Abstract
Background
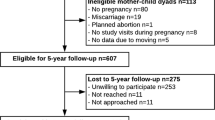
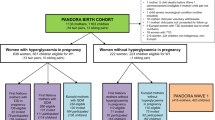
High glycemic index (GI) and glycemic load (GL) as indicators of carbohydrate quality and quantity have been found to increase risk of metabolic outcomes in adults. Whether carbohydrate quality may influence metabolic programming already in early life is unknown. We examined the association of maternal GI and GL with offspring body mass index (BMI) in the first 7 years of life among 68,471 mother–offspring dyads from the Danish National Birth Cohort (DNBC). In a sub-cohort of offspring with clinical data (n = 1234) that included 608 dyads exposed to gestational diabetes mellitus (GDM), we also examined the relation to metabolic health at 9–16 years.
Methods
Maternal GI and GL were quantified using a mid-pregnancy food frequency questionnaire. We used birth weight and length to calculate offspring’s ponderal index. Age- and sex-specific BMI z scores at 5 mo, 12 mo, and 7 y were standardized against WHO reference data. In the clinical cohort, we quantified body composition, HOMA-IR, and HOMA-B. We used multivariable mixed linear and Poisson regression to model the associations.
Results
Median (IQR) of GI and GL were 83 (63–111) and 241 (180–333) g/day, respectively. We found that GI (Q4 vs. Q1:1.09, 95%CI: 1.03, 1.15) and GL (Q4 vs. Q1:1.10, 95%CI: 1.05, 1.16) modestly increased the relative risk of large-for gestational age (LGA). In the clinical sub-cohort, we observed a potential increase in offspring HOMA-IR, adiposity, and metabolic syndrome z score with higher maternal GI and GI. These associations were stronger among the GDM-exposed offspring, but the CI included the null value.
Conclusion
We found associations of GI and GL in pregnancy with offspring LGA. Potential long-term benefits to offspring exposed to GDM need to be confirmed in larger, well-powered studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Roseboom T, de Rooij S, Painter R. The Dutch famine and its long-term consequences for adult health. Early Hum Dev. 2006;82:485–91.
Dabelea D, Mayer-Davis EJ, Saydah S, Imperatore G, Linder B, Divers J, et al. Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA. 2014;311:1778–86.
Dehghan M, Akhtar-Danesh N, Merchant AT. Childhood obesity, prevalence and prevention. Nutr J. 2005;4:24.
Lobstein T, Baur L, Uauy R. Obesity in children and young people: a crisis in public health. Obes Rev. 2004;5(Suppl 1):4–104.
Tzanetakou IP, Mikhailidis DP, Perrea DN. Nutrition during pregnancy and the effect of carbohydrates on the offspring’s metabolic profile: in search of the “perfect maternal diet”. Open Cardiovasc Med J. 2011;5:103–9.
Wolever TM, Jenkins DJ, Jenkins AL, Josse RG. The glycemic index: methodology and clinical implications. Am J Clin Nutr. 1991;54:846–54.
Hu FB, Manson JE, Stampfer MJ, Colditz G, Liu S, Solomon CG, et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med. 2001;345:790–7.
Hu FB, van Dam RM, Liu S. Diet and risk of Type II diabetes: the role of types of fat and carbohydrate. Diabetologia. 2001;44:805–17.
Schulze MB, Manson JE, Ludwig DS, Colditz GA, Stampfer MJ, Willett WC, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927–34.
Willett W, Manson J, Liu S. Glycemic index, glycemic load, and risk of type 2 diabetes. Am J Clin Nutr. 2002;76:274s–80s.
Fung TT, Hu FB, Pereira MA, Liu S, Stampfer MJ, Colditz GA, et al. Whole-grain intake and the risk of type 2 diabetes: a prospective study in men. Am J Clin Nutr. 2002;76:535–40.
Salmeron J, Ascherio A, Rimm EB, Colditz GA, Spiegelman D, Jenkins DJ, et al. Dietary fiber, glycemic load, and risk of NIDDM in men. Diabetes Care. 1997;20:545–50.
Scholl TO, Chen X, Khoo CS, Lenders C. The dietary glycemic index during pregnancy: influence on infant birth weight, fetal growth, and biomarkers of carbohydrate metabolism. Am J Epidemiol. 2004;159:467–74.
Louie JC, Brand-Miller JC, Markovic TP, Ross GP, Moses RG. Glycemic index and pregnancy: a systematic literature review. J Nutr Metab. 2010;2010:282464.
Dabelea D. The predisposition to obesity and diabetes in offspring of diabetic mothers. Diabetes Care. 2007;30(Suppl 2):S169–74.
Catalano PM, Hauguel-De Mouzon S. Is it time to revisit the Pedersen hypothesis in the face of the obesity epidemic? Am J Obstet Gynecol. 2011;204:479–87.
Olsen J, Melbye M, Olsen SF, Sorensen TI, Aaby P, Andersen AM, et al. The Danish National Birth Cohort—its background, structure and aim. Scand J Public Health. 2001;29:300–7.
Zhang C, Hu FB, Olsen SF, Vaag A, Gore-Langton R, Chavarro JE, et al. Rationale, design, and method of the Diabetes & Women’s Health study—a study of long-term health implications of glucose intolerance in pregnancy and their determinants. Acta Obstet Gynecol Scand. 2014;93:1123–30.
Grunnet LG, Hansen S, Hjort L, Madsen CM, Kampmann FB, Thuesen ACB, et al. Adiposity, dysmetabolic traits, and earlier onset of female puberty in adolescent offspring of women with gestational diabetes mellitus: a clinical study within the Danish national birth cohort. Diabetes Care. 2017;40:1746–55.
Olsen SF, Mikkelsen TB, Knudsen VK, Orozova-Bekkevold I, Halldorsson TI, Strom M, et al. Data collected on maternal dietary exposures in the Danish National Birth Cohort. Paediatr Perinat Epidemiol. 2007;21:76–86.
Willett WC. Nutritional Epidemiology. 2nd edn. New York, USA: Oxford University Press; 1998.
Mikkelsen TB, Osler M, Olsen SF. Validity of protein, retinol, folic acid and n-3 fatty acid intakes estimated from the food-frequency questionnaire used in the Danish National Birth Cohort. Public Health Nutr. 2006;9:771–8.
Pryer JA, Vrijheid M, Nichols R, Kiggins M, Elliott P. Who are the ‘low energy reporters’ in the dietary and nutritional survey of British adults? Int J Epidemiol. 1997;26:146–54.
Department of Health Report on Health and Social Subjects. Dietary reference values for food, energy andnutrients for the United Kingdom. London, United Kingdom: HMSO; 1991.
Brion MJ, Ness AR, Rogers I, Emmett P, Cribb V, Davey Smith G, et al. Maternal macronutrient and energy intakes in pregnancy and offspring intake at 10 y: exploring parental comparisons and prenatal effects. Am J Clin Nutr. 2010;91:748–56.
Knudsen VK, Heitmann BL, Halldorsson TI, Sorensen TI, Olsen SF. Maternal dietary glycaemic load during pregnancy and gestational weight gain, birth weight and postpartum weight retention: a study within the Danish National Birth Cohort. Br J Nutr. 2013;109:1471–8.
Foster-Powell K, Holt SH, Brand-Miller JC. International table of glycemic index and glycemic load values: 2002. Am J Clin Nutr. 2002;76:5–56.
Zhu Y, Olsen SF, Mendola P, Yeung EH, Vaag A, Bowers K, et al. Growth and obesity through the first 7 y of life in association with levels of maternal glycemia during pregnancy: a prospective cohort study. Am J Clin Nutr. 2016;103:794–800.
Knudsen LB, Olsen J. The Danish Medical Birth Registry. Dan Med Bull. 1998;45:320–3.
WHO Multicentre Growth Reference Study Group. WHO Child Growth Standards: length/height-for-age,weight-for-age, weight-for length, weight-for-height and body mass index-for-age: methods and development.Geneva, Switzerland: World Health Organization; 2006.
de Onis M, Onyango AW, Borghi E, Siyam A, Nishida C, Siekmann J. Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organ. 2007;85:660–7.
Hulshof KF, Brussaard JH, Kruizinga AG, Telman J, Lowik MR. Socio-economic status, dietary intake and 10 y trends: the Dutch National Food Consumption Survey. Eur J Clin Nutr. 2003;57:128–37.
Oken y, Levitan EB, Gillman MW. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes (2005). 2008;32:201–10.
Reynolds RM, Osmond C, Phillips DI, Godfrey KM. Maternal BMI, parity, and pregnancy weight gain: influences on offspring adiposity in young adulthood. J Clin Endocrinol Metab. 2010;95:5365–9.
Mihalopoulos NL, Holubkov R, Young P, Dai S, Labarthe DR. Expected changes in clinical measures of adiposity during puberty. J Adolesc Health. 2010;47:360–6.
Hopkins SA, Cutfield WS. Exercise in pregnancy: weighing up the long-term impact on the next generation. Exerc Sport Sci Rev. 2011;39:120–7.
Strom M, Mortensen EL, Halldorson TI, Osterdal ML, Olsen SF. Leisure-time physical activity in pregnancy and risk of postpartum depression: a prospective study in a large national birth cohort. J Clin Psychiatry. 2009;70:1707–14.
Bland JM, Altman DG. Multiple significance tests: the Bonferroni method. BMJ (Clin Res Ed). 1995;310:170.
Moses RG, Barker M, Winter M, Petocz P, Brand-Miller JC. Can a low-glycemic index diet reduce the need for insulin in gestational diabetes mellitus? A randomized trial. Diabetes Care. 2009;32:996–1000.
Louie JC, Markovic TP, Perera N, Foote D, Petocz P, Ross GP, et al. A randomized controlled trial investigating the effects of a low-glycemic index diet on pregnancy outcomes in gestational diabetes mellitus. Diabetes Care. 2011;34:2341–6.
Markovic TP, Muirhead R, Overs S, Ross GP, Louie JC, Kizirian N, et al. Randomized controlled trial investigating the effects of a low-glycemic index diet on pregnancy outcomes in women at high risk of gestational diabetes mellitus: The GI Baby 3 Study. Diabetes Care. 2016;39:31–8.
Okubo H, Crozier SR, Harvey NC, Godfrey KM, Inskip HM, Cooper C, et al. Maternal dietary glycemic index and glycemic load in early pregnancy are associated with offspring adiposity in childhood: the Southampton Women’s Survey. Am J Clin Nutr. 2014;100:676–83.
Danielsen I, Granstrom C, Haldorsson T, Rytter D, Hammer Bech B, Henriksen TB, et al. Dietary glycemic index during pregnancy is associated with biomarkers of the metabolic syndrome in offspring at age 20 years. PLoS ONE. 2013;8:e64887.
Bigornia SJ, LaValley MP, Benfield LL, Ness AR, Newby PK. Relationships between direct and indirect measures of central and total adiposity in children: what are we measuring? Obesity (Silver Spring). 2013;21:2055–62.
Brettschneider AK, Ellert U, Schaffrath Rosario A. Comparison of BMI derived from parent-reported height and weight with measured values: results from the German KiGGS study. Int J Environ Res Public Health. 2012;9:632–47.
Oken E. Maternal and child obesity: the causal link. Obstet Gynecol Clin North Am. 2009;36:361–77. ix-x
Schmatz M, Madan J, Marino T, Davis J. Maternal obesity: the interplay between inflammation, mother and fetus. J Perinatol: Off J Calif Perinat Assoc. 2010;30:441–6.
Brantsaeter AL, Haugen M, Alexander J, Meltzer HM. Validity of a new food frequency questionnaire for pregnant women in the Norwegian Mother and Child Cohort Study (MoBa). Matern Child Nutr. 2008;4:28–43.
Erkkola M, Karppinen M, Javanainen J, Rasanen L, Knip M, Virtanen SM. Validity and reproducibility of a food frequency questionnaire for pregnant Finnish women. Am J Epidemiol. 2001;154:466–76.
Fein SB, Labiner-Wolfe J, Shealy KR, Li R, Chen J, Grummer-Strawn LM. Infant Feeding Practices Study II: study methods. Pediatrics. 2008;122(Suppl 2):S28–35.
McGowan CA, Curran S, McAuliffe FM. Relative validity of a food frequency questionnaire to assess nutrient intake in pregnant women. J Human Nutr Diet. 2014;27(Suppl 2):167–74.
Mouratidou T, Ford F, Fraser RB. Validation of a food-frequency questionnaire for use in pregnancy. Public Health Nutr. 2006;9:515–22.
Fawzi WW, Rifas-Shiman SL, Rich-Edwards JW, Willett WC, Gillman MW. Calibration of a semi-quantitative food frequency questionnaire in early pregnancy. Ann Epidemiol. 2004;14:754–62.
Acknowledgements
Funding
Funding for this study was provided by a fellowship grant from the Danish Diabetes Academy supported by the Novo Nordisk Foundation (EM); grants from the Danish Council for Strategic Research (09–067124 (Center for Fetal Programming) and 09–075611); the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health (contract #HHSN275201000020C); the Innovation Fund Denmark (11–115923) and Rigshospitalet, Copenhagen University Hospital.
Author contributions
The authors’ contributions were as follows—EM, AAV, CZ, and SFO: study concept and design; SH, LGG, MS, AAB, LH, FBK, CMM, and ACBT: took part in the planning and conductance of the clinical follow-up study; EM: conducted the statistical analyses and drafted the manuscript; SH, LGG, AAB, LH, FBK, CCM, and CG: contributed to the statistical analyses and methods description; EM, SH, LGG, MS, BHB, TIH, AAV, and CZ: contributed critical advice and revisions of the manuscript; and EM and SFO: responsibility for the entire contents of the manuscript. All authors had full access to study data. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Maslova, E., Hansen, S., Grunnet, L.G. et al. Maternal glycemic index and glycemic load in pregnancy and offspring metabolic health in childhood and adolescence—a cohort study of 68,471 mother–offspring dyads from the Danish National Birth Cohort. Eur J Clin Nutr 73, 1049–1062 (2019). https://doi.org/10.1038/s41430-018-0316-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-018-0316-6
This article is cited by
-
Precision stratification of prognostic risk factors associated with outcomes in gestational diabetes mellitus: a systematic review
Communications Medicine (2024)
-
Association between metabolic syndrome and gestational diabetes mellitus in women and their children: a systematic review and meta-analysis
Endocrine (2021)