Abstract
Background
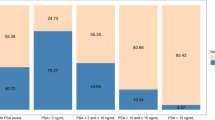
Prostate cancer (PCa) is the most common cancer in North American men. Beyond the established contribution of androgens to disease progression, growing evidence suggest that oestrogen-related pathways might also be of clinical importance. The aim of this study was to explore the association of urinary oestrogen levels with clinical outcomes.
Methods
Urine samples from the prospective multi-institutional PROCURE cohort were collected before RP for discovery (n = 259) and validation (n = 253). Urinary total oestrogens (unconjugated + conjugated), including oestrone and oestradiol, their bioactive and inactive catechol and methyl derivatives (n = 15), were measured using mass spectrometry (MS).
Results
The median follow-up time for the discovery and replication cohorts was 7.6 and 6.5 years, respectively. Highly significant correlations between urinary oestrogens were observed; however, correlations with circulating oestrogens were modest. Our findings indicate that higher levels of urinary oestriol and 16-ketoestradiol were associated with lower risk of BCR. In contrast, higher levels of 2-methoxyestrone were associated with an increased risk of development of metastasis/deaths.
Conclusions
Our data suggest that urinary levels of oestriol and 16-ketoestradiol metabolites are associated with a more favourable outcome, whereas those of 2-methoxyestrone are associated with an elevated risk of metastasis after RP. Further studies are required to better understand the impact of oestrogens on disease biology and as easily accessible urine-based risk-stratification markers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Miller, K. D., Nogueira, L., Mariotto, A. B., Rowland, J. H., Yabroff, K. R., Alfano, C. M. et al. Cancer treatment and survivorship statistics, 2019. CA Cancer J. Clin. 69, 363–385 (2019).
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A. & Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Pishgar, F., Ebrahimi, H., Saeedi Moghaddam, S., Fitzmaurice, C. & Amini, E. Global, regional and national burden of prostate cancer, 1990 to 2015: results from the Global Burden of Disease Study 2015. J. Urol. 199, 1224–1232 (2018).
Lorent, M., Maalmi, H., Tessier, P., Supiot, S., Dantan, E. & Foucher, Y. Meta-analysis of predictive models to assess the clinical validity and utility for patient-centered medical decision making: application to the cancer of the prostate risk assessment (CAPRA). BMC Med. Inf. Decis. Mak. 19, 2 (2019).
Fraser, M., Sabelnykova, V. Y., Yamaguchi, T. N., Heisler, L. E., Livingstone, J., Huang, V. et al. Genomic hallmarks of localized, non-indolent prostate cancer. Nature 541, 359–364 (2017).
Pound, C. R., Partin, A. W., Eisenberger, M. A., Chan, D. W., Pearson, J. D. & Walsh, P. C. Natural history of progression after PSA elevation following radical prostatectomy. J. Am. Med. Assoc. 281, 1591–1597 (1999).
Audet-Walsh, E., Bellemare, J., Lacombe, L., Fradet, Y., Fradet, V., Douville, P. et al. The impact of germline genetic variations in hydroxysteroid (17-beta) dehydrogenases on prostate cancer outcomes after prostatectomy. Eur. Urol. 62, 88–96 (2012).
Audet-Walsh, E., Bellemare, J., Nadeau, G., Lacombe, L., Fradet, Y., Fradet, V. et al. SRD5A polymorphisms and biochemical failure after radical prostatectomy. Eur. Urol. 60, 1226–1234 (2011).
Huggins, C. & Clark, P. J. Quantitative studies of prostatic secretion: II. the effect of castration and of estrogen injection on the normal and on the hyperplastic prostate glands of dogs. J. Exp. Med. 72, 747–762 (1940).
Schileyko, A. A. & Solovyeva, E. N. On the taxonomic position of the enigmatic genus Tonkinodentus Schileyko, 1992 (Chilopoda, Scolopendromorpha): the first molecular data. Zookeys 840, 133–155 (2019).
Ryan, C. J., Smith, M. R., de Bono, J. S., Molina, A., Logothetis, C. J., de Souza, P. et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N. Engl. J. Med. 368, 138–148 (2013).
Ryan, C. J., Smith, M. R., Fizazi, K., Saad, F., Mulders, P. F., Sternberg, C. N. et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 16, 152–160 (2015).
Beer, T. M., Armstrong, A. J., Rathkopf, D., Loriot, Y., Sternberg, C. N., Higano, C. S. et al. Enzalutamide in men with chemotherapy-naive metastatic castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur. Urol. 71, 151–154 (2017).
Scher, H. I., Fizazi, K., Saad, F., Taplin, M. E., Sternberg, C. N., Miller, K. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 367, 1187–1197 (2012).
Bosland, M. C., Ford, H. & Horton, L. Induction at high incidence of ductal prostate adenocarcinomas in NBL/Cr and Sprague-Dawley Hsd:SD rats treated with a combination of testosterone and estradiol-17 beta or diethylstilbestrol. Carcinogenesis 16, 1311–1317 (1995).
Bosland, M. C. & Mahmoud, A. M. Hormones and prostate carcinogenesis: androgens and estrogens. J. Carcinog. 10, 33 (2011).
McPherson, S. J., Ellem, S. J. & Risbridger, G. P. Estrogen-regulated development and differentiation of the prostate. Differentiation 76, 660–670 (2008).
McPherson, S. J., Wang, H., Jones, M. E., Pedersen, J., Iismaa, T. P., Wreford, N. et al. Elevated androgens and prolactin in aromatase-deficient mice cause enlargement, but not malignancy, of the prostate gland. Endocrinology 142, 2458–2467 (2001).
Weng, C., Cai, J., Wen, J., Yuan, H., Yang, K., Imperato-McGinley, J. et al. Differential effects of estrogen receptor ligands on regulation of dihydrotestosterone-induced cell proliferation in endothelial and prostate cancer cells. Int. J. Oncol. 42, 327–337 (2013).
Yu, L., Shi, J., Cheng, S., Zhu, Y., Zhao, X., Yang, K. et al. Estrogen promotes prostate cancer cell migration via paracrine release of ENO1 from stromal cells. Mol. Endocrinol. 26, 1521–1530 (2012).
Yang, L., Gaikwad, N. W., Meza, J., Cavalieri, E. L., Muti, P., Trock, B. et al. Novel biomarkers for risk of prostate cancer: results from a case-control study. Prostate 69, 41–48 (2009).
Ellem, S. J., Schmitt, J. F., Pedersen, J. S., Frydenberg, M. & Risbridger, G. P. Local aromatase expression in human prostate is altered in malignancy. J. Clin. Endocrinol. Metab. 89, 2434–2441 (2004).
Montgomery, R. B., Mostaghel, E. A., Vessella, R., Hess, D. L., Kalhorn, T. F., Higano, C. S. et al. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 68, 4447–4454 (2008).
Ross, R. W., Oh, W. K., Xie, W., Pomerantz, M., Nakabayashi, M., Sartor, O. et al. Inherited variation in the androgen pathway is associated with the efficacy of androgen-deprivation therapy in men with prostate cancer. J. Clin. Oncol. 26, 842–847 (2008).
Adams, M. J., Johnson, S. A., Lefevre, P., Levesque, V., Hayward, V., Andre, T. et al. Finger pad friction and its role in grip and touch. J. R. Soc. Interface 10, 20120467 (2013).
Duan, L., Chen, Z., Lu, J., Liang, Y., Wang, M., Roggero, C. M. et al. Histone lysine demethylase KDM4B regulates the alternative splicing of the androgen receptor in response to androgen deprivation. Nucleic Acids Res. 47, 11623–11636 (2019).
Cussenot, O., Azzouzi, A. R., Nicolaiew, N., Fromont, G., Mangin, P., Cormier, L. et al. Combination of polymorphisms from genes related to estrogen metabolism and risk of prostate cancers: the hidden face of estrogens. J. Clin. Oncol. 25, 3596–3602 (2007).
Zhu, B. T., Han, G. Z., Shim, J. Y., Wen, Y. & Jiang, X. R. Quantitative structure-activity relationship of various endogenous estrogen metabolites for human estrogen receptor alpha and beta subtypes: Insights into the structural determinants favoring a differential subtype binding. Endocrinology 147, 4132–4150 (2006).
Zhu, B. T. & Conney, A. H. Is 2-methoxyestradiol an endogenous estrogen metabolite that inhibits mammary carcinogenesis? Cancer Res. 58, 2269–2277 (1998).
Zhu, B. T. & Conney, A. H. Functional role of estrogen metabolism in target cells: review and perspectives. Carcinogenesis 19, 1–27 (1998).
Han, D. F., Zhou, X., Hu, M. B., Xie, W., Mao, Z. F., Chen, D. E. et al. Polymorphisms of estrogen-metabolizing genes and breast cancer risk: a multigenic study. Chin. Med. J. 118, 1507–1516 (2005).
Cavalieri, E. L. & Rogan, E. G. Unbalanced metabolism of endogenous estrogens in the etiology and prevention of human cancer. J. Steroid Biochem. Mol. Biol. 125, 169–180 (2011).
Lee, M. M., Gomez, S. L., Chang, J. S., Wey, M., Wang, R. T. & Hsing, A. W. Soy and isoflavone consumption in relation to prostate cancer risk in China. Cancer Epidemiol. Biomark. Prev. 12, 665–668 (2003).
Saeed, M., Rogan, E. & Cavalieri, E. Mechanism of metabolic activation and DNA adduct formation by the human carcinogen diethylstilbestrol: the defining link to natural estrogens. Int. J. Cancer 124, 1276–1284 (2009).
Cavalieri, E. & Rogan, E. Catechol quinones of estrogens in the initiation of breast, prostate, and other human cancers: keynote lecture. Ann. N. Y. Acad. Sci. 1089, 286–301 (2006).
Saeed, M., Gunselman, S. J., Higginbotham, S., Rogan, E. & Cavalieri, E. Formation of the depurinating N3adenine and N7guanine adducts by reaction of DNA with hexestrol-3’,4’-quinone or enzyme-activated 3’-hydroxyhexestrol. Implications for a unifying mechanism of tumor initiation by natural and synthetic estrogens. Steroids 70, 37–45 (2005).
Cavalieri, E., Frenkel, K., Liehr, J. G., Rogan, E. & Roy, D. Estrogens as endogenous genotoxic agents–DNA adducts and mutations. J. Natl. Cancer Inst. Monogr. 27, 75–93 (2000).
Sissung, T. M., Danesi, R., Price, D. K., Steinberg, S. M., de Wit, R., Zahid, M. et al. Association of the CYP1B1*3 allele with survival in patients with prostate cancer receiving docetaxel. Mol. Cancer Ther. 7, 19–26 (2008).
Markushin, Y., Gaikwad, N., Zhang, H., Kapke, P., Rogan, E. G., Cavalieri, E. L. et al. Potential biomarker for early risk assessment of prostate cancer. Prostate 66, 1565–1571 (2006).
Rogan, E. G. & Cavalieri, E. L. Estrogen metabolites, conjugates, and DNA adducts: possible biomarkers for risk of breast, prostate, and other human cancers. Adv. Clin. Chem. 38, 135–149 (2004).
Cavalieri, E. L., Devanesan, P., Bosland, M. C., Badawi, A. F. & Rogan, E. G. Catechol estrogen metabolites and conjugates in different regions of the prostate of Noble rats treated with 4-hydroxyestradiol: implications for estrogen-induced initiation of prostate cancer. Carcinogenesis 23, 329–333 (2002).
D’Amato, R. J., Lin, C. M., Flynn, E., Folkman, J. & Hamel, E. 2-Methoxyestradiol, an endogenous mammalian metabolite, inhibits tubulin polymerization by interacting at the colchicine site. Proc. Natl Acad. Sci. USA 91, 3964–3968 (1994).
Fotsis, T., Zhang, Y., Pepper, M. S., Adlercreutz, H., Montesano, R., Nawroth, P. P. et al. The endogenous oestrogen metabolite 2-methoxyoestradiol inhibits angiogenesis and suppresses tumour growth. Nature 368, 237–239 (1994).
Lakhani, N. J., Sarkar, M. A., Venitz, J. & Figg, W. D. 2-Methoxyestradiol, a promising anticancer agent. Pharmacotherapy 23, 165–172 (2003).
Zhu, B. T. & Liehr, J. G. Inhibition of the catechol-O-methyltransferase-catalyzed O-methylation of 2- and 4-hydroxyestradiol by catecholamine: implications for the mechanism of estrogen-induced carcinogenesis. Arch. Biochem. Biophys. 304, 248–256 (1993).
Lévesque E., Caron, P., Lacombe, L., Turcotte, V., Simonyan, D., Fradet, Y. et al. comprehensive analysis of steroid hormones and progression of localized high-risk prostate cancer. Cancer Epidemiol. Biomark. Prev. 28, 701–706 (2019).
Brimo, F., Aprikian, A., Latour, M., Tetu, B., Doueik, A., Scarlata, E. et al. Strategies for biochemical and pathologic quality assurance in a large multi-institutional biorepository; the experience of the PROCURE Quebec Prostate Cancer Biobank. Biopreserv Biobank. 11, 285–290 (2013).
Levesque, E., Caron, P., Lacombe, L., Turcotte, V., Simonyan, D., Fradet, Y. et al. A comprehensive analysis of steroid hormones and progression of localized high-risk prostate cancer. Cancer Epidemiol. Biomark. Prev. 28, 701–706 (2019).
Wissing, M., Chevalier, S., McKercher, G., Laprise, C., Aprikian, S., O’Flaherty, A. et al. The relationship between body-mass index, physical activity, and pathologic and clinical outcomes after radical prostatectomy for prostate cancer. World J. Urol. 37, 789–798 (2019).
Audet-Delage, Y., Gregoire, J., Caron, P., Turcotte, V., Plante, M., Ayotte, P. et al. Estradiol metabolites as biomarkers of endometrial cancer prognosis after surgery. J. Steroid Biochem. Mol. Biol. 178, 45–54 (2018).
Franke, A. A., Custer, L. J., Morimoto, Y., Nordt, F. J. & Maskarinec, G. Analysis of urinary estrogens, their oxidized metabolites, and other endogenous steroids by benchtop orbitrap LCMS versus traditional quadrupole GCMS. Anal. Bioanal. Chem. 401, 1319–1330 (2011).
Petrick, J. L., Hyland, P. L., Caron, P., Falk, R. T., Pfeiffer, R. M., Dawsey, S. M. et al. Associations between prediagnostic concentrations of circulating sex steroid hormones and esophageal/gastric cardia adenocarcinoma among men. J. Natl Cancer Inst. 111, 34–41 (2019).
Eliassen, A. H., Missmer, S. A., Tworoger, S. S. & Hankinson, S. E. Endogenous steroid hormone concentrations and risk of breast cancer: does the association vary by a woman’s predicted breast cancer risk? J. Clin. Oncol. 24, 1823–1830 (2006).
Dobbs, R. W., Malhotra, N. R., Greenwald, D. T., Wang, A. Y., Prins, G. S. & Abern, M. R. Estrogens and prostate cancer. Prostate Cancer Prostatic Dis. 22, 59–65 (2019).
Cavalieri, E. L. & Rogan, E. G. Depurinating estrogen-DNA adducts, generators of cancer initiation: their minimization leads to cancer prevention. Clin. Transl. Med. 5, 12 (2016).
Toren, P., Hoffman, A., Ding, K., Joncas, F. H., Turcotte, V., Caron, P. et al. Serum sex steroids as prognostic biomarkers in patients receiving androgen deprivation therapy for recurrent prostate cancer: a post hoc analysis of the PR.7 trial. Clin. Cancer Res. 24, 5305–5312 (2018).
Di Zazzo, E., Galasso, G., Giovannelli, P., Di Donato, M. & Castoria, G. Estrogens and their receptors in prostate cancer: therapeutic implications. Front. Oncol. 8, 2 (2018).
Levesque, E., Laverdiere, I., Audet-Walsh, E., Caron, P., Rouleau, M., Fradet, Y. et al. Steroidogenic germline polymorphism predictors of prostate cancer progression in the estradiol pathway. Clin. Cancer Res. 20, 2971–2983 (2014).
Salonia, A., Abdollah, F., Capitanio, U., Suardi, N., Briganti, A., Gallina, A. et al. Serum sex steroids depict a nonlinear u-shaped association with high-risk prostate cancer at radical prostatectomy. Clin. Cancer Res. 18, 3648–3657 (2012).
Muti, P., Westerlind, K., Wu, T., Grimaldi, T., De Berry, J. 3rd, Schunemann, H. et al. Urinary estrogen metabolites and prostate cancer: a case-control study in the United States. Cancer Causes Control 13, 947–955 (2002).
Barba, M., Yang, L., Schunemann, H. J., Sperati, F., Grioni, S., Stranges, S. et al. Urinary estrogen metabolites and prostate cancer: a case-control study and meta-analysis. J. Exp. Clin. Cancer Res. 28, 135 (2009).
Teas, J., Cunningham, J. E., Fowke, J. H., Nitcheva, D., Kanwat, C. P., Boulware, R. J. et al. Urinary estrogen metabolites, prostate specific antigen, and body mass index among African-American men in South Carolina. Cancer Detect. Prev. 29, 494–500 (2005).
Kosti, O., Xu, X., Veenstra, T. D., Hsing, A. W., Chu, L. W., Goldman, L. et al. Urinary estrogen metabolites and prostate cancer risk: a pilot study. Prostate 71, 507–516 (2011).
Schumacher, G. & Neuhaus, P. The physiological estrogen metabolite 2-methoxyestradiol reduces tumor growth and induces apoptosis in human solid tumors. J. Cancer Res. Clin. Oncol. 127, 405–410 (2001).
Alfaro, A., Leon, A., Guajardo-Correa, E., Reuquen, P., Torres, F., Mery, M. et al. MgO nanoparticles coated with polyethylene glycol as carrier for 2-Methoxyestradiol anticancer drug. PLoS ONE 14, e0214900 (2019).
Acknowledgements
The authors are thankful to all participating patients and staff at each site associated with the PROCURE Biobank who have made this scientific contribution possible.
Author information
Authors and Affiliations
Contributions
Study concept, design and supervision: E.L. and C.G. Steroid measurements: P.C., V.T. and C.G. Statistical analysis: D.S. Interpretation of the data: J.P.E., L.L., P.C., V.T., D.S., A.A., F.S., M.C., S.C., C.G. and E.L. Drafting of the paper: J.P.E., C.G. and E.L. Critical revision of the paper for important intellectual content: J.P.E., L.L., P.C., V.T., D.S., A.A., F.S., M.C., S.C., C.G. and E.L. Patients’ recruitment and clinical data: F.S., L.L., M.C., S.C. and A.A. Obtaining funding: E.L., C.G. and L.L.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All participants provided written informed consent and the CHU de Quebec research ethics committee approved the research protocol, and the study was performed in accordance with the Declaration of Helsinki.
Consent to publish
Not applicable.
Data availability
The datasets analysed during this study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Funding information
This work was supported by research grants from Prostate Cancer Canada (D2013-22 to E.L., C.G. and L.L.), the Cancer Research Society (to C.G.) and the Canada Research Chair Program (to C.G.). E.L. holds a CIHR Clinician-Scientist Award and was the holder of the PCC rising star award (RS2013-59). C.G. holds the Canada Research Chair in Pharmacogenomics (Tier I). The results are based on samples and patient data obtained from the PROCURE Biobank, supported by donations in a partnership with the Cancer Research Society of Canada.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Emond, JP., Lacombe, L., Caron, P. et al. Urinary oestrogen steroidome as an indicator of the risk of localised prostate cancer progression. Br J Cancer 125, 78–84 (2021). https://doi.org/10.1038/s41416-021-01376-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01376-z
This article is cited by
-
Extensive metabolic consequences of human glycosyltransferase gene knockouts in prostate cancer
British Journal of Cancer (2023)