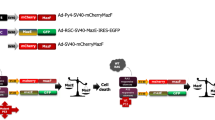
Abstract
Delivery of RNAi-mediating shRNA molecules for gene silencing via bacteria, i.e. by transkingdom RNAi (tkRNAi) technology, is suggested to be a powerful alternative technique. In this work, the efficiency of bacterial delivery of shRNAs directed against HPV16-E7-specific mRNA to oral squamous carcinoma cells (OSCCs) was evaluated. E. coli were transfected with a plasmid encoding the inv locus and the Hlya gene to enable the bacteria to enter carcinoma cells and to escape from endocytotic vesicles. The bacterial penetration to the target cells was confirmed by DAPI staining. The HPV16-E7 mRNA expression in bacteria-treated OSCCs dropped to 61% of the controls as measured by qRT-PCR. Corresponding inhibition of the HPV16-E7 protein was confirmed by western blotting. The IC50 of bacteria-treated OSCCs was reduced to more than 75%. Flow cytometry assays showed higher total apoptosis and caspase-3 activation (6.6-fold and 8.4-fold respectively) in OSCCs following exposure to anti-HPV-E7 bacteria compared to anti-GFP bacteria (2-fold and 2.9-fold, respectively). In conclusion, it was demonstrated for the first time that tkRNAi technology is also useful for treatment of squamous carcinoma cells. Anti-HPV16-E7 shRNA-encoding bacteria can efficiently deliver RNAi effectors to OSCCs mediating a strong and specific gene silencing associated with triggering cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Yu T, Wu Y, Helman JI, Wen Y, Wang C, Li L. CXCR4 promotes oral squamous cell carcinoma migration and invasion through inducing expression of MMP-9 and MMP-13 via the ERK signaling pathway. Mol Cancer Res. 2011;9:161–72.
Chi AC, Day TA, Neville BW. Oral cavity and oropharyngeal squamous cell carcinoma—an update. CA Cancer J Clin. 2015;65:401–21.
Chaturvedi AK, Engels EA, Pfeiffer RM, Hernandez BY, Xiao W, Kim E, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J Clin Oncol. 2011;29:4294–301.
McMurray HR, Nguyen D, Westbrook TF, McAnce DJ. Biology of human papillomaviruses. Int J Exp Pathol. 2001;82:15–33.
Jung HS, Rajasekaran N, Ju W, Shin YK. Human papillomavirus: current and future RNAi therapeutic strategies for cervical cancer. J Clin Med. 2015;4:1126–55.
Charette ST, McCance DJ. The E7 protein from human papillomavirus type 16 enhances keratinocyte migration in an Akt-dependent manner. Oncogene. 2007;26:7386–90.
Boulenouar S, Weyn C, Van Noppen M, Moussa Ali M, Favre M, Delvenne Po, et al. Effects of HPV-16 E5, E6 and E7 proteins on survival, adhesion, migration and invasion of trophoblastic cells. Carcinogenesis. 2010;31:473–80.
Jiang M, Milner J. Selective silencing of viral gene expression in HPV-positive human cervical carcinoma cells treated with siRNA, a primer of RNA interference. Oncogene. 2002;21:6041–8.
Liu YC, Cai ZM, Zhang XJ. Reprogrammed CRISPR-Cas9 targeting the conserved regions of HPV6/11 E7 genes inhibits proliferation and induces apoptosis in E7-transformed keratinocytes. Asian J Androl. 2016;18:475–9.
Hu J, Ge W, Xu J. HPV 16 E7 inhibits OSCC cell proliferation, invasion, and metastasis by upregulating the expression of miR-20a. Tumour Biol. 2016;37:9433–40.
LaRocca CJ, Han J, Salzwedel AO, Davydova J, Herzberg MC, Gopalakrishnan R, et al. Oncolytic adenoviruses targeted to human papilloma virus-positive head and neck squamous cell carcinomas. Oral Oncol. 2016;56:25–31.
Lage H. Therapeutic potential of RNA interference in drug-resistant cancers. Future Oncol. 2009;5:169–85.
Zhou J, Shum KT, Burnett JC, Rossi JJ. Nanoparticle-based delivery of RNAi therapeutics: progress and challenges. Pharmaceuticals (Basel). 2013;6:85–107.
Wang T, Shigdar S, Shamaileh HA, Gantier MP, Yin W, Xiang D, et al. Challenges and opportunities for siRNA-based cancer treatment. Cancer Lett. 2017;387:77–83.
Aagaard L, Rossi JJ. RNAi therapeutics: principles, prospects and challenges. Adv Drug Deliv Rev. 2007;59:75–86.
Deng Y, Wang CC, Choy KW, Du Q, Chen J, Wang Q, et al. Therapeutic potentials of gene silencing by RNA interference: principles, challenges, and new strategies. Gene. 2014;538:217–27.
Xiang S, Fruehauf J, Li CJ. Short hairpin RNA-expressing bacteria elicit RNA interference in mammals. Nat Biotechnol. 2006;24:697–702.
Lage H, Fruehauf JH. Delivery of therapeutic RNA molecules to cancer cells by bacteria. Ther Deliv. 2011;2:441–9.
Pogorzelski M, Ting S, Gauler TC, Breitenbuecher F, Vossebein I, Hoffarth S, et al. Impact of human papilloma virus infection on the response of head and neck cancers to anti-epidermal growth factor receptor antibody therapy. Cell Death Dis. 2014;5:e1091.
Ahmed O, Krühn A, Lage H. Delivery of siRNAs to cancer cells via bacteria. Methods Mol Biol. 2015;1218:117–29.
Chang JT, Kuo TF, Chen YJ, Chiu CC, Lu YC, Li HF, et al. Highly potent and specific siRNAs against E6 or E7 genes of HPV16- or HPV18-infected cervical cancers. Cancer Gene Ther. 2010;17:827–36.
Priebsch A, Rompe F, Tönnies H, Kowalski P, Surowiak P, Stege A, et al. Complete reversal of ABCG2-depending atypical multidrug resistance by RNA interference in human carcinoma cells. Oligonucleotides. 2006;16:263–74.
Krühn A, Wang A, Fruehauf JH, Lage H. Delivery of short hairpin RNAs by transkingdom RNA interference modulates the classical ABCB1-mediated multidrug-resistant phenotype of cancer cells. Cell Cycle. 2009;8:3349–54.
Vichai V, Kirtikara K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat Protoc. 2006;1:1112–6.
Guo H, Zhang J, Inal C, Nguyen T, Fruehauf JH, Keates AC, et al. Targeting tumor gene by shRNA-expressing Salmonella-mediated RNAi. Gene Ther. 2011;18:95–105.
Acknowledgements
This work was supported in part by “Deutscher Akademischer Austauschdienst” (DAAD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ahmed, O.B., Lage, H. Bacteria-mediated delivery of RNAi effector molecules against viral HPV16-E7 eradicates oral squamous carcinoma cells (OSCC) via apoptosis. Cancer Gene Ther 26, 166–173 (2019). https://doi.org/10.1038/s41417-018-0054-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-018-0054-x