Abstract
Spinal cord injuries (SCI) are traumatic events with limited treatment options. Following injury, the lesion site experiences a drastic change to both its structure and vasculature which reduces its ability for tissue regeneration. Despite the lack of clinical options, researchers are investigating therapies to induce neuronal regeneration. Cell-based therapies have long been assessed in the context of SCI to promote neuronal protection and repair. Vascular endothelial growth factor (VEGF) not only demonstrates this ability, but also demonstrates angiogenic potential to promote blood vessel formation. While there have been numerous animal studies investigating VEGF, further research is still warranted to pinpoint its role following SCI. This review aims to discuss the literature surrounding the role of VEGF following SCI and its potential in promoting functional recovery.
Similar content being viewed by others
Introduction
Regenerative therapies have been extensively explored to treat traumatic spinal cord injury (SCI), yet few therapies have been translated clinically. Following injury, changes to the structural architecture of the spinal cord decrease the potential for neural regeneration. However, the subsequent glial scar formation and reduction of vasculature to the lesion site may be repaired by exogenous means [1,2,3]. Through cell-based therapies, researchers have been able to study potential treatment options [4]. For example, an ongoing clinical trial investigates the efficacy of adipose tissue-derived mesenchymal stem cells on individuals with chronic SCI [5]. Such therapies are purported to induce a microenvironment more permissible for neural regeneration through their downstream effects. In addition to stem cells, vascular endothelial growth factor (VEGF) is a secretory protein that has intrigued researchers due to its potential for neural regeneration [6].
Angiogenesis, or the formation of new blood vessels, plays an important role in the typical wound healing process, but is reduced around the lesion site following SCI [6]. VEGF not only demonstrates the ability to promote angiogenesis, but also shows potential for neuronal protection and repair [6] (Fig. 1). A study by Basu et al. revealed that the addition of VEGF to Schwann cells improved their phagocytosis of myelin debris and led to significantly lower level of pro-inflammatory cytokines [7]. Subsequent reports have shown its involvement in other spine diseases, including cervical degenerative myelopathy, neurogenic bladder, spinal cord contusion, and discogenic low back pain [8, 9]. This review aims to discuss the literature surrounding the role of VEGF following SCI and its potential in promoting functional recovery. Overall, the authors hope to shed light onto VEGF and encourage further research of its regenerative potential following SCI.
Pathophysiology of SCI
To understand the role of VEGF following SCI, one must consider the mechanism of secondary injury. Upon traumatic impact, the vertebral column displaces leading to transection or contusion of the spinal cord. While primary injury is related to the initial insult of the spinal cord, secondary injury is mediated by the biochemical, physiological, and mechanical changes to the neuronal tissue at the injury site. Secondary injury may manifest through ischemia, inflammation, demyelination, glial scarring, and damage to the vasculature, all of which reduce the ability for tissue regeneration (Fig. 2) [10, 11].
The acute phase of secondary injury initiates when the spinal cord is penetrated as an influx of inflammatory markers and firing of mechanoreceptors trigger signals to the rest of the central nervous system [12]. Chemokines and cytokines such as interleukin (IL)-1, IL-6, tumor necrosis factor (TNF)-α, and interferon (IFN)-γ are recruited to the injury site, inducing a pro-inflammatory state within the local microenvironment. As a secondary cascade of neuroglia and macrophages initiates 24–48 h after injury, so does the sub-acute phase [13]. For the upcoming weeks, the lesion site will experience macrophage infiltration, astrocyte activation, and neural cell differentiation [14]. However, the lack of neurotrophic factors, such as VEGF, and presence of inhibitory factors, such as TNF-α, result in an environment unfavorable for tissue regeneration. Rather, axonal demyelination and glial scar formation ensue. The glial scar dissociates nerve tissue from pro-inflammatory cells, and the initial inflammatory response begins to dissipate. During the chronic phase, inhibitory factors and maturation of the glial scar represent molecular and physical barriers that prevent axon regeneration [14].
Discovery and properties of VEGF
In 1983, Senger et al. described a “Vascular Permeability Factor” when studying fluid accumulation in tumor cells [15]. This permeability factor would soon be coined ‘vascular endothelial growth factor’ by Ferrara and Henzel to reflect its specificity and function [16]. Over the next decade, scientists discovered that VEGF can display different downstream effects by transcribing into different isoforms such as VEGF111, 121, 145, 165, 189, and 206 [17]. For example, VEGF121 is highly diffusible as it lacks heparin-binding, while VEGF189 is highly basic, possesses heparin-binding, and is mostly isolated to the extracellular matrix. VEGF165 on the other hand possesses intermediate properties [18]. Furthermore, the secretory protein sub-family is comprised of seven members (VEGF-A to VEGF-F and placental growth factor), with VEGF-A the most characterized member and specifically VEGF165 as the most biologically active isoform [19]. Once released, VEGF binds to tyrosine kinase receptors VEGFR-1, VEGFR-2, and VEGFR-3. Most notably, VEGFR-1 and VEGFR-2 are present on vascular endothelial cells and upon VEGF binding, downstream PI3k/Akt and MEK/ERK signaling pathways are triggered which have associations with vascularizing neuronal networks (Fig. 3) [20].
Therapeutic potential following SCI
A study by James et al. sheds light into the unique role of VEGF in spinal hemodynamics. The authors reported that during development, VEGF is essential to control the location and timing of blood vessel sprouting and ingression from the perineural vascular plexus (PNVP) [21]. As blood vessels from the PNVP invade the spinal cord, VEGF triggers patterning cues to locally guide vessel sprouting in the CNS [22]. Such changes to the CNS vasculature are not limited to development. In another study, Batholdi et al. assessed the vascular changes following partial transection of the spinal cord in adult rats [23]. The authors noted that transected spinal cords had an early upregulation of VEGF mRNA, which would continue to rise and showed a peak at 24 h. In situ hybridization followed by counterstaining demonstrated that VEGF positive cells were predominantly located in the region of highly reactive astrocytes surrounding the lesion center. Furthermore, a local transient sprouting of vessels would occur over the next several days and seemed to correlate with the spatial distribution of VEGF mRNA. This integral study identified a distinct correlation between VEGF expression patterns and angiogenic responses following SCI [23].
In a similar study, Ritz et al. analyzed protein profiles to gain insight into the temporal expression of certain angiogenic genes [24]. After one week following injury, VEGF and other growth factors such as PDGF-BB, Ang-1 and PlGF, were downregulated. However, other proteins including HGF, CCN1/CYR61, and TGF-β1 demonstrated upregulation. Although the downregulated factors returned to normal values at two-week follow-up, VEGF proved to be the only exception. Time-dependent expression profiling revealed that the downregulation of angiogenic factors following SCI may explain the impairment of both vessel stabilization and blood flow restoration. This study highlights how future considerations of therapeutic applications must leverage angiogenic factors to prevent ischemia in secondary injury [24].
With enhanced knowledge of cellular changes following SCI, researchers began to leverage specific targets to mitigate injury progression. Promoting a robust angiogenic response after SCI has the potential to spare spinal cord tissue, promote neuronal repair, and lead to better functional motor outcomes. Administrating exogenous VEGF following SCI could potentially be the solution to induce neural regeneration and growth. For example, a study by Widenfalk et al. administered intralesional VEGF injection following contusion injury in the rat model to assess blood vessel architecture, apoptosis, and tissue sparing [25]. Rats were delegated into three treatment groups: VEGF (proangiogenic), Ringer’s (control), or angiostatin (antiangiogenic). Angiogenesis was induced in treatment group rats through VEGF administration. Proangiogenic rats demonstrated higher blood vessel density in the lesion area and a decrease in apoptotic cells six weeks after injury [25]. BrdU, a nucleoside analog, was used to label and monitor cell proliferation. There was no significant difference in BrdU levels between groups, indicating that VEGF does not directly influence cell proliferation. Rather, the authors suggest that the beneficial effects seen after VEGF administration were primarily attributable to protection and repair of blood vessels and prevention of apoptosis [25]. While endogenous VEGF is heavily downregulated in mature tissue to prevent tumor angiogenesis, such studies demonstrate the potential of exogenous administration.
Functional outcomes after VEGF treatment following SCI
The primary indication of treatment efficacy lies in its ability to restore motor function. As the cascade of events occurring after SCI is complex and time-dependent, scientists have speculated which modality of administration is optimal. Using the rat model, the predominant test subject for SCI studies, the Basso, Beattie, Bresnahan (BBB) Locomotor Rating Scale assesses functional recovery following SCI [26, 27]. While other assessments do exist to measure behavioral outcomes, such as the rota-rod and catwalk test, BBB scoring is a widely used outcome measure in the literature (Table 1) [27, 28]. These scores can be compared between treatment and control groups to gain insight into functional recovery. Several studies administer VEGF through either implantation or injection, which could also be coupled with other neurotrophic factors to induce a synergistic effect. The current literature sparks some hope, yet many questions are still left unanswered regarding VEGF’s ability to promote recovery.
Lone injection
In the study by Widenfalk et al., hindlimb motor function was also investigated following intralesional VEGF injection. Results showed that stimulating angiogenesis through exogenous VEGF treatment led to significantly improved BBB scores up to six weeks after injury compared to both Ringer’s and angiostatin treatment [25]. This method of administering VEGF after inducing injury to the spinal cord would be a common theme in studies over the following decades. In 2015, a study by Wang et al. also aimed to investigate functional recovery after SCI [29]. Following contusion injury, treatment group rats received intralesional injection of VEGF165 and compared to control rats which were injected with PBS. All injections were administered 24 h post injury as the acute phase was reported to exhibit unfavorable effects following VEGF treatment. As early as 1 day post injection, the VEGF-treated rats demonstrated significant improvements in BBB scores, which persisted at the day 7 and day 21 follow-up. Wang et al. concluded that VEGF165 treatment enhanced functional outcomes following SCI by reducing loss of motor neurons [29].
A study by Liu et al. expanded on this protocol and integrated zinc-finger protein (ZFP) transcription factors into their treatment by using recombinant adenoviral and adeno-associated viral vectors (AAV-ZFP-VEGF). Adult rats received laminectomies and exposed spinal cords were subjected to a force to induce SCI. Immediately, animals were injected with either AAV-ZFP-VEGF or a similar vector with GFP as control. Rats injected with ZFP-VEGF not only had more tissue preserved after SCI, but also had significantly higher BBB scores 6 weeks post injury [30]. Their group then investigated the effect of delaying such treatment in a subsequent study. Using the same bio-engineered vectors, injections were administered 24 h post injury. Using the catwalk test to assess functional outcomes, they reported a dramatic improvement in both hindlimb weight support and swing speed [31].
While these studies report positive results after exogenous VEGF administration following a SCI, results by Sundberg et al. demonstrate differing results. Although VEGF-treated rats did have significantly increased tissue sparing, white matter sparing, and oligodendrogenesis compared to controls 8 weeks post injury, there were no observed differences in functional recovery as measured by BBB, inclined plane, and grid walk neurobehavioral assays [20]. However, the authors do offer the possibility that differences in outcome may have been more prominent at time points beyond eight weeks, as the oligodendrogenesis found after VEGF delivery could have induced remyelination and led to motor function recovery. Furthermore, as injection immediately followed injury, it could be possible that the acute phase of injury reduced the potential benefits of VEGF on the injured tissue [19].
Synergistic effect of VEGF-coupled therapy
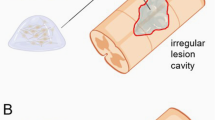
Coupling VEGF with additional therapeutics may ostensibly induce enhanced functional outcomes through a complementary effect on the target tissue, but in reality the nature of SCI is complex and often equivocal. However, given the hypothesis that VEGF promotes angiogenesis, regeneration, and functional outcomes after SCI, it is rational to assess potential synergistic effects by combining additional angiogenic or neurotrophic factors. Platelet-derived growth factor (PDGF), for example, was reported by Chehrehasa et al. to reduce lesion cavity sites in rat spinal cords when coupled with VEGF injections at the injury site [32]. The authors utilized a coupled neutropic therapeutic (VEGF + PDGF) treatment to assess locomotor recovery after treatment group and control rats were subjected to contusion injury. All rats received a miniosmotic pump with a catheter tip placed adjacent to the site of injury, with the control group receiving saline and treatment group receiving VEGF + PDGF for 7 consecutive days. After 4 weeks of BBB scoring, the authors reported growth factor-treated rats had significantly higher scores on days 21 and 28. Such quick locomotive recovery led Chehrehasa et al. to suspect that this combination of growth factors enhances preservation of spinal cord tissue thus reducing functional impairment [32].
PDGF is not the only therapeutic that demonstrated functional improvement when combined with VEGF. A study by Zeng et al. utilized neural stem cells (NSCs) to assess functional recovery following SCI in rats [28]. The authors reported that NSCs had previously demonstrated enhanced vascularization, neuroprotection, and production of VEGF at the injury site. Furthermore, while NSCs are limited by their low survival rate and insufficient regenerative ability, recombinant variants have demonstrated the capacity to overcome this limitation. However, it should be noted that such studies utilized the VEGF165 isoform. To investigate if other isoforms produced similar positive results, Zeng et al. introduced VEGF189 into a lentiviral vector and cultured it with NSCs for two weeks. The transgene NSCs were then delivered to rats (VNSC group) intrathecally via an infusion pump catheter post-contusion injury. Before and after injury, the rota-rod test was performed to measure the number of seconds each rat can remain on a rod turning at an accelerating speed. The time spent on the rod following injury was compared to baseline measurements and reported as latency. Rats in the VNSC group had a significantly higher latency than all other groups each week for 3 weeks post treatment. At week three, VNSC rats had approximately 75% latency, while NSC-treated and PBS-treated groups had an approximate average latency of 55% and 10%, respectively. Overall, the authors reported that VNSC-treated rats had greater recovery with reduced pain [28].
In a similar fashion, Povysheva et al. in 2016 used an Escherichia coli vector to deliver VEGF and angiogenin (ANG) to rodent SCI models [33]. ANG is similar to VEGF as they are both members of the vertebrate-secreted RNAase gene family and induce angiogenesis and neurotrophic effects. While early BBB scoring did not show any significance, days 25, 27, and 29 post injury did demonstrate higher BBB scores for treatment group rats compared to control. Ultimately, Povysheva et al. concluded that the administration of vectors encoding both VEGF and ANG stimulated functional recovery after traumatic SCI in their rat model [33].
Implantation through grafts or scaffolds
While many studies utilized injections for their treatment protocols, other modalities do exist. Kim et al. transplanted a VEGF-overexpressing NSC (F3.VEGF) engraftment into the injured spinal cord of rats [34]. They aimed to investigate if such a therapy resulted in proliferation of endogenous glial progenitor cells. Given that progenitor cells proliferate into glial cells in response to injury, they theorized that the graft could promote glial cell proliferation. Maturation of these glial cells could potentially result in neuroregeneration as they replace astrocytes and oligodendrocytes lost due to spinal cord injury. F3.VEGF grafts were transplanted 2 mm rostral and caudal to the injury epicenter 1 week following contusion injury and then monitored for 6 weeks. Rats transplanted with treatment grafts showed an increased concentration of VEGF within the injury site, which is often both costly and challenging through direct injection of VEGF. Treatment group rats also demonstrated statistically higher concentration of newly born oligodendrocytes, but there was no difference in astrocyte production. Locomotive recovery was assessed using weekly BBB scoring. Compared to control groups, F3.VEGF rats had significantly higher BBB scores at all time points starting 3 weeks after transplantation. Ultimately, Kim et al. reported that this treatment in rats enhanced functional recovery by increasing angiogenesis and neuroprotection after SCI [34].
Similarly, a study by Wang et al. transplanted a collagen scaffold bridge bound to collagen-binding VEGF (CBD-VEGF) into the injury site [35]. The authors’ rationale for such a delivery system considered that VEGF has a short-lived half-life and spreads easily to surrounding areas, but uncontrolled VEGF activity risks tumorigenesis at the delivery site [35]. As collagen is virtually ubiquitous throughout, collagen-binding domains readily bind to areas of interest within the body. Therefore, utilizing a collagen-bound VEGF transplant system may be advantageous by ensuring that the VEGF is released in a controlled, sustained manner. BBB scoring revealed that rats implanted with CBD-VEGF performed significantly better than control rats, but this did not occur until the 7-week mark. Final scores were assessed for both right and left hindlimb, with CBD-VEGF rats averaging significantly higher than the control group. Therefore, the delivery system with stable binding of VEGF may be creating a more suitable microenvironment, guiding axon regeneration, and promoting the formation of new vessels at the injury site, leading to improved functional recovery in SCI [35].
Implantation was further investigated by Xu et al. in 2020. In their study, a homotropic graft of tissue-engineered acellular spinal cord scaffold (ASCS) was implanted into rats post-SCI to co-deliver VEGF165 and neurotrophin-3 (NT-3) [36]. Neurotrophins like neurotrophin-3 are key molecules for the development of the nervous system, but also exhibit osteogenic and angiogenic effects mediated by factors including VEGF [37]. As the mechanism of action of NT-3 may be regulated by VEGF, this study aimed to investigate their synergistic roles on angiogenesis, anti-inflammation, and neural repair through both neural and vascular approaches. Bone mesenchymal stem cells (BMSCs) were also incorporated in the study to determine if growth factors enhanced VEGF/NT-3 release. Hemisection was performed on rat models, followed by transplantation of ASCS at the site of injury. Control, VEGF/NT-3 (VNA), BMSC, VEGF/NT-3+BMSC (VNBC) were the 4 groups that rats were divided into. The two groups treated with VEGF/NT-3 demonstrated reduced inflammation, reduced gliosis, increased angiogenesis, and increased axonal outgrowth with no difference in BMSC administration. However, BMSC did play a significant role in locomotive recovery as seen through BBB scoring. While treatment with VEGF/NT-3, in general, showed statistically significant improvement in motor function, the addition of BMSC further increased BBB scores. At 8 weeks post-SCI, the VNBC group average BBB score was statistically greater than that of the VNA group [37].
Future considerations
As previous studies examined the role of different VEGF isoforms, it is worthwhile to investigate other upstream targets. For example, Shinozaki et al. in 2014 noted that although the receptors VEGF-R1 and VEGF-R2 share common ligands, they are regulated differently after SCI [20]. In their study, VEGF-R1-neutralized mice were undistinguishable to control mice in acute apoptosis, chronic histology, and chronic behavioral assessments. However, they did have significantly lower acute vascular permeability and less acute infiltration of inflammatory cells. On the other hand, VEGF-R2-neutralized mice had more acute apoptosis, less chronic residual tissue, and worse functional recovery than control mice. This suggests that although VEGF as a ligand can promote angiogenesis and neurogenesis, the effects are dependent on which receptor it binds to. While VEGF-R1 seems to influence vascular permeability and infiltration of inflammatory cells, VEGF-R2 confers neuroprotective effects. For SCI and other neurological disorders, maximizing treatment efficacy may require tailored targeting of receptor subtypes.
While VEGF administration demonstrates potential for neural regeneration, scientists must consider the possibility of side effects. VEGF has been noted to promote excessive growth of myelinated axon pathways, particularly nociceptive sensory pathways, which can lead to sensitization and the development of neuropathy [19]. Such complications were found in the study by Sundberg et al. which used the von Frey test to assess sensitivity to stimuli. Mechanical allodynia with hind paw stimulation was compared between VEGF-treated rats and control rats following SCI. The authors reported significantly more rats treated with VEGF developed chronic allodynia [19]. This finding is further supported by a study by Nesic-Taylor et al., in which exogenous VEGF administration significantly increased the incidence of developing pain after SCI [38]. Considering that many SCI patients develop chronic pain from the injury alone, the benefits of treating SCI with VEGF may be at the cost of a higher risk of developing chronic pain conditions. Subsequent studies should explore the development of sensitization and allodynia, as well as other complications that may affect the cost-benefit of receiving treatment.
Conclusion
The current literature demonstrates that exogenous VEGF administration may have the potential to improve functional outcomes following SCI. The previous decades have afforded scientists and researchers great insight into the role of VEGF following SCI. Further clinical translation of this therapeutic strategy would require much more research to be done, along with demonstration of treatment safety and efficacy in a sufficiently powered randomized control trial.
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
References
Anjum A, Yazid MD, Fauzi Daud M, Idris J, Ng AMH, Selvi Naicker A, et al. Spinal cord injury: pathophysiology, multimolecular interactions, and underlying recovery mechanisms. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21207533.
Ahuja CS, Nori S, Tetreault L, Wilson J, Kwon B, Harrop J, et al. Traumatic spinal cord injury-repair and regeneration. Neurosurgery. 2017;80:S9–S22.
Ahuja CS, Wilson JR, Nori S, Kotter MRN, Druschel C, Curt A, et al. Traumatic spinal cord injury. Nat Rev Dis Prim. 2017;3:17018.
Ding D-C, Chang Y-H, Shyu W-C, Lin S-Z. Human umbilical cord mesenchymal stem cells: a new era for stem cell therapy. Cell Transplant. 2015;24:339–47.
Bydon M, Dietz AB, Goncalves S. CELLTOP clinical trial: first report from a phase 1 trial of autologous adipose tissue-derived mesenchymal stem cells in the treatment of paralysis due to traumatic spinal cord injury. Mayo Clin Proc. 2020;95:406–14.
Storkebaum E, Lambrechts D, Carmeliet P. VEGF: once regarded as a specific angiogenic factor, now implicated in neuroprotection. Bioessays. 2004;26:943–54.
Basu S, Choudhury IN, Nazareth L, Chacko A, Shelper T, Vial M-L, et al. In vitro modulation of Schwann cell behavior by VEGF and PDGF in an inflammatory environment. Sci Rep. 2022. https://doi.org/10.1038/s41598-021-04222-7.
Blume C, Geiger MF, Müller M, Clusmann H, Mainz V, Kalder J, et al. Decreased angiogenesis as a possible pathomechanism in cervical degenerative myelopathy. Sci Rep. 2021;11:2497.
Qiu S, Shi C, Anbazhagan AN. Absence of VEGFR‐1/Flt‐1 signaling pathway in mice results in insensitivity to discogenic low back pain in an established disc injury mouse model. J Cell Physiol. 2020;235:5305–17.
Liu Y, Liu Q, Li R. Three-dimensional changes in cervical spinal cord microvasculature during the chronic phase of hemicontusion spinal cord injury in rats. World Neurosurg. 2019;126:e385–e391.
Volkman R, Offen D. Concise review: mesenchymal stem cells in neurodegenerative diseases. Stem Cells. 2017;35:1867–80.
Bartanusz V, Jezova D, Alajajian B, Digicaylioglu M. The blood-spinal cord barrier: morphology and clinical implications. Ann Neurol. 2011;70:194–206.
Kwiecien JM, Dabrowski W, Dąbrowska-Bouta B. Prolonged inflammation leads to ongoing damage after spinal cord injury. https://doi.org/10.1101/865089.
Fan B, Wei Z, Yao X, Shi G, Cheng X, Zhou X, et al. Microenvironment imbalance of spinal cord injury. Cell Transplant. 2018;27:853–66.
Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science. 1983;219:983–5.
Ferrara N, Henzel WJ. Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem Biophys Res Commun. 1989;161:851–8.
Peach CJ, Mignone VW, Arruda MA, Alcobia DC, Hill SJ, Kilpatrick LE, et al. Molecular pharmacology of VEGF-A isoforms: binding and signalling at VEGFR2. Int J Mol Sci. 2018. https://doi.org/10.3390/ijms19041264.
Houck KA, Ferrara N, Winer J, Cachianes G, Li B, Leung DW. The vascular endothelial growth factor family: identification of a fourth molecular species and characterization of alternative splicing of RNA. Mol Endocrinol. 1991;5:1806–14.
Sundberg LM, Herrera JJ, Narayana PA. Effect of vascular endothelial growth factor treatment in experimental traumatic spinal cord injury: in vivo longitudinal assessment. J Neurotrauma. 2011;28:565–78.
Shinozaki M, Nakamura M, Konomi T, Kobayashi Y, Takano M, Saito N, et al. Distinct roles of endogenous vascular endothelial factor receptor 1 and 2 in neural protection after spinal cord injury. Neurosci Res. 2014;78:55–64.
James JM, Gewolb C, Bautch VL. Neurovascular development uses VEGF-A signaling to regulate blood vessel ingression into the neural tube. Development. 2009;136:833–41.
Himmels P, Paredes I, Adler H, Karakatsani A, Luck R, Marti HH, et al. Motor neurons control blood vessel patterning in the developing spinal cord. Nat Commun. 2017;8:14583.
Bartholdi D, Rubin BP, Schwab ME. VEGF mRNA induction correlates with changes in the vascular architecture upon spinal cord damage in the rat. Eur J Neurosci. 1997;9:2549–60.
Ritz M-F, Graumann U, Gutierrez B, Hausmann O. Traumatic spinal cord injury alters angiogenic factors and TGF-beta1 that may affect vascular recovery. Curr Neurovasc Res. 2010;7:301–10.
Widenfalk J, Lipson A, Jubran M, Hofstetter C, Ebendal T, Cao Y, et al. Vascular endothelial growth factor improves functional outcome and decreases secondary degeneration in experimental spinal cord contusion injury. Neuroscience. 2003;120:951–60.
Kjell J, Olson L. Rat models of spinal cord injury: from pathology to potential therapies. Dis Model Mech. 2016;9:1125–37.
Basso DM, Beattie MS, Bresnahan JC. A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma. 1995;12:1–21.
Zeng Y, Han H, Tang B, Chen J, Mao D, Xiong M. Transplantation of recombinant vascular endothelial growth factor (VEGF)189-neural stem cells downregulates transient receptor potential vanilloid 1 (TRPV1) and Improves Motor Outcome in Spinal Cord Injury. Med Sci Monit. 2018;24:1089–96.
Wang H, Wang Y, Li D, Liu Z, Zhao Z, Han D, et al. VEGF inhibits the inflammation in spinal cord injury through activation of autophagy. Biochem Biophys Res Commun. 2015;464:453–8.
Liu Y, Figley S, Spratt SK, Lee G, Ando D, Surosky R, et al. An engineered transcription factor which activates VEGF-A enhances recovery after spinal cord injury. Neurobiol Dis. 2010;37:384–93.
Figley SA, Liu Y, Karadimas SK. Delayed administration of a bio-engineered zinc-finger VEGF-A gene therapy is neuroprotective and attenuates allodynia following traumatic spinal cord injury. PLoS ONE. 2014;9:e96137.
Chehrehasa F, Cobcroft M, Young YW, Mackay-Sim A, Goss B. An acute growth factor treatment that preserves function after spinal cord contusion injury. J Neurotrauma. 2014;31:1807–13.
Povysheva T, Shmarov M, Logunov D, Naroditsky B, Shulman I, Ogurcov S, et al. Post–spinal cord injury astrocyte-mediated functional recovery in rats after intraspinal injection of the recombinant adenoviral vectors Ad5-VEGF and Ad5-ANG. J Neurosurg Spine. 2017;27:105–15.
Kim HM, Hwang DH, Lee JE, Kim SU, Kim BG. Ex vivo VEGF delivery by neural stem cells enhances proliferation of glial progenitors, angiogenesis, and tissue sparing after spinal cord injury. PLoS ONE. 2009;4:e4987.
Wang L, Shi Q, Dai J, Gu Y, Feng Y, Chen L, et al. Increased vascularization promotes functional recovery in the transected spinal cord rats by implanted vascular endothelial growth factor-targeting collagen scaffold. J Orthop Res. 2018;36:1024–34.
Xu Z-X, Zhang L-Q, Zhou Y-N, Chen X-M, Xu W-H. Histological and functional outcomes in a rat model of hemisected spinal cord with sustained VEGF/NT-3 release from tissue-engineered grafts. Artif Cells, Nanomed, Biotechnol. 2020;48:362–76.
Su Y-W, Chung R, Ruan C-S. Neurotrophin-3 induces BMP-2 and VEGF activities and promotes the bony repair of injured growth plate cartilage and bone in rats. J Bone Min Res. 2016;31:1258–74.
Nesic O, Sundberg LM, Herrera JJ, Mokkapati VUL, Lee J, Narayana PA. Vascular endothelial growth factor and spinal cord injury pain. J Neurotrauma. 2010;27:1793–803.
Author information
Authors and Affiliations
Contributions
CZ and GK conceived the idea and were responsible for writing. CO and AKG were responsible for the literature search and data extraction. RJ and FM were responsible for manuscript editing. SG was responsible for conception and design of figures. MB supervised the project and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zamanian, C., Kim, G., Onyedimma, C. et al. A review of vascular endothelial growth factor and its potential to improve functional outcomes following spinal cord injury. Spinal Cord 61, 231–237 (2023). https://doi.org/10.1038/s41393-023-00884-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-023-00884-4