Abstract
Paeoniflorin-6′-O-benzene sulfonate (CP-25) is a new ester derivative of paeoniflorin with improved lipid solubility and oral bioavailability, as well as better anti-inflammatory activity than its parent compound. In this study we explored whether CP-25 exerted therapeutic effects in collagen-induced arthritis (CIA) mice through regulating B-cell activating factor (BAFF)-BAFF receptors-mediated signaling pathways. CIA mice were given CP-25 or injected with biological agents rituximab or etanercept for 40 days. In CIA mice, we found that T cells and B cells exhibited abnormal proliferation; the percentages of CD19+ total B cells, CD19+CD27+-activated B cells, CD19+BAFFR+ and CD19+TACI+ cells were significantly increased in PBMCs and spleen lymphocytes. CP-25 suppressed the indicators of arthritis, alleviated histopathology, accompanied by reduced BAFF and BAFF receptors expressions, inhibited serum immunoglobulin levels, decreased the B-cell subsets percentages, and prevented the expressions of key molecules in NF-κB signaling. Furthermore, we showed that treatment with CP-25 reduced CD19+TRAF2+ cell expressions stimulated by BAFF and decreased TRAF2 overexpression in HEK293 cells in vitro. Thus, CP-25 restored the abnormal T cells proliferation and B-cell percentages to the normal levels, and normalized the elevated levels of IgA, IgG2a and key proteins in NF-κB signaling. In comparison, rituximab and etanercept displayed stronger anti-inflammatory activities than CP-25; they suppressed the elevated inflammatory indexes to below the normal levels in CIA mice. In summary, our results provide evidence that CP-25 alleviates CIA and regulates the functions of B cells through BAFF-TRAF2-NF-κB signaling. CP-25 would be a soft immunomodulatory drug with anti-inflammatory effect.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease characterized by pain, swelling, and joint destruction, leading to the functional disability of joints. The pathogenesis of RA is complex and is highly involved in the interactions of cells and cell mediators [1, 2]. Recent progress in B cells studies revealed that B cells play a critical role in the pathogenesis of RA and other autoimmune diseases [3]. B cells are the effectors of humoral immunity and the producers of antibodies, which generate the rheumatoid factor (RF) and anticitrullinated protein antibodies [1, 4,5,6]. These cells act as antigen-presenting cells (APCs) and regulate dendritic cells and cytokine secretion. Different B-cell subsets are distinguished mainly through the differential expression of different markers on the cell surface [7,8,9,10,11,12]. CD19 is a B-cell-specific surface antigen that regulates the threshold for B-cell activation and is expressed by early pre-B cells and B cells through the phase from heavy chain rearrangement until the differentiation into plasma cells [8]. CD27 is vital surface marker for mutated memory B cells and promotes memory B cells to differentiate into plasma cells [10, 11]. CD138 is a crucial surface marker for plasma cells [13, 14].
The B-cell activating factor (BAFF) belongs to the tumor necrosis factor (TNF) ligand superfamily, which is important for B-cell development and function [15,16,17]. The three different members of receptors for BAFF include the B-cell maturation antigen (BCMA), the B-cell activating factor receptor (BAFFR), and the transmembrane activator and calcium modulator and cyclophilin ligand interactor (TACI) [18]. As type I single transmembrane receptors, all of the three receptors are expressed on memory B cells [15]. BAFFR, which is specific only for BAFF, is a crucial receptor for the survival of mature B cells and is expressed initially in developing B cells coming from the bone marrow [19]. BCMA is expressed primarily on B lymphocytes and tonsillar memory B cells and is restricted to plasmablasts and plasma cells, promoting plasma cell survival [8, 15, 19]. TACI is expressed predominantly on all peripheral B cells and activated T cells. Accordingly, the expression of TACI is also high on plasma cells [8, 15, 19].
It has been well demonstrated that BAFF binding BAFFR promotes human mesangial cells proliferation, and the BAFF−BAFFR interaction triggers the rapid phosphorylation of Akt and mitogen-activated protein kinase p38 kinase, leading to the phosphorylation of the nuclear factor of kappa B65 (p-NF-κB65) activation in human mesangial cells [20]. Furthermore, the BAFFR signal activates the classical and alternative NF-κB signaling pathways, resulting in the expression of downstream genes that are crucial for B-cell survival [21]. In addition, NF-κB regulates inflammation, innate and adaptive immune response, apoptosis, and controls the gene expression for cell proliferation and tumor cell invasion [22]. In the BAFF signaling pathway, the tumor necrosis receptor-associated factor 2 (TRAF2) increases NF-κB2 activation [23]. The three BAFF receptors recruit a species of TRAF to their cytoplasmic domains [24]. TRAF2 is an adaptor molecule that mediates signaling from TNFR family members and participates in the regulation and homeostasis of immune cells [25, 26]. The TRAF2 gene is significantly increased in the peripheral blood mononuclear cells (PBMCs) of RA patients [26, 27].
Paeoniflorin-6′-O-benzene sulfonate (CP-25) originates from the chemistry construct modification of paeoniflorin (Pae) (Fig. 1), which is the effective natural active ingredient of the total glucosides of peony (TGP) [28, 29]. Previous studies in our lab have demonstrated that Pae exerts its anti-inflammatory effects by regulating immune cell responses [30]. The lipid solubility and oral bioavailability of CP-25 was better than those of Pae [31], and the anti-inflammatory effects of CP-25 were better than those of Pae [32, 33]. CP-25 decreased the clinical and histopathological scores and lymphocyte proliferation and suppressed the inflammation progression of adjuvant-induced arthritis (AA) rats and collagen-induced arthritis (CIA) mice [34, 35]. In addition, CP-25 decreased the levels of proinflammatory cytokines, such as IL-1β, IL-6, IL-17, and TNF-alpha, and enhanced the production of the anti-inflammatory cytokine TGF-β1 [34]. Rituximab is a chimeric monoclonal antibody against CD20 that is expressed on B cells and is used in the treatment of B-cell malignancies and RA [14]. Etanercept is an anti-TNF-alpha agent, which reduces the percentage of B cells in the peripheral blood of RA patients by neutralizing TNF-alpha, which is crucial for B-cell proliferation [36]. Etanercept inhibits memory B-cell induction by interfering with germinal center formation and the follicular dendritic cell network [37]. Whether the therapeutic effect of CP-25 on CIA regulates the function of B cells or CP-25 regulates the B-cell function through the BAFF-BAFF receptor-TRAF2-NF-κB signaling pathway is unknown. Compared with biological agents (rituximab or etanercept), the characteristics and advantages of CP-25 are unclear. This study aimed to demonstrate the regulatory effect of CP-25 on B-cell function by investigating the modulatory effect of CP-25 on BAFF/BAFF receptor-TRAF2-NF-κB signaling in CIA mice.
Materials and methods
Animals
The DBA/1 mice (male, 18 ± 2 g) were purchased from the Shanghai Slack Experimental Animal Limited Liability Company (Certificate No. SCXK [HU] 2012-0002). The mice were housed under standard laboratory conditions. All research adhered to the principles of the laboratory animals care guidelines and was approved by the Ethics Review Committee for Animal Experimentation of the Institute of Clinical Pharmacology, Anhui Medical University (No. 20140186, China).
Reagents
The Roswell Park Memorial Institute (RPMI)-1640 medium, Dulbecco’s modified Eagle's medium (DMEM), and fetal bovine serum (FBS) were from Gibco Co. (CA, USA). The CCK-8 kit was purchased from Tianjin Baiying Biological Technology Co., Ltd. (China). The enzyme-linked immunosorbent assay (ELISA) kits for BAFF and recombinant mouse BAFF were purchased from the Research & Development Co., Ltd. (Minneapolis, MN, USA). Chicken type II collagen (CII) was purchased from Chondrex, Inc. (USA). Lipopolysaccharides and concanavalin A (ConA) were purchased from Sigma Chemical Co. (USA). The T/B lymphocyte separation fluid was purchased from Dakewe biotechnology Co., Ltd. Freund’s complete adjuvant was purchased from Sigma. Anti-mouse FITC-CD19, PE-CD27 and APC-CD138, PE-BAFFR, and PE-TACI antibodies were from BD Inc. (USA). The Brilliant Violet 421 anti-mouse CD19 antibody was purchased from Biolegend Co., Ltd., and the TRAF2 (H-10) Alexa Fluor 647 antibody was purchased from Santa Cruz Biotechnology Inc. The cell fixation/permeabilization kit, the HEK 293 cells and the BCA protein quantitation kit were purchased from Thermo Scientific (NO.NCI3225CH). The Lipofectamine™ 3000 transfection reagent kit was purchased from Invitrogen. Inc. The PIRES2-EGFP vector was purchased from BD Inc. The PIRES2-EGFP-TRAF2 plasmid was synthesized by the lab of the Institute of Clinical Pharmacology of Anhui Medical University. The β-actin antibody was purchased from ZSGB-BIO. The mitogen-activated protein kinase kinase3 (MKK3), MKK6, and phosphorylated p38 (p-P38) antibodies were obtained from Santa Cruz Biotechnology Inc. The p-NF-κB65 antibody was purchased from Cell Signaling. The protein antibody array of mouse immunoglobulin was obtained from RayBiotech, Inc.
Drugs
The CP-25 [C29H32O13S, MW: 620] white crystalline powder, purity >98%, was supplied by the chemistry lab of Institute of Clinical Pharmacology of Anhui Medical University (Hefei, Anhui Province, China). Etanercept was provided from Shanghai Guojian Pharmaceutical Co., Ltd. (China). Rituximab was purchased from Roche Pharma (Schweiz) Ltd.
Induction and treatment of CIA
Chicken CII (2 mg/mL) was added in 0.1 mol/L acetic acid overnight at 4 °C under sterile conditions. The chicken CII mixture was then emulsified with an equal volume of Freund’s complete adjuvant (4 mg/mL). This emulsion of CII (0.1 mL) was injected intradermally into the back and base of the tail of the DBA/1 mice on day 0 to induce CIA, and then a booster injection followed on day 21 [35]. The model success rate of the mice was 95%, and some failed CIA mice models were screened out from the study on day 29. After the onset of arthritis on day 29, the CIA mice were randomly divided into five groups. There were 12 mice in each group. The CIA mice were given CP-25 at a dose of 35 mg/kg by an intragastric administration once every day or were treated with rituximab and etanercept at a dose of 5 mg/kg by an intraperitoneal injection [34, 35]. The normal mice and CIA model mice were given an equal volume of normal sodium.
Clinical assessment of CIA
An evaluation of the severity of the CIA was performed by two independent observers with no knowledge of the treatment protocol. Beginning on day 24 after immunization, the mice were evaluated every 3 days using two clinical parameters, including an arthritis index (AI) assessment and a swollen joint count (SJC). After the onset of inflammation, the AI of the CIA mice in each group was evaluated once every 3 days as follows: 0 = no swelling or redness; 1 = phalanx joints slightly swollen and redness; 2 = phalanx joints and paw swelling and redness; 3 = phalanx joints and paw swelling and redness except the ankle joints; and 4 = whole paws and ankle joint swelling and redness. All feet were measured, the maximum score value of each mouse was 16. Each paw has five phalanx joints and one ankle or wrist joint, and thus, the maximum SJC for each mouse was 24 [35, 38]. Additionally, the weights of the mice were also evaluated once every 3 days after the immunization.
The histopathological examination of the spleen and joints
Paraffin sections of the spleen and joint were stained with hematoxylin and eosin. Six mice were analyzed in each group. The histological changes were evaluated by two independent observers. Five compartments were evaluated in the spleen, including the periarteriolar lymphoid sheaths (PALs), lymphoid follicles, marginal zone, red pulp and the total number of germinal centers (GCs). The five compartments were assessed in the joints for synovium hyperplasia, pannus, cartilage erosion, and infiltrated inflammatory cells. The grading scheme consisted of ordinal categories ranging from 0 (no effect) to 4 (severe effect) [8].
The measure of the thymus index and spleen index
The normal and CIA mice were sacrificed, and the thymus and spleen were aseptically obtained. The thymus index or spleen index was shown by the ratios of the weight of the thymus or spleen divided by the weight of mouse.
Analysis of T and B lymphocyte proliferation
T lymphocytes from the thymus and B lymphocytes from the spleen were separated by the lymphocyte separation fluid in each group, and the concentration of the cells was adjusted at 1×106/mL. The T or B lymphocyte suspensions (200 μL) in RPMI 1640 supplemented with 10% fetal calf serum were seeded into 96-well culture plates (1×105 cells per well) and were cultured for 48 h. Triplicates were designed. A total of 10 µL of CCK-8 was added into each well. The cells were incubated at 37 °C under 5% CO2 for 2 h. Then, the absorbance was measured at 450 nm, and the results were analyzed to assess the effect of CP-25, rituximab, and etanercept on T- and B-cell proliferation.
The percentages of B-cell subsets and the BAFF receptors in the PBMCs and spleen lymphocytes were analyzed by flow cytometry
PBMCs were obtained from the blood of the mice using erythrocyte lysate. The lymphocytes in the spleens were separated using lymphocytes separation fluid. A 100 μL of the cell suspension was then transferred into a 12 mm × 75 mm polystyrene round-bottom tube. The antibody combinations of CD19−FITC, CD138-APC and CD27-PE or CD19−FITC and BAFFR-PE or CD19−FITC and TACI-PE were added into the round-bottom tubes. After 40 min of incubation at room temperature in the dark, the cells were washed and analyzed immediately by flow cytometry. Cell-associated fluorescence was analyzed with a FACScan instrument (Epics XL, Beckman Coulter, USA) and associated Cell Quest software (Beckman Coulter, USA).
The level of BAFF was measured by ELISA
The level of BAFF in the serum was measured using an ELISA kit. Each serum sample was assayed in triplicate. First, 80 μL of the assay diluents was added into each well of the 96-well plate. Then, 40 μL of the standard, control, or samples was added into each well and incubated for 2 h at room temperature. After washing five times with a buffer, the mouse antibodies against BAFF were added into each well and were incubated for 2 h at room temperature. Then, the substrate solution was added into each well and was incubated for 30 min. The optical density was measured at 450 nm.
The immunoglobulin levels were measured by protein chip array systems
The levels of serum IgA, IgE, IgD, IgM, IgG1, IgG2a, IgG2b, and IgG3 were analyzed by protein chip array systems (RayBiotech, USA). The operation steps were as follows: (1) The glass chip was air dried completely at room temperature. (2) A diluent (100 μL) was added into each well and was incubated at room temperature for 30 min to block the slides. The samples (100 μL) were added into each well and were incubated for 1−2 h. (3) The detection antibody cocktail (80 μL) was added into each well. The glass chip was incubated for 1−2 h. (4) Cy3 equivalent dye-conjugated streptavidin (80 μL) was added into each well. The device was covered with aluminum foil to avoid exposure to light or was incubated in a dark room for 1 h. (5) For the fluorescence detection, the signals were visualized through the usage of a laser scanner that was equipped with a Cy3 wavelength, such as Axon GenePix. The quantitative data were analyzed by Quantibody® Q-Analyzer software.
The percentages of CD19+TRAF2+ cells in the spleens of the mice were analyzed by flow cytometry
The lymphocytes in the spleens of the normal mice were separated using a lymphocyte separation fluid. The concentration of the cells was adjusted to 1×107/ml. The lymphocyte suspensions (2000 μL) in RPMI 1640 supplemented with 10% fetal calf serum were seeded into 12-well culture plates (5×106 cells per well). The lymphocytes were first stimulated with mouse BAFF (60 ng/mL) at 37 °C under a 5% CO2 environment for 12 h and were then treated with CP-25(10−5 mol/L) or rituximab (10 μg/mL) or etanercept (10 μg/mL). After culturing for 48 h, the lymphocytes were stained with Brilliant Violet 421 anti-mouse-labeled CD19 antibody at room temperature in the dark for 30 min. After surface staining, the cells were fixed and permeabilized by a commercial cell fixation/permeabilization kit and then were stained with an Alexa Fluor 647-labeled TRAF2 intracellular antibody. After incubation for 30 min at room temperature in the dark, the cells were washed, transferred into a 12×75 mm polystyrene round-bottom tube and then analyzed by flow cytometry.
B-cell magnetic separation
The B cells in the spleens were isolated using magnetic cell separation (MACS) by a positive selection. The lymphocytes (108 cells) in the spleen were isolated by lymphocyte separation fluid and were incubated with PE-anti-CD19 for 20 min in 500 µL of MACS buffer, followed by an incubation with anti-PE beads for an additional 20 min. The stained cell suspension was applied onto an LS column (Mitenyi Biotec, Germany). The labeled B cells were collected after washing with degassed buffer three times.
The expressions of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 in the spleen B cells were analyzed by a Western blot
The spleen B cells isolated by MACS were lysed in cell lysis buffer with PMSF 1 mM at 4 °C for 30 min followed by centrifugation at 13622 × g for 10 min at 4 °C. The supernatants were diluted to 4 mg protein/mL and were kept frozen at −80 °C until use. A total of 50 μg of denatured protein was isolated by 10% SDS-PAGE and was transferred onto polyvinylidene fluoride membranes (PVDF membrane, Millipore, USA) in an ice−water environment. The membranes were blocked with blocking buffer (0.05% Tween 20-PBS with 5% nonfat milk) for 2 h at room temperature and were then incubated with primary antibodies targeting rabbit monoclonal TRAF2 (1:500), MKK3, MKK6, p-P38, and p-NF-κB65 (1:500) and mouse monoclonal anti-β-actin (1:500) at 4 °C overnight. Then, the membranes were incubated with anti-rabbit or anti-mouse secondary antibodies conjugated with HRP (1:60,000) for 2 h at 37 °C. The detection of the membrane was achieved by measuring the chemiluminescence of the blotting agent on the film. Finally, the densities of the bands were quantified with a computerized densitometer (ImageJ Launcher, Broken Symmetry Software). The equivalent protein loading and transfer efficiency were verified by staining for β-actin.
The overexpression of TRAF2 in HEK 293 cells was observed by fluorescence microscopy and analyzed by a Western blot
HEK 293 cell suspensions in DMEM supplemented with 10% fetal calf serum were seeded into six-well culture plates. The concentration of the cells was adjusted to 5×106/mL. Then, the cells were cultured at 37 °C under 5% CO2 for 8 h to attach to the six-well culture plates. The PIRES2-EGFP vector plasmid and PIRES2-EGFP-TRAF2 plasmid were transfected into the cells by a Lipofectamine™ 3000 transfection reagent kit. After culturing under 5% CO2 environment at 37 °C for 24 h, the cells were treated with CP-25(10−4 mol/L) or CP-25(10−5 mol/L) or etanercept (10 μg/mL). The cell culture was continued at 37 °C under 5% CO2 for 24 h, and then the cells were observed. In addition, the transfected cells were lysed in cell lysis buffer with PMSF at 4 °C for 30 min, and then centrifugation followed at 14 000 × g for 10 min at 4 °C. The supernatants were transferred into 1.5 mL tubes and detected by a BCA protein quantitation kit. The denatured protein was isolated by 10% SDS-PAGE and transferred onto a PVDF membrane. The membranes were blocked with Tween 20-PBS with 5% nonfat milk for 2 h at room temperature. After incubation with the primary antibodies of rabbit monoclonal TRAF2 (1:500) and mouse monoclonal anti-β-actin (1:500) at 4 °C overnight, the membranes were incubated with secondary antibodies conjugated with HRP (1:60,000) for 2 h at 37 °C. The detection of the membrane was observed and analyzed by the ImageJ launcher of a computerized densitometer and ImageJ software.
Statistical analysis
The data in the figures are presented as the mean ± standard deviation (SD). An analysis of variance (ANOVA) (SPSS Software Products, USA) was used to determine the significant differences between the groups. P values less than 0.05 were considered significant.
Results
CP-25 decreased the AI and SJC and recovered the low weight of the CIA mice
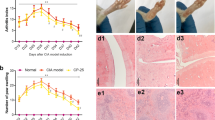
The results showed that the onset of inflammation appeared approximately at day 29 after the primary immunization. The forefeet and hind feet appeared red and swollen in sequence. The AI and SJC were increased with the development of arthritis. The peak of the swelling paws in the CIA mice appeared on day 41 after the primary immunization (Fig. 2a, b). CP-25 decreased the AI and SJC and recovered the low weight of the CIA mice. The paw swelling of the mice in the CP-25 group gradually decreased from day 63. The AI of the CP-25 group was significantly reduced on the 69th day. The SJC in the rituximab and etanercept groups was reduced on day 55. The AI in the rituximab group showed a decrease on day 63. Etanercept decreased AI on day 55. Compared with CP-25, the time for the inhibitory effects of rituximab and etanercept on paw swelling and AI was early (Fig. 2c–e).
The AI, SJC and weights were observed in CIA mice and the effect of CP-25. a The arthritis index (AI) of individual CIA mice from day 29 to day 69 after immunization. b The swollen joint counts (SJC) of individual CIA mice from day 29 to day 69 after immunization. c The AI of CIA mice were assessed and the effects of CP-25. d The SJC of CIA mice were observed and the effects of CP-25. e The weights of CIA mice were observed and the effects of CP-25. **P < 0.01 vs. Normal, #P < 0.05, ##P < 0.01 vs. CIA. $P < 0.05 vs. CP-25
CP-25 alleviated spleen histopathology changes
The histopathology examinations in the spleen were observed by H&E staining. More GCs, infiltrated inflammatory cells, and lymphoid follicular hyperplasia were observed in the CIA mice. Compared with normal mice, an increased PALs, marginal zone and red pulp were also found. After treatment with CP-25 (35 mg/kg), rituximab (5 mg/kg), and etanercept (5 mg/kg), the red pulp congestion was alleviated, the number of GCs decreased, and the PALs were reduced (Fig. 3a, b).
Histopathological evaluation of spleen and joint in CIA mice and the effect of CP-25. a In normal mice, → shows red pulp, ↓ shows white pulp. In CIA mice, → shows germinal center, ↓ shows lymphoid follicular hyperplasia, ↙ periarteriolar lymphoid sheaths, ↘ shows red medullary congestion. b The histological appearances of spleen were assessed. #P < 0.05 vs. CIA (n = 6). c In normal mice, ↓ shows synovial membrane. In CIA mice, ↑ shows synovium hyperplasia, ↓ shows inflammatory cell infiltration, ← shows vascular pannus, ↖ shows cartilage damage. d The histological appearances of joints were assessed. #P < 0.05 vs. CIA (n = 6)
CP-25 improved joint histopathology changes
The histopathological changes of the joints were analyzed by H&E staining, and the results showed that there were differences among the CP-25, rituximab and etanercept-treated mice and the CIA mice. A monolayer synovium or a two-layer synovium was observed, and there were no inflammatory cells in the normal mice. The synovium of the CIA mice showed hyperplasia, a pannus was formed, cartilage was destroyed, and inflammatory cells infiltrated into the synovium, and scores of inflammation were elevated. These abnormalities were mitigated significantly after the administration of CP-25, rituximab, and etanercept. CP-25 reduced synovium hyperplasia, cartilage erosion, and inflammation scores. Rituximab and etanercept decreased synovium hyperplasia, bone erosion, the infiltrated inflammatory cells, and inflammation scores (Fig. 3c, d).
The regulatory effects of CP-25 on the spleen index and thymus index
Compared with normal mice, the spleen index and thymus index obviously increased. CP-25, rituximab, and etanercept significantly inhibited the thymus index and the spleen index. There was a significant difference between CP-25 and etanercept. The effect of etanercept on the thymus index was stronger than the effect of CP-25 (Fig. 4a).
The effects of CP-25 on thymus/spleen indices and T/B cells proliferations in CIA mice. a Effects of CP-25 on indices of thymus and spleen in CIA mice. **P < 0.01 vs. Normal. #P < 0.05 vs. CIA. $P < 0.05 vs. CP-25. b Effects of CP-25 on proliferations of T cell and B cell in CIA mice. **P < 0.01 vs. Normal. ##P < 0.01 vs. CIA. $$P < 0.01 vs. CP-25
The effect of CP-25 on the thymus T-cell and spleen B-cell proliferation
T-cell and B-cell proliferation was measured by CCK-8. The proliferation of the T cells and B cells in the CIA mice was significantly higher than that in the normal mice. CP-25, rituximab, and etanercept inhibited the abnormal proliferation of the T cells and B cells. The inhibitory effect of rituximab and etanercept on T-cell proliferation was stronger than that of CP-25. Rituximab and etanercept strongly inhibited T-cell proliferation to under the normal levels. There was a significant difference between the normal group and the rituximab group or the normal group and the etanercept group. CP-25 mildly downregulated T-cell proliferation to a normal level (Fig. 4b).
The percentage of B-cell subsets and the BAFF receptor expression in the different inflammatory phase of CIA mice in the PBMCs and the effect of CP-25
The percentage of B-cell subsets and the BAFF receptor expression in the PBMCs was analyzed by flow cytometry. The percentage of CD19+ total B cells, CD19+CD27+-activated B cells, CD19+BAFFR+, and CD19+TACI+ cells increased on day 45 after primary immunization, and the percentage of CD19−CD27+CD138+ plasma cells was elevated on day 35. The percentages of CD19+ total B cells and CD19+TACI+ cells were reduced in the CP-25 group on day 65, the percentage of CD19+CD27+-activated B cells and CD19+BAFFR+ cells decreased on day 45, and the percentage of CD19−CD27+CD138+ plasma cells decreased on day 35. The suppressive effects of rituximab and etanercept on the B-cell subsets were stronger than that of CP-25. There was a significant difference between the CP-25 group and the rituximab group or the CP-25 group and the etanercept group. Rituximab and etanercept inhibited the CD19+CD27+-activated B cells and the CD19−CD27+CD138+ plasma cells percentages below the normal levels. There was a significant difference between the normal group and the rituximab group or the normal group and the etanercept group in the CD19+CD27+-activated B cells and the CD19−CD27+CD138+ plasma cells (Fig. 5a–e).
The effects of CP-25 on B cells and BAFF receptors in PBMCs of CIA mice by flow cytometry. a CD19+ total B-cell percentages in PBMCs were analyzed and the effect of CP-25. b The effect of CP-25 on CD19+CD27+-activated B-cell percentages in PBMCs. c CD27+CD19–CD138+plasma cells percentages in PBMCs were analyzed and the effect of CP-25. d The effect of CP-25 on CD19+BAFFR+ cell expressions in PBMCs. e The effect of CP-25 on CD19+TACI+ cell expressions in PBMCs. *P < 0.05, **P < 0.01 vs. Normal. #P < 0.05, ##P < 0.01 vs. CIA. $P < 0.05 vs. CP-25
The percentage of B-cell subsets and BAFF receptor expression in the spleen lymphocytes of the CIA mice and the effect of CP-25
Compared with normal mice, the percentage of the CD19+ total B cells, CD19+CD27+-activated B cells, CD19−CD27+CD138+ plasma cells, CD19+BAFFR+ cells, and CD19+TACI+ cells increased in the CIA mice. CP-25, rituximab, and etanercept inhibited the percentages of the CD19+ total B cells, CD19+CD27+-activated B cells and CD19−CD27+CD138+ plasma cells and reduced the CD19+BAFFR+ and CD19+TACI+ cell percentages. Etanercept inhibited the CD19+CD27+-activated B-cell percentages under normal levels compared with the normal group. There was no significant difference between CP-25 and rituximab or CP-25 and etanercept (Fig. 6a–e).
The effects of CP-25 on B cells and BAFF receptors in spleen lymphocytes of CIA mice by flow cytometry. a The percentages of B-cell subsets were observed. b The percentage of CD19+ total B cells, CD27+CD19–CD138+plasma cells in spleen lymphocytes of CIA mice were analyzed and the effect of CP-25. c CD19+CD27+-activated B-cell percentages in spleen lymphocytes of CIA mice were analyzed and the effect of CP-25. d The expressions of BAFF receptors were observed. e The percentages of CD19+BAFFR+ cell and CD19+TACI+ cell in spleen lymphocytes of CIA mice and the effect of CP-25. *P < 0.05, **P < 0.01 vs. Normal
The serum level of BAFF and the effect of CP-25
The serum level of BAFF in mice was detected by ELISA. As expected, we found a significant elevation of BAFF in the CIA mice. CP-25, rituximab, and etanercept downregulated the level of BAFF. However, there was no significant difference among the three drugs (Fig. 7a).
The effect of CP-25 on BAFF levels, immunoglobulin expressions. a The effect of CP-25 on BAFF levels in CIA mice. b The effect of CP-25 on IgE and IgG3 levels in CIA mice. c The IgG1 levels in CIA mice and the effect of CP-25. d The levels of serum IgA, IgG2a, and IgG2b in CIA mice and the effect of CP-25. e The levels of serum IgD and IgM in CIA mice and the effect of CP-25. *P < 0.05, **P < 0.01 vs. Normal. #P < 0.05, ##P < 0.01 vs. CIA. $P < 0.05, $$P < 0.01 vs. CP-25
The immunoglobulin levels and the effect of CP-25
The levels of IgA, IgE, IgD, IgM, IgG1, IgG2a, IgG2b, and IgG3 were analyzed by protein array systems. Compared with normal mice, the levels of IgA, IgE, IgD, IgG1, IgG2a, and IgG2b were significantly increased in the CIA mice, but the levels of IgM and IgG3 showed no change between the normal mice and the CIA mice. CP-25, rituximab, and etanercept decreased the levels of IgA, IgD, IgG1, IgG2a, and IgG2b. Interestingly, rituximab and etanercept decreased the level of IgE, but CP-25 had no effect on IgE. These three drugs had no effect on the IgG3 and IgM levels. Rituximab and etanercept decreased the levels of IgA and IgG2a below normal levels. There was a significant difference between the normal group and the rituximab group or the normal group and the etanercept group. The effect of rituximab and etanercept on the levels of IgD, IgA, and IgG2a was stronger than that of CP-25. There was a significant difference between the CP-25 group and the rituximab group or the CP-25 group and the etanercept group. Etanercept strongly inhibited the levels of IgG1 under the normal level. CP-25 mildly downregulated the levels of IgA, IgG1, and IgG2a in the CIA mice to a normal level (Fig. 7b–e).
The percentage of CD19+TRAF2+ cells in the spleens of the mice and the effect of CP-25
The percentage of CD19+TRAF2+ cells was analyzed by flow cytometry. Compared with the control group, the expression of CD19+TRAF2+ cells increased after stimulation by BAFF. CP-25, rituximab, and etanercept reduced the expression of CD19+TRAF2+ cells. There was a significant difference between the CP-25 group and the etanercept group. The suppressive effect of etanercept on the expression of CD19+TRAF2+ cells was stronger than that of CP-25. Etanercept and rituximab inhibited the expression of CD19+TRAF2+ cells below normal levels (Fig. 8a, b).
The effect of CP-25 on the expressions of CD19+TRAF2+ cells of mice. a The effect of CP-25 on the expressions of CD19+TRAF2+ cells of mice by flow cytometry. b The percentages of CD19+TRAF2+ cells were observed and the effect of CP-25. The expressions of CD19+TRAF2+ cells were increased after BAFF induction CP-25, rituximab, and etanercept reduced the expressions of CD19+TRAF2+ cells stimulated by BAFF. **P < 0.01 vs. Control. ##P < 0.01 vs. BAFF. $P < 0.05 vs. CP-25
The overexpression of TRAF2 in HEK 293 cells and the effect of CP-25
The expression of TRAF2 was observed and analyzed by fluorescence microscopy and a Western blot. Compared with the control (HEK 293 cell) group and the PIRES2-EGFP vector group, the expression of TRAF2 was increased in the PIRES2-EGFP-TRAF2 plasmid transfection group. CP-25 and etanercept significantly decreased the overexpression of TRAF2 (Fig. 9a–c). There was no significant difference between the CP-25 group and the etanercept group.
The overexpression of TRAF2 in HEK 293 cell and the effect of CP-25. a The figure showed transfection of PIRES2-EGFP-TRAF2 plasmid and the effect of CP-25. b The overexpression of TRAF2 was analyzed by Western blotting. c The effect of CP-25 on the expressions of TRAF2. **P < 0.01 vs. Control. ##P < 0.01 vs. PIRES2-EGFP. &P < 0.05 vs. PIRES2-EGFP-TRAF2
The expression of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 in the B cells and the effect of CP-25
The expressions of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 were analyzed by Western blots. Compared with normal mice, the expressions of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 were obviously increased in the CIA mice. Rituximab and etanercept decreased the elevated expression of TRAF2, MKK3, MKK6, p-P38, p-NF-κB65 in the spleens of the CIA mice. Moreover, CP-25 reduced the expression of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65, and the inhibitory effect of rituximab on the expression of TRAF2, MKK6, and p-P38 was stronger than that of CP-25. The inhibitory effect of etanercept on the expression of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 was stronger than that of CP-25 (Fig. 10a, b). Etanercept inhibited the expression of MKK6 and p-P38 below normal levels. CP-25 regulated the expression of the MKK6 and p-P38 expressions to normal levels.
The effect of CP-25 on the NF-κB signaling molecules expressions. a The expression of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 in B cells of mice were analyzed by Western blotting. b The effect of CP-25 on the expressions of key molecules in NF-κB signaling. **P < 0.01 vs. Normal. #P < 0.05, ##P < 0.01 vs. CIA. $P < 0.05, $$P < 0.01 vs. CP-25
Discussion
RA is an autoimmune disease that affects multiple joints and causes physical disability. Studies of drug therapies for RA have made remarkable progress recently [39]. The CIA mice model is very similar to the pathogenesis of human RA and is a recognized animal model for studying the pathology of RA and therapeutic drugs [8]. In this study, the AI and SJC of the CIA mice increased with the process of inflammation. The pathological changes of spleen and joint synovium in the CIA mice were also abnormal. These findings were consistent with a previous study [35].
T cells and B cells play important roles in autoimmune diseases, including RA. Recent studies have demonstrated a strong correlation between the duration of the disease and the percentage of memory B cells in RA patients [40]. In our study, T cells and B cells showed an abnormal proliferation in the CIA model. The percentages of CD19+ total B cells, CD19+CD27+-activated B cells, and CD19−CD27+CD138+ plasma cells were elevated in the PBMCs and spleen lymphocytes of the CIA mice. It was clarified that B cells were crucial to the process of arthritis. Increased plasma cells result in the abnormal production of immunoglobulins, and immunoglobulins are also involved in the pathology of RA. In a previous study in our lab, the IgD level increased in the RA patients, and IgD stimulated the activation of the PBMCs in RA patients [12]. IgM is one major type of B-cell receptor (BCR) expressed on most of the B cells from the immature to mature stage. IgM-deficient mice exhibited a significantly reduced incidence of CIA and an attenuated disease severity [41]. The elevated BAFF in transgenic mice resulted in an increase in serum immunoglobulins, such as IgA, IgE, IgM, IgG [42]. Here, the levels of immunoglobulin (IgA, IgD, IgE, IgM, IgG1, IgG2a, IgG2b, and IgG3) in the mice were measured. The levels of IgA, IgE, IgD, IgG1, IgG2a, and IgG2b were elevated in the CIA mice. These results indicated that the activation and differentiation of B cells were abnormal during the process of CIA.
BAFF is one of the major factors regulating B-cell development and homeostasis through the binding of its three receptors [43]. In CIA mice and an AA rat model, the BAFF levels were significantly high [30, 38]. The study by Jia et al. showed that the mRNA expressions of BAFF and BAFFR were increased in the mesenteric lymph nodes in AA rats [43]. In the research by Tue G et al., BAFF exerted a positive effect on the maturation and proliferation of B cells [44]. These findings were consistent with our results that the serum BAFF levels in CIA mice were enhanced, which might lead to the abnormal proliferation, activation, and differentiation of B cells. Activated BAFFR mediates the classical and nonclassical NF-κB signaling pathways [21]. TRAF2 is a multifunctional regulator that regulates the canonical and noncanonical NF-κB pathways in B cells [23]. In this study, we analyzed the canonical NF-κB pathway by the expressions of key molecules in CIA mice. Moreover, we detected the expression of CD19+TRAF2+ cells stimulated by BAFF in the mice and measured the overexpression of TRAF2 in the HEK 293 cells. We found that the expressions of BAFFR and TACI were upregulated on the B cells in the PBMCs and spleen lymphocytes in the CIA mice. CD19+TRAF2+ cell expression increased in the spleen of the mice stimulated by BAFF in vitro. The expressions of TRAF2, MKK3, MKK6, p-P38, and p-NF-κB65 in the B cells of the spleen obviously increased in the CIA mice. This finding suggests that the NF-κB signaling pathways mediated by BAFF in the B cells of the CIA mice were abnormal.
In clinics, therapeutic drugs for RA include nonsteroidal anti-inflammatory drugs, steroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, biological agents, traditional Chinese medicine, and natural medicine. The therapeutic effects of drugs have been achieved, but the adverse reactions of some drugs were also observed. Adverse reactions, such as malignant tumors and infections, have been found with biological agents, including a BAFF monoclonal antibody, a CD20 monoclonal antibody (rituximab), and the inhibitors of TNF-alpha (etanercept) [45,46,47,48,49]. CP-25 is a new ester derivative from the chemistry construct modifications of Pae, and Pae is from Chinese traditional medicine [28]. Previous studies in our lab have demonstrated that CP-25 alleviated the inflammatory process by regulating the abnormal immune system in experimental arthritis [34, 35]. In this study, we further clarified the effective mechanisms of CP-25 on CIA mice, specifically to investigate the regulatory effects of CP-25 on B-cell function. Additionally, we also compared the effect features of CP-25 with biological agents (rituximab or etanercept). CP-25 reduced the percentage of total B cells, the activated B cells and the plasma cells, decreased the expression of the BAFF receptors, downregulated the level of BAFF, inhibited the activation of the NF-κB signaling pathway in the CIA mice, and decreased the expression of CD19+TRAF2+ cells in the mice and the overexpression of TRAF2 in vitro. These findings suggest that CP-25 downregulated the function of B cells by adjusting the BAFF-BAFF receptor-TRAF2-NF-κB signaling pathways.
The effect of rituximab and etanercept on B-cell function was stronger than that of CP-25. Rituximab and etanercept strongly inhibited the B-cell subsets, the BAFF receptor expression, the level of immunoglobulins, and so on, to be under the normal level. There was a significant difference between the CP-25 group and rituximab group or the CP-25 group and etanercept group. The effect of CP-25 was mild. These results suggest that CP-25 might be a soft immunomodulatory drug with an anti-inflammatory effect [50]. Meanwhile, the dysfunction of T/B cells, caused by rituximab and etanercept, may be one of the mechanisms of the adverse reactions in the treatment of RA.
Conclusion
In conclusion, BAFF plays an important role in B-cell activation by mediating TRAF2-NF-κB signaling in the pathological progress of CIA. CP-25 has a therapeutic effect on CIA mice by decreasing AI and SJC and inhibiting T/B-cell proliferation. CP-25 inhibited activated B-cell function by downregulating the percentage of B-cell subsets, the BAFF receptor expression, the BAFF levels and the immunoglobulin levels and inhibiting the activation of the NF-κB signaling pathways (Fig. 11). These findings suggest that CP-25 might be a soft immunomodulatory drug with an anti-inflammatory effect. The effect of rituximab and etanercept on the function of B cells was stronger than that of CP-25, which might be one of the mechanisms of the adverse reaction in the immune system for the biological agent. This study might provide new insight for the development of anti-inflammatory immune drugs for the treatment of RA.
To summary the effect of CP-25, rituximab and etanercept on BAFF/BAFF receptor-TRAF2-NF-κB signaling in B cell on CIA mice. The whole criteria of CIA mice were increased, the function of B cells was abnormal, and NF-κB signaling pathways mediated by BAFF in B cells of CIA mice were activated. CP-25 reduced the whole criteria, T/B cells proliferations, and downregulated abnormal B-cell function and the key molecules of BAFF-NF-κB signaling pathways. The action intensity of rituximab and etanercept on B cells was strong, which might be one of mechanisms of adverse reaction. AI arthritis index, BAFF B-cell activating factor, BAFFR B-cell activating factor receptor, CIA collagen-induced arthritis, CREB cAMP response element binding protein, MKK3 mitogen-activated protein kinase kinase 3, MKK6 mitogen-activated protein kinase kinase 6, p-P38 phosphorylated p38 mitogen-activated protein kinase, NF-κB nuclear factor of kappa B, IκB inhibit of nuclear factor of kappa B, SJC swollen joint counts, TACI transmembrane activator and calcium modulator and cyclophilin ligand interactor, TRAF2 tumor necrosis factor receptor-associated factors 2
References
Mease PJ. B cell-targeted therapy in autoimmune disease: rationale, mechanisms, and clinical application. J Rheumatol. 2008;35:1245–55.
Shu JL, Zhang F, Zhang LL, Wei W. G protein coupled receptors signaling pathways implicate in inflammatory and immune response of rheumatoid arthritis. Inflamm Res. 2017;66:379–87.
Salazar-Camarena DC, Ortiz-Lazareno PC, Cruz A, Oregon-Romero E, Machado-Contreras JR, Muñoz-Valle JF, et al. Association of BAFF, APRIL serum levels, BAFFR, TACI and BCMA expression on peripheral B cell subset with clinical manifestations in systemic lupus erythematsus. Lupus. 2016;25:582–92.
Pillai S, Mattoo H, Cariappa A. B cells and autoimmunity. Curr Opin Immunol. 2011;23:721–31.
Liu Z, Davidson A. BAFF and selection of autoreactive B cells. Trends Immunol. 2011;32:388–94.
Cohen SB. Targeting the B cell in rheumatoid arthritis. Best Prac Res Clin Rheumatol. 2010;24:553–63.
Leandro MJ. B-cell subpopulations in humans and their differential susceptibility to depletion with anti-CD20 monoclonal antibodies. Arthritis Res Ther. 2013;15(Suppl 1):S3 https://doi.org/10.1186/ar3908.
Liu Y, Zhang L, Wu Y, Tong T, Zhao W, Li P, et al. Therapeutic effects of TACI-Ig on collagen-induced arthritis by regulating T and B lymphocytes function in DBA/1 mice. Eur J Pharmacol. 2011;654:304–14.
Rossi JF. Targeted therapies in adult B-cell malignancies. Biomed Res Int. 2015;2015:217593 https://doi.org/10.1155/2015/217593.
Han BK, Olsen NJ, Bottaro A. The CD27-CD70 pathway and pathogenesis of autoimmune disease. Semin Arthritis Rheum. 2016;45:496-501.
Roll P, Palanichamy A, Kneitz C, Dorner T, Tony HP. Regeneration of B cell subsets after transient B cell depletion using anti-CD20 antibodies in rheumatoid arthritis. Arthritis Rheum. 2006;54:2377–86.
Wu Y, Chen W, Chen H, Zhang L, Chang Y, Yan S. et al. The elevated secreted immunoglobulin D enhanced the activation of peripheral blood mononuclear cells in rheumatoid arthritis. PLoS ONE. 2016;11:e0147788 https://doi.org/10.1371/journal.pone.0147788.
Byun JK, Moon SJ, Jhun JY, Kim EK, Park JS, Youn J, et al. Rebamipide attenuates autoimmune arthritis severity in SKG mice via regulation of B cell and antibody production. Clin Exp Immunol. 2014;178:9–19.
Teng YK, Levarht EW, Hashemi M, Bajema IM, Toes RE, Huizinga TW, et al. Immunohistochemical analysis as a means to predict responsiveness to rituximab treatment. Arthritis Rheum. 2007;56:3909–18.
Morais SA, Vilas-Boas A, Isenberg DA. B-cell survival factors in autoimmune rheumatic disorders. Ther Adv Musculoskelet Dis. 2015;7:122–51.
Mackay F, Leung H. The role of the BAFF/APRIL system on T cell function. Semin Immunol. 2006;18:284–9.
Li PP, Liu DD, Liu YJ, Song SS, Wang QT, Chang Y, et al. BAFF/BAFF-R involved in antibodies production of rats with collagen-induced arthritis via PI3K-Akt-mTOR signaling and the regulation of paeoniflorin. J Ethnopharmacol. 2012;141:290–300.
Mackay F, Silveira PA, Brink R. B cells and the BAFF/APRIL axis: fast-forward on autoimmunity and signaling. Curr Opin Immunol. 2007;19:327–36.
Scholz JL, Crowley JE, Tomayko MM, Steinel N, O’Neill PJ, Quinn WJ 3rd, et al. BLyS inhibition eliminates primary B cells but leaves natural and acquired humoral immunity intact. Proc Natl Acad Sci U S A. 2008;105:15517–22.
Zheng N, Wang D, Ming H, Zhang H, Yu X. BAFF promotes proliferation of human mesangial cells through interaction with BAFF-R. BMC Nephrol. 2015;16:1-10.
Kreuzaler M, Rauch M, Salzer U, Birmelin J, Rizzi M, Grimbacher B, et al. Soluble BAFF levels inversely correlate with peripheral b cell numbers and the expression of BAFF receptors. J Immunol. 2012;188:497–503.
Ma J, Mi C, Wang KS, Lee JJ, Jin X. 4′, 6-Dihydroxy-4-methoxyisoaurone inhibits TNF-alpha-induced NF-κB activation and expressions of NF-κB-regulated target gene products. J Pharmacol Sci. 2015; pii: S1347-8613(15)00206-6.
Grech AP, Amesbury M, Chan T, Gardam S, Basten A, Brink R. TRAF2 differentially regulates the canonical and noncanonical pathways of NF-B activation in mature B cells. Immunity. 2004;21:629–42.
Vallabhapurapu S, Matsuzawa A, Zhang W, Tseng PH, Keats JJ, Wang H, et al. Nonredundant and complementary functions of TRAF2 and TRAF3 in an ubiquitination cascade that activates NIK-dependent alternative NF-kappa B signaling. Nat Immunol. 2008;9:1364–70.
Etemadi N, Chopin M, Anderton H, Tanzer MC, Rickard JA, Abeysekera W, et al. TRAF2 regulates TNF and NF-κB signalling to suppress apoptosis and skin inflammation independently of Sphingosine kinase 1. eLife. 2017;6:e29849
Cabal-Hierro L, Lazo PS. Signal transduction by tumor necrosis factor receptors. Cell Signal. 2012;24:1297–305.
Raghav SK, Gupta B, Agrawal C, Chaturvedi VP, Das HR. Expression of TNF-alpha and related signaling molecules in the peripheral blood mononuclear cells of rheumatoid arthritis patients. Mediators Inflamm. 2006;2006:R12682.
Wang C, Yuan J, Wei W. Study on paeoniflorin-6′O-benzenesulfonate sphysicochemical property. Acta Univ Med Anhui. 2014;49:202–5.
Chang Y, Wei W, Zhang L, Xu HM. Effects and mechanisms of total glucosides of paeony on synoviocytes activities in rat collagen-induced arthritis. J Ethnopharmacol. 2009;121:43–48.
Chang Y, Zhang L, Wang C, Jia XY, Wei W. Paeoniflorin inhibits function of synoviocytes pretreated by rIL-1alpha and regulates EP4 receptor expression. J Ethnopharmacol. 2011;137:1275–82.
Wang C, Yuan J, Zhang LL, Wei W. Pharmacokinetic comparisons of Paeoniflorin and Paeoniflorin-6′-O-benzenesulfonate in rats via different routes of administration. Xenobiotica. 2016;46:1142–50.
Li Y, Sheng K, Chen J, Wu Y, Zhang F, Chang Y, et al. Regulation of PGE2 signaling pathways and TNF-alpha signaling pathways on the function of bone marrow-derived dendritic cells and the effects of CP-25. Eur J Pharmacol. 2015;769:8–21. https://doi.org/10.1016/j.ejphar.2015.09.036.
Yang XD, Wang C, Zhou P, Yu J, Asenso J, Ma Y, et al. Absorption characteristic of paeoniflorin-6′-O-benzene sulfonate (CP-25) in in situ single-pass intestinal perfusion in rats. Xenobiotica. 2016;46:775–83.
Chang Y, Jia XY, Wei F, Wang C, Sun XJ, Xu S, et al. CP-25, a novel compound, protects against autoimmune arthritis by modulating immune mediators of inflammation and bone damage. Sci Rep. 2016;6:26239 https://doi.org/10.1038/srep26239.
Wang QT, Wu YJ, Huang B, Ma YK, Song SS, Zhang LL, et al. Etanercept attenuates collagen-induced arthritis by modulating the association between BAFFR expression and the production of splenic memory B cells. Pharmacol Res. 2013;68:38–45.
Conigliaro P, Triggianese P, Perricone C, Chimenti MS, Di Muzio G, Ballanti E, et al. Restoration of peripheral blood natural killer and B cell levels in patients affected by rheumatoid andpsoriatic arthritis during Etanercept treatment. Clin Exp Immunol. 2014;177:234–43.
Daridon C, Burmester GR, Dörner T. Anticytokine therapy impacting on B cells in autoimmune diseases. Curr Opin Rheumatol. 2009;21:205–10.
Wang D, Chang Y, Wu Y, Zhang L, Yan S, Xie G, et al. Therapeutic effects of TACI-Ig on rat with adjuvant arthritis. Clin Exp Immunol. 2011;163:225–34.
Kojima T, Ishikawa H, Tanaka S, Haga N, Nishida K, Yukioka M, et al. Characteristics of functional impairment in patients with long-standing rheumatoid arthritis based on range of motion of joints: baseline data from a multicenter prospective observational cohort study to evaluate the effectiveness of joint surgery in the treat-to-target era. Modernrheumatology. 2017;1−8. https://doi.org/10.1080/14397595.
Fekete A, Soos L, Szekanecz Z, Szabo Z, Szodoray P, Barath S, et al. Disturbances in B- and T-cell homeostasis in rheumatoid arthritis: suggested relationships with antigen-driven immune responses. J Autoimmun 2007;29:154–63.
Zheng B, Zhang X, Guo L, Han S. IgM plays an important role in induction of collagen-induced arthritis. Clin Exp Immunol. 2007;149:579–85.
Khare SD, Sarosi I, Xia XZ, McCabe S, Miner K, Solovyev I, et al. Severe B cell hyperplasia and autoimmune disease in TALL-1 transgenic mice. Proc Natl Acad Sci U S A. 2000;97:3370–5.
Jia X, Wei F, Sun X, Chang Y, Xu S, Yang X, et al. CP-25 attenuates the inflammatory response of fibroblast-like synoviocytes co-cultured with BAFF-activated CD4+T cells. J Ethnopharmacol. 2016;189:194–201. https://doi.org/10.1016/j.jep.2016.05.034.
Nguyen TG, Morris JM. Signals from activation of B-cell receptor with anti-IgD can override the stimulatory effects of excess BAFF on mature B cells in vivo. Immunol Lett. 2014;161:157–64.
Chiu YM, Lang HC, Lin HY, Yang MT, Fang CH, Yang YW, et al. Risk of tuberculosis, serious infection and lymphoma with disease-modifying biologic drugs in rheumatoid arthritis patients in Taiwan. Int J Rheum Dis. 2014;9(Suppl 3):9–19. https://doi.org/10.1111/1756-185X.12539.
Davies R, Southwood TR, Kearsley-Fleet L, Lunt M, Hyrich KL. Medically significant infections are increased in patients with juvenile idiopathic arthritis treated with Etanercept: results from the British Society for Paediatric and Adolescent Rheumatology Etanercept Cohort Study. Arthritis Rheumatol. 2015;67:2487–94.
Fleischmann R, Kremer J, Cush J, Schulze-Koops H, Connell CA, Bradley JD, et al. Placebo-controlled trial of Tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med. 2012;367:495–507.
Atzeni F, Batticciotto A, Masala IF, Talotta R, Benucci M, Sarzi-Puttini P. Infections and biological therapy in patients with rheumatic diseases. Isr Med Assoc J. 2016;18:164–7.
Yun H, Xie F, Delzell E, Levitan EB, Chen L, Lewis JD, et al. Comparative risk of hospitalized infection a68(1):ted with biologic agents in rheumatoid arthritis patients enrolled in medicare. Arthritis Rheumatol. 2016;68:56–66.
Wei W. Soft regulation of inflammatory immune responses. Chin Pharmacol Bull. 2016;32:297–303.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant numbers 81330081, 81473223, and 81673444) and by the Anhui Province Postdoctoral Science Foundation (grant number 2016B134).
Author information
Authors and Affiliations
Contributions
J-lS, X-zZ, and LH conducted the study, wrote this paper and analyzed the data. CW, X-yT, YT, Y-jW, Q-tW, FZ, J-yC, YC, H-xW, L-lZ and WW analyzed the data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Shu, Jl., Zhang, Xz., Han, L. et al. Paeoniflorin-6′-O-benzene sulfonate alleviates collagen-induced arthritis in mice by downregulating BAFF-TRAF2-NF-κB signaling: comparison with biological agents. Acta Pharmacol Sin 40, 801–813 (2019). https://doi.org/10.1038/s41401-018-0169-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-018-0169-5
Keywords
This article is cited by
-
Deficiency of β-arrestin2 exacerbates inflammatory arthritis by facilitating plasma cell formation
Acta Pharmacologica Sinica (2021)
-
CP-25, a compound derived from paeoniflorin: research advance on its pharmacological actions and mechanisms in the treatment of inflammation and immune diseases
Acta Pharmacologica Sinica (2020)