were used to test developmental toxicity, locomotor behavior, and total cell RNA sequencing. The Tg(olig2:egfp) (#frzcc1072),20 Tg(mbp:egfp) (#frzcc1055),21 and Tg(claudink:gal4-vp16;uas:mgfp) (#frzcc1013)22 transgenic zebrafish lines were used to investigate defects in oligodendrocytes and myelin.
Zebrafish embryos were raised and maintained in the E3 medium (5 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl2, 0.33 mM MgSO4) using standard procedures and staged according to hours post fertilization (hpf).23 A total of 0.003% (weight/volume) 1-phenyl-2-thiourea in the E3 medium was used to prevent the production of pigment in zebrafish embryos.
This study was approved by the Korea University Institutional Animal Care and Use Committee (approval no.: KOREA-2021-0007). All experiments were conducted following the guidelines of the Animal Care Ethics Committee of the Korea University Medical Center and the National Institute of Health guidelines.
Drug exposure and developmental toxicity
We purchased 0.2 mg/ml CHG (Anatomical Therapeutic Chemical code: D08AC02) from Firson (Cheonan, Korea). To test the developmental toxicity of CHG, ten zebrafish embryos were exposed to 4–120 hpf per concentration (i.e., 0.1, 0.25, 0.5, and 1 ng/ml). The hatching rate, mortality, and malformation of the embryos/larvae were monitored three times daily until the end of the exposure. The experiments were repeated three times. A separate set of experiments using the same exposure conditions was conducted to confirm neurotoxicity at 120 hpf by immunohistochemistry analysis.
Image acquisition and analysis
Transgenic zebrafish larvae were embedded in 2% low-melting agarose in a dish containing E3 medium with tricaine (Cat #A5040, Sigma). All fluorescent images were captured using a spinning disk confocal microscope (CSU-X1, Nikon). Confocal images were processed using NIS-Elements AR Analysis 4.30 software (Nikon, Tokyo, Japan). The number of oligodendrocytes and length of the myelin sheath were obtained using the cell counter and length measurements of NIS-Elements.
Transmission electron microscopy (TEM)
Larval tissues were prepared using standard procedures as previously described.24 Zebrafish larvae were fixed in 10% formaldehyde/2.5% glutaraldehyde/0.1 M phosphate buffer, pH 7.4, at 4 °C overnight. The larvae were then postfixed in 1% osmium tetroxide, sequentially dehydrated, and embedded in eponate-12 resin (Cat #18006, Ted Pella). Sections of 1-μm thickness were obtained using a Reichert-Jung Ultracut E ultramicrotome (Leica) and stained with toluidine blue. Sections with a thickness of 60 nm were collected on formvar-coated slot grids, stained with uranyl acetate-lead citrate, and imaged using an H-7500 transmission electron microscope (80 kV, Hitachi, Tokyo, Japan).
Locomotor analysis
All behavioral experiments were conducted between 11:00 am and 3:00 pm in a temperature-controlled room maintained at 27–28 °C. At 5 days post fertilization (dpf), larvae from each group were individually introduced into 48-well plates, each well containing 500 μl of E3 medium. All larvae used in the behavioral analysis were acclimatized for 1 h. Locomotion was recorded using a Daniovision video tracking system (Noldus, Wageningen, The Netherlands) and analyzed using the EthoVision XT 12 system (Noldus, Wageningen, The Netherlands). For the light/dark locomotion test, after acclimatization to the light state for 20 min, the animals were exposed to two cycles of 5 min of dark and 5 min of light and then acclimated to the dark state for 20 min. Each test was independently repeated three times.
Quantitative reverse transcription-polymerase chain reaction (qRT-PCR)
Total RNA was extracted from 5 dpf control and CHG-exposed larvae using TRIzol reagent (Invitrogen) and reverse-transcribed into cDNA using a reverse transcription kit (ImProm-IITM Reverse Transcriptase, Promega). qRT-PCR was performed using a Light Cycler 96 instrument (Roche). The qRT-PCR mixture included 1 μl of diluted cDNA and 1 μl of a mixture of forward and reverse primers, 5 μl of the FastStart Essential DNA Green Master reaction mix (Roche Life Science), and 3 μl of nuclease-free water. The following primers were used for qRT-PCR in this study: β-actin 1 F 5’-ACCCAGACATCAGGGAGTG-3’, R 5’-CATCCCAGTTGGTCACAATAC-3’, mbpa F 5’-CCGTCGTGGAGACGTCAA-3’, mbpa R 5’-CGAGGAGAGGACACAAAGCT-3’, plp1b F 5’-TGCAAAGCACCAGAGTTCCA-3’, plp1b R 5’-AGGGCAAGAAGGGTGATTCC-3’, scrt2 F 5’-ACTACGAGTCGGCCTGCTT-3’, scrt2 R 5’-GGGATTTTTGCGAGCATTAAA-3’, sema7a F 5’-ACCTCTTTCCTGAAGGCACG-3’, sema7a R 5’-ATCCCTCCAGTCCTCAGCAT.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 7.0c software (GraphPad Software, San Diego, CA, USA). One-way analysis of variance (ANOVA) with Tukey’s multiple comparison test was performed to analyze multiple groups, and two-way ANOVA with Tukey’s multiple comparison test was used to analyze survival rate, hatching rate, and velocity under light/dark conditions. Data are expressed as the mean ± standard error of the mean, and statistical significance was set at p < 0.05. Statistical significance is indicated as follows: *p < 0.05, **p < 0.01, ***p < 0.001, and n.s., not significant.
Results
Developmental toxicity of CHG
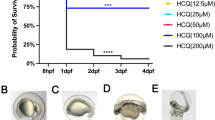
To examine the neurotoxicity of CHG, we first determined the exposure concentration of CHG without developmental toxicity. Zebrafish embryos at 4 hpf were treated with different concentrations of CHG (0.01–10 ng/ml). The effects of CHG on the development of zebrafish embryos are shown in Fig. 1. Concentrations >2 ng/ml were lethal to all embryos within a day (data not shown). In contrast, there were no obvious effects on survival, hatching, and morphology at a concentration <0.25 ng/ml. From these results, we selected 0.25 and 0.5 ng/ml as the appropriate concentration without morphological abnormalities to observe in myelin and behavior.
To test the developmental toxicity of CHG, zebrafish embryos were exposed to 4–120 h post fertilization per concentration (i.e., 0.1, 0.25, 0.5, and 1 ng/ml). a, b The survival and hatching rates of zebrafish larvae exposed to chlorhexidine. c Detailed morphological observations of zebrafish larvae upon early CHG exposure. We selected 0.25 and 0.5 ng/ml as the appropriate concentration without morphological abnormalities to observe in myelin and behavior. Each experiment was repeated independently three times.
Oligodendrocyte defects caused by CHG in the brain of zebrafish
To investigate the effect of CHG on myelin, we analyzed the development of oligodendrocytes that produce myelin sheaths in the central nervous system (CNS). Tg(olig2:EGFP) zebrafish expressed EGFP in oligodendrocyte lineage cells, including oligodendrocyte precursor and premature oligodendrocytes. In the hindbrain region, Olig2+ oligodendrocyte lineage cells were decreased in CHG-exposed larvae compared to control larvae (Fig. 2a–c, m) (control: 121.2 ± 1.79; 0.25 ng/ml CHG: 102.7 ± 3.33; 0.5 ng/ml CHG: 91.47 ± 6.05). In addition, we confirmed the production of mature oligodendrocytes using Tg(mbpa:EGFP), which expresses EGFP in fully mature oligodendrocytes. Similar to the result of olig2+ cells, myelin basic protein (MBP)+ mature oligodendrocytes are significantly reduced in the hindbrain of CHG-exposed larvae in a concentration-dependent manner (Fig. 2d–f, n) (control: 51.1 ± 1.33; 0.25 ng/ml CHG: 47.9 ± 0.97; 0.5 ng/ml CHG: 35.9 ± 1.76).
Tg(olig2:EGFP) zebrafish express oligodendrocyte precursor cells and premature oligodendrocyte cells. In the hindbrain region, Olig2+ oligodendrocyte lineage cells were decreased in CHG-exposed larvae compared to control larvae in the hindbrain region (a–c) and the spinal cord (g–i). Tg(mbpa:EGFP) zebrafish expresses fully mature oligodendrocytes. MBP+ oligodendrocyte lineage cells were decreased in CHG-exposed larvae compared to control larvae in the hindbrain region (d–f) and the spinal cord (j–l). m–p Olig2+ cells, MBP+ mature oligodendrocytes are significantly reduced in the hindbrain and the spinal cord of CHG-exposed larvae in a concentration-dependent manner, and data are expressed as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.0001.
Oligodendrocyte defects caused by CHG in the spinal cord of zebrafish
We then confirmed the neurotoxic effect of CHG on oligodendrocytes in the spinal cord. CHG exposure reduced the number of dorsally migrating Olig2+ cells compared to the control larvae (Fig. 2g–i, o) (control: 10.59 ± 1.84; 0.25 ng/ml CHG: 8.69 ± 2.14; 0.5 ng/ml CHG: 8.4 ± 1.4). Moreover, the number of MBP+ cells are significantly decreased following treatment with 0.25 and 0.5 ng/ml of CHG (Fig. 2j–l, p) (control: 19.04 ± 1.134; 0.25 ng/ml CHG: 15.47 ± 0.50; 0.5 ng/ml CHG: 15.5 ± 0.80). These results revealed that exposure to CHG during early embryonic development induces defects in oligodendrocytes in the CNS.
Delay in myelin synthesis
Single oligodendrocytes produce a limited number of myelin sheaths, which elongate their length to cover target axons during myelination.25 To examine whether CHG affects myelin synthesis in oligodendrocytes, we analyzed the myelin sheath of dorsally located oligodendrocytes in the spinal cord using Tg(claudink:gal4-vp16;uas:mGFP) (Fig. 3a–c). The number of myelin sheath per oligodendrocyte was similar in control and CHG-exposed larvae (Fig. 3d) (control: 13.44 ± 0.88; 0.25 ng/ml CHG: 11.31 ± 0.5; 0.5 ng/ml CHG: 12.94 ± 0.54). However, in CHG-treated larvae, the average length of myelin sheath produced per single oligodendrocyte was reduced in a concentration-dependent manner (Fig. 3e) (control: 15.84 ± 1.09; 0.25 ng/ml CHG: 13.2 ± 1.54; 0.5 ng/ml CHG: 11.35 ± 1.16). This suggests that CHG exposure delayed myelination.
a-c Myelin sheath of dorsally located oligodendrocytes in the spinal cord using Tg(claudink:gal4-vp16;uas:mGFP). d The number of myelin sheaths per oligodendrocyte was similar in control and CHG-exposed larvae. e The average length of myelin sheath per oligodendrocyte was reduced in a concentration-dependent manner in CHG-treated larvae and data. *P < 0.05. This suggests that CHG exposure delayed myelination. The values are presented as the mean ± SEM.
Ultra-microstructural changes of myelin
Next, TEM images of transverse sections of the spinal cord and the posterior lateral line (PLL) were analyzed to reveal ultra-microstructural changes in myelin. A compact myelin sheath was wrapped around the axons in the spinal cord and the PLL (Fig. 4a, b). However, uncompacted, abnormal myelin structures were observed in the CHG-exposed nerves (Fig. 4c, d). The myelin sheath in CHG-treated larvae was looser than the myelin sheath in control larvae, and several splitting and fluid-accumulated vacuoles were observed between the myelin sheaths (Fig. 4c, d). The pathology of myelin is similar to that of HCP, which is a topical antiseptic reagent.26 Therefore, exposure to CHG can induce structural changes in the myelin.
Transmission electron microscopy on transverse sections of the spinal cord and posterior longitudinal ligament (PLL). a, b A compact myelin sheath was wrapped around the axons in the spinal cord and the PLL. c, d Uncompact abnormal myelin structures were observed in the CHG-exposed nerves. c′, c′′, d′ The myelin sheath in CHG-treated larvae was looser than the myelin sheath in control larvae, and several splitting (arrows) and fluid-accumulated vacuoles (asterisks) were observed between the myelin sheaths.
Neurobehavioral locomotor response
We evaluated locomotor activity under alternating light and dark conditions to verify the effects of CHG exposure on behavior. Larvae treated with CHG 0.25 and 0.5 ng/ml each showed decreased locomotion than control larvae (Fig. 5a). In particular, locomotor activity in the light status was significantly decreased. In addition, the total distance moved and mean velocity decreased, and the total time unmoved increased significantly in CHG-treated larvae (Fig. 5b–d). These behavioral changes did not differ between 0.25 ng/ml-treated larvae and 0.5 ng/ml-treated larvae. Together, these results indicate that neurotoxicity induced by exposure to CHG-induced locomotion changes.
Locomotor activity under alternating light and dark conditions. a Larvae treated with CHG 0.25 and 0.5 ng/ml each showed decreased locomotion than control larvae. In particular, locomotor activity in the light status was significantly decreased. **P < 0.01, ***P < 0.001. b–d The total distance moved and mean velocity decreased, and the total time unmoved increased significantly in CHG-treated larvae. These behavioral changes did not differ between 0.25 ng/ml-treated larvae and 0.5 ng/ml-treated larvae. *P < 0.05, ***P < 0.001. The values are presented as the mean ± SEM.
Changes in the expression level of the myelin-related gene
Next, we tested whether CHG exposure induced changes in the expression of myelin-related genes using qRT-PCR (Fig. 6). The expression of myelin basic protein a (mbpa) and proteolipid protein 1b (plp1b), which encode major myelin proteins, was significantly downregulated by CHG exposure, consistent with the histological analysis. The scratch family transcriptional repressor 2 (scrt2) gene is strongly expressed in Schwann cells.27 It was also downregulated in CHG-exposed larvae. semaphorin 7a (sema7a) is expressed in the oligodendrocyte lineage and is known to be involved in oligodendrocyte guidance and neuroinflammation.28 The expression of sema7a was downregulated by exposure to CHG, although the difference was not statistically significant. These qRT-PCR results indicate that CHG exposure reduced the expression of genes involved in myelination.
CHG exposure-induced changes in the expression of myelin-related genes using qRT-PCR The expression of mbpa and plp1b, markers for myelination, was significantly downregulated in CHG-exposed larvae. The scrt2 gene, which is expressed in Schwann cells, is also downregulated in CHG-exposed larvae. The expression of sema7a was downregulated by exposure to CHG, although the difference was not statistically significant. *P < 0.05, **P = 0.01.
Discussion
This study was designed to investigate the neurotoxic potential of CHG in the developing nervous system and emphasize its effect on oligodendrocyte development and myelination. We have shown that exposure to CHG significantly induces delayed myelination, structural myelin changes, and locomotor alterations. Embryonic exposure to CHG decreased myelination, in association with downregulated mbpa, plp1b, and scrt2 gene expression. To our knowledge, this report is the first attempt to investigate the myelin toxicity of CHG in zebrafish larvae.
Overall developmental toxicity to CHG in zebrafish was shown by reductions in hatchability and survival rates.29 In this study, zebrafish embryos at 4 hpf were treated with different concentrations of CHG (0.01–10 ng/ml), and concentrations >2 ng/ml were lethal for all embryos. In contrast, there were no obvious effects on survival, hatching, and morphology at a concentration <0.25 ng/ml. Zebrafish larvae treated with CHG 0.25 and 0.5 ng/ml showed decreased locomotion compared to control larvae. Myelin defects and neurotoxicity affect the locomotor behavior of zebrafish larvae.26
Limited data are available on the toxicity of chlorhexidine in humans.30 CHG is considered to have low toxicity due to poor absorption after oral ingestion.31 Toxicity from oral ingestion of chlorhexidine has been reported to cause gastrointestinal irritation at low concentrations (<20%) and corrosive effects at high concentrations (>20%).32 The LD50 of chlorhexidine digluconate in mice was 2.5 g/kg (oral), 0.02 g/kg (intravenous), and 0.63 g/kg (subcutaneous).30 A value has not been established for the safe concentration of CHG in the blood. In previous human studies, blood levels of CHG have been described in 17 preterm infants born at <32 weeks of gestation after skin exposure to CHG, with concentrations ranging from 0 to 214 ng/ml. The clinical significance is still unknown, even if CHG is detected in the blood.33
In the CNS, we found that the expression of olig2+ cells and MBP+ mature oligodendrocytes associated with myelination was significantly reduced in CHG-exposed larvae in a concentration-dependent manner. In addition, the average length of the myelin sheath per oligodendrocyte was reduced, suggesting that CHG exposure delayed the myelination process. Several previous studies have demonstrated that abnormal CNS or peripheral nervous system development significantly influences toxic agent-induced neurotoxicity in zebrafish embryos.34
Chlorhexidine has been widely used as a topical antiseptic since the 1950s, even in pediatric patients. Due to the limited data evidence regarding the safety of preterm infants in the NICU, there are no guidelines for preferred antiseptics.35 Adverse effects have not been reported in studies involving adults as well as neonates when trace amounts of CHG are absorbed. No data suggest that trace levels of CHG in the blood are clinically important. Although many studies have concluded that CHG is absorbed in infants without any neurotoxicity,8,36,37,38,39 preterm infants, particularly those <32 weeks of gestation, have immature skin with increased permeability, a vulnerable nervous system, and decreased drug clearance, all of which may increase the risk of CHG adverse events.33 In this study, the myelin sheaths in CHG-treated larvae were looser than the myelin sheaths in control larvae, and several splitting and fluid-accumulated vacuoles were observed between the myelin sheaths. This pathology of myelin is similar to that of HCP, another topical antiseptic that is absorbed through the skin of newborns and causes vacuolar encephalopathy.12,40 In addition, the expression of mbpa, plp1b, and scrt2 was significantly downregulated, which is a marker of myelination.
Taken together, the results revealed that systemic absorption of CHG may lead to neurotoxicity through the downregulation of myelination. CHG is a topical agent applied to the skin and differs from the experimental method performed in zebrafish, but exposure to a developing neurological system at a high concentration may cause neurotoxicity.
In conclusion, concerns about the potential neurotoxicity of CHG remain. In this study, the neurotoxicity of CHG was investigated in zebrafish larvae. CHG exposure significantly induced delayed myelination, structural myelin changes, and locomotor alterations by downregulating myelination-related genes. Further studies are warranted to investigate the mechanisms of potential myelin toxicity and the appropriate threshold for serum chlorhexidine concentrations.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Milstone, A. M., Passaretti, C. L. & Perl, T. M. Chlorhexidine: expanding the armamentarium for infection control and prevention. Clin. Infect. Dis. 46, 274–281 (2008).
Sinha, A., Sazawal, S., Pradhan, A., Ramji. S. & Opiyo, N. Chlorhexidine skin or cord care for prevention of mortality and infections in neonates. Cochrane Database Syst. Rev. CD007835 (2015).
Mullany, L. C., Darmstadt, G. L. & Tielsch, J. M. Safety and impact of chlorhexidine antisepsis interventions for improving neonatal health in developing countries. Pediatr. Infect. Dis. J. 25, 665–675 (2006).
Johnson, J. et al. The impact of chlorhexidine gluconate bathing on skin bacterial burden of neonates admitted to the neonatal intensive care unit. J. Perinatol. 39, 63–71 (2019).
Johnson, J. et al. Trends in chlorhexidine use in US neonatal intensive care units: results from a follow-up national survey. Infect. Control Hosp. Epidemiol. 37, 1116–1118 (2016).
O’Grady, N. P. et al. Guidelines for the prevention of intravascular catheter-related infections. The Hospital Infection Control Practices Advisory Committee, Center for Disease Control and Prevention, U.S. Pediatrics 110, e51 (2002).
Vanzi, V. & Pitaro, R. Skin injuries and chlorhexidine gluconate-based antisepsis in early premature infants: a case report and review of the literature. J. Perinat. Neonatal Nurs. 32, 341–350 (2018).
Garland, J. S. et al. Pilot trial to compare tolerance of chlorhexidine gluconate to povidoneiodine antisepsis for central venous catheter placement in neonates. J. Perinatol. 29, 808–813 (2009).
Chapman, A. K. et al. Absorption and tolerability of aqueous chlorhexidine gluconate used for skin antisepsis prior to catheter insertion in preterm neonates. J. Perinatol. 33, 768–771 (2013).
Milstone, A. M. et al. Chlorhexidine inhibits L1 cell adhesion molecule-mediated neurite outgrowth in vitro. Pediatr. Res. 75, 8–13 (2014).
Lockhart, J. D. How toxic is hexachlorophene? Pediatrics 50, 229–235 (1972).
Shuman, R. M., Leech, R. W. & Alvord, E. C. Jr Neurotoxicity of hexachlorophene in the human: I. A clinicopathologic study of 248 children. Pediatrics 54, 689–695 (1974).
Shuman, R. M., Leech, R. W. & Alvord, E. C. Jr Neurotoxicity of topically applied hexachlorophene in the young rat. Arch. Neurol. 32, 315–319 (1975).
Kimbrough, R. D. Review of the toxicity of hexachlorophene, including its neurotoxicity. J. Clin. Pharm. 13, 439–444 (1973).
van Tilborg, E. et al. Origin and dynamics of oligodendrocytes in the developing brain: Implications for perinatal white matter injury. Glia 66, 221–238 (2018).
Hüppi, P. S. et al. Quantitative magnetic resonance imaging of brain development in premature and mature newborns. Ann. Neurol. 43, 224–235 (1998).
Veldman, M. B. & Lin, S. Zebrafish as a developmental model organism for pediatric research. Pediatr. Res. 64, 470–476 (2008).
d’Amora, M. & Giordani, S. The utility of zebrafish as a model for screening developmental neurotoxicity. Front Neurosci. 12, 976 (2018).
Preston, M. A. & Macklin, W. B. Zebrafish as a model to investigate CNS myelination. Glia 63, 177–193 (2015).
Shin, J., Park, H. C., Topczewska, J. M., Mawdsley, D. J. & Appel, B. Neural cell fate analysis in zebrafish using olig2 BAC transgenics. Methods Cell Sci. 25, 7–14 (2003).
Jung, S. H. et al. Visualization of myelination in GFP-transgenic zebrafish. Dev. Dyn. 239, 592–597 (2010).
Münzel, E. J. et al. Claudin k is specifically expressed in cells that form myelin during development of the nervous system and regeneration of the optic nerve in adult zebrafish. Glia 60, 253–270 (2012).
Kimmel, C. B., Ballard, W. W., Kimmel, S. R., Ullmann, B. & Schilling, T. F. Stages of embryonic development of the zebrafish. Dev. Dyn. 203, 253–310 (1995).
Kim, H. T. et al. Specific plasticity of parallel fiber/Purkinje cell spine synapses by motor skill learning. Neuroreport 13, 1607–1610 (2002).
Almeida, R. G., Czopka, T., Ffrench-Constant, C. & Lyons, D. A. Individual axons regulate the myelinating potential of single oligodendrocytes in vivo. Development 138, 4443–4450 (2011).
Persson, L. A., Norlander, B. & Kristensson, K. Studies on hexachlorophene-induced myelin lesions in the trigeminal root transitional region in developing and adult mice. Acta Neuropathol. 42, 115–120 (1978).
Marin, F. & Nieto, M. A. The expression of scratch genes in the developing and adult brain. Dev. Dyn. 235, 2586–2591 (2006).
Cohen, R. I., Rottkamp, D. M., Maric, D., Barker, J. L. & Hudson, L. D. A role for semaphorins and neuropilins in oligodendrocyte guidance. J. Neurochem. 85, 1262–1278 (2003).
Wang, Q. et al. Bioconcentration, metabolism and neurotoxicity of the organophorous flame retardant 1,3-dichloro 2-propyl phosphate (TDCPP) to zebrafish. Aquat. Toxicol. 158, 108–115 (2015).
Liebert, M. A. Final report on the safety assessment of chlorhexidine/chlorhexidine diacetate/chlorhexidine dihydrochloride/chlorhexidine digluconate. J. Am. Coll. Toxicol. 12, 201–223 (1993).
Winrow, M. J. Metabolic studies with radiolabelled chlorhexidine in animals and man. J. Periodontal. Res. Suppl. 12, 45–48 (1973).
Chlorhexidine Salts Monograph. Micromedex Health Care Series, Version 125 (Thomson Health Care, Colorado, 2005).
Chapman, A. K., Aucott, S. W. & Milstone, A. M. Safety of chlorhexidine gluconate used for skin antisepsis in the preterm infant. J. Perinatol. 32, 4–9 (2012).
Wang, X. et al. The developmental neurotoxicity of polybrominated diphenyl ethers: effect of DE-71 on dopamine in zebrafish larvae. Environ. Toxicol. Chem. 34, 1119–1126 (2015).
Loveday, H. P. et al. Epic3: national evidence-based guidelines for preventing healthcare-associated infections in NHS hospitals in England. J. Hosp. Infect. 86, S1–S70 (2014). Suppl 1.
Cowen, J., Ellis, S. H. & McAinsh, J. Absorption of chlorhexidine from the intact skin of newborn infants. Arch. Dis. Child. 54, 379 (1979).
Andersen, C., Hart, J., Vemgal, P. & Harrison, C. Prospective evaluation of a multi-factorial prevention strategy on the impact of nosocomial infection in very-low-birthweight infants. J. Hosp. Infect. 61, 162–167 (2005).
Sankar, M. J. et al. Does skin cleansing with chlorhexidine affect skin condition, temperature and colonization in hospitalized preterm low birth weight infants?: a randomized clinical trial. J. Perinatol. 29, 795–801 (2009).
Lee, A. et al. Blood concentrations of chlorhexidine in hospitalized children undergoing daily chlorhexidine bathing. Infect. Control Hosp. Epidemiol. 32, 395–397 (2011).
Kopelman, A. E. Cutaneous absorption of hexachlorophene in low-birth-weight infants. J. Pediatrics 82, 972–975 (1973).
Acknowledgements
FRZCC #1013, 1055, and 1072 were provided by the Fluorescent Reporter Zebrafish Cooperation Center (FRZCC), Korea.
Funding
This research was supported by the Korea Environment Industry & Technology Institute (KEITI) through the Technology Development Project for Safety Management of Household Chemical Products funded by the Korean Ministry of Environment (MOE) (2020002960007, NTIS-1485017544) and Korea University Ansan Hospital (Grant Nos. O2000281 and O1903601).
Author information
Authors and Affiliations
Contributions
E.K.C. and S.K. were involved in the design, execution, and interpretation of experiments. Y.C. was involved in implementing the animal model. B.M.C. designed the study, advised, and contributed to the editing of the manuscript. E.K.C. wrote the first draft of the manuscript. All authors edited drafts of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Choi, E.K., Choi, B.M., Cho, Y. et al. Myelin toxicity of chlorhexidine in zebrafish larvae. Pediatr Res 93, 845–851 (2023). https://doi.org/10.1038/s41390-022-02186-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02186-6