Abstract
Objectives
To study the efficacy and the long-term outcomes of treating micro and macrocystic orbital and/or adnexal lymphatic malformations (OA-LM) with protocol-based bleomycin sclerotherapy.
Methods
A retrospective interventional study of 69 eyes having OA-LM treated with non-image guided transcutaneous or transconjunctival bleomycin sclerotherapy (1IU/ml aqueous solution) between December 2014 and December 2018. Based on clinical regression, the outcomes were classified as excellent, good, fair and poor.
Results
The mean age at presentation was 20 ± 16 years (median 16; range 1 month to 70 years). The orbital-palpebral variant was the most common presentation, seen in 29 patients (42%). Microcystic morphology was seen in 34(49%), macrocystic in 22 (32%) and mixed cyst in 13 (19%) patients. Mean units of bleomycin injected per patient were 9 ± 8 IU (median 5.5 IU, range 1–38 IU). Mean number of treatment sessions required were 2 ± 1 (median 2, range 1–6). The response was excellent in 43 (62%), good in 12 (17%), fair in 9 (13%) and poor in 5 (7%) patients. These responses were comparable across the morphological subgroups (p = 0.24, chi-square test). Adverse reactions noted were inflammation in 11 eyes (16%) and peri-ocular pigmentation in 15 (22%). There was a sustained tumour regression over a mean follow-up duration of 3.5 years (median 3; range 1.5–5 years).
Conclusions
Seventy-nine percent of eyes with OA-LM showed a good outcome with transcutaneous and/or transconjunctival non-image guided bleomycin sclerotherapy with no serious adverse events. The results were promising over long-term follow-up.
Similar content being viewed by others
Introduction
Orbital and/or adnexal lymphatic malformations (OA-LM), previously known as lymphangiomas, are benign vascular lesions with a predilection for the head and neck region [1]. These lesions account for ~4% of space occupying lesions in the orbit [2]. They are benign, thin-walled, ill-defined and cystic vascular lesions [3]. The ill-defined and vascular nature of the lesion poses a challenge during surgical debulking [4, 5]. Other treatment options include observation, sclerotherapy and systemic therapy [4,5,6,7,8]. The outcome is dependent on the length of time the lesion has been present, its size, its position in the orbit and its relationship to the other important structures in the orbit, such as the globe, optic nerve or the extraocular muscles. The management of these lesions has seen a paradigm change in recent times, with a shift from surgical debulking to intralesional sclerotherapy with one of the several sclerosing agents [9]. Lymphatic malformations can occur as isolated lymphatic lesions or as complex lymphatic lesions containing lymphatic as well as venous, capillary or arterial components [7]. Sclerotherapy in the presence of a venous or arterial component is contraindicated [10].
Sclerotherapy has gained popularity in the management of the macrocystic component of LM [2, 9,10,11,12,13,14,15,16,17,18,19]. Percutaneous intralesional injection of bleomycin has been shown to be effective in the treatment of macrocystic orbital LM by inducing inflammation and destroying the endothelial cells with resultant fibrosis and thrombosis of vascular channels [20, 21]. However, its role in the microcystic variant of the lesion is unclear [2, 9,10,11,12,13,14,15,16,17,18,19]. In addition, the long term efficacy, percentage regression in the tumour volume with sclerotherapy and the recurrence rates after sclerotherapy have not been reported. We report here our experience with a protocol-based treatment algorithm using bleomycin for sclerotherapy in OA-LM. To the best of our knowledge, this is the largest series reported till date. We also discuss its efficacy in treating OA-LM as a percutaneous intralesional injection, without image guidance, in both the micro and macrocystic subsets of the malformation.
Method
This is a retrospective study of 69 cases of OA-LM treated within the network of a tertiary eye care institute in India. Institutional Review Board approval was obtained for the study and the study adhered to the tenets of the Declaration of Helsinki. The medical records of patients with OA-LM who received bleomycin sclerotherapy between December 2014 and December 2018 were reviewed. Data was collected for particulars of age, gender, presenting symptoms and signs, imaging features, bleomycin dose, clinical response, treatment outcome, complications and follow-up. A comprehensive eye examination was performed in all patients. Hemodynamic tests such as the Valsalva manoeuvre, positional proptosis, palpable thrill and audible bruit were performed to rule out venous varices and high-flow vascular malformations. Pre-operative imaging included Doppler ultrasonography to check the flow within the lesion and orbital MRI/CT with and without Valsalva manoeuvre. Patients underwent either a CT or an MRI based on the affordability of the scan. In these patients, all imaging was non-contrast enhanced as patients with a venous component had been ruled out. The T1 fat saturation MRI sequences were used to look for haemorrhages and T2 to look at the morphology of the lesion. Clinical photographs were taken for all patients prior to the first sitting of sclerotherapy and during each follow-up visit. Based on the orbital imaging characteristics, the lesions were classified into microcystic (cysts <1 cm in diameter), macrocystic (cysts larger than 1 cm in diameter), and mixed cyst (combination of microcystic and macrocystic characteristics). Bleomycin sclerotherapy was planned for all the above morphologic variants.
The following data were collected for each patient: number of treatment sessions, cumulative dose of bleomycin, pre- and post-treatment exophthalmometry (in mm), percentage volume reduction based upon available imaging studies, adverse treatment events and any recurrence of the lesion over the post-treatment follow-up period. All the pre and post-operative imaging was done on the same computed tomography (CT) or a magnetic resonance imaging (MRI) machine (CT:GE 128, MRI: 3 T GE Pioneer). Tumour volume was calculated by an experienced orbital radiologist using a built-in software in the MRI or CT scan device.
Reconstitution of Bleomycin
A commercially available preparation of bleomycin sulphate powder was reconstituted with normal saline to deliver a strength of 1 IU/ml of aqueous bleomycin solution for intralesional injection. Four cc of reconstituted bleomycin (4IU) and 1 cc of 2% lidocaine with adrenaline were mixed in a 5-cc syringe for orbital and adnexal injections.
Dose of sclerosant
The amount of bleomycin injected at any one session was based upon the location of the lesion and our group’s initial experience with the use of this sclerosant in the treatment of LM [3, 22]. Conjunctival lesions and localized palpebral lesions received 2 IU; diffuse palpebral and localized orbital lesions received 4 IU; diffuse orbital lesions received 6 IU and orbital-palpebral lesions received 8 IU of bleomycin. Patients with additional temporal fossa, frontal and cheek LM had additional 2 IU injected into each component of the lesion respectively. None of the patients received a cumulative dose of more than 400 IU of bleomycin [23].
Sclerosant injection
All eyes received non-image guided transcutaneous, transconjunctival or combined intralesional sclerosant injections. Under aseptic precautions, a 24 G needle loaded on a 10-cc syringe was introduced into the lesion and the contents were aspirated. While keeping the tip of the aspiration needle within the cystic lumen, the 10cc syringe was disengaged and the pre-determined dose of bleomycin aqueous solution (1 IU/ml) was injected into the macrocyst using the pre-loaded 5-cc syringe. The technique of injection is as reported for management of retention cysts [24]. For microcystic disease, multiple injections were performed with small aliquots injected into the anatomic region with the microcysts. For patients with eyelid and conjunctival lesions, a 26 G needle was used for the injection of bleomycin. Patients were discharged on the same day on oral paracetamol for 3 days and topical lubricating eye drops for a month.
Treatment protocol
All eyes received non-image guided transcutaneous, transconjunctival or combined sclerosant injection. A flow chart of the treatment protocol is presented in Table 1. Response to sclerotherapy was graded as excellent (>75% clinical regression), good (>50–75% clinical regression), fair (>25–50% clinical regression) and poor (25% or less clinical regression) (Fig. 1). For lesions that were purely orbital, the clinical regression was judged objectively by reduction in the proptosis. The desired response was reduction in the proptosis to within 1 mm of the contralateral normal eye. For lesions with additional palpebral and/or conjunctival components, the clinical outcome assessment was based on a comparison of standard and high-magnification pre- and post-injection photographs, depicting the extent of eyelid involvement and the clock hours of conjunctival involvement. The desired end point of treatment for conjunctival and eyelid components was complete disappearance of the lesion. If any residual lesion was noted on clinical examination, the imaging was repeated after three sittings of intralesional sclerotherapy to look for the areas with residual lesions. If there was a definite continued clinical response, sclerotherapy was continued until the complete disappearance of the lesion or until no further reduction in lesion size was noted. If after three consecutive injections no clinical change was observed when compared to the previous visit or there was excessive resistance while injecting within the lesion (indicative of fibrosis), then the patient was considered to be unresponsive to further sclerotherapy and was scheduled for surgical debulking of the residual disease. Patients with complete tumour regression were followed up at 3-month intervals for the first year and at 6-month intervals thereafter. The radiologic response to treatment was assessed based on post-operative imaging if available. The maximum cumulative dose of bleomycin allowed was 400 IU [23]. The treatment protocol has been highlighted in Table 1.
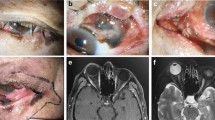
Clinical grading of outcome of bleomycin sclerotherapy as (A–D) excellent, (E–H) good, (I–L) fair and (M–P) poor. A–D Photographs of patient 1 with excellent regression. A, B Pre-bleomycin standard and birds view demonstrating left proptosis, C, D Post-bleomycin standard and bird’s eye view demonstrating >75% regression. E–H Photographs of patient 2 with good regression. E, F Pre-bleomycin standard and bird’s eye view demonstrating right proptosis with hypoglobus and right brow and frontal lesion, G, H Post-bleomycin standard and bird’s eye view demonstrating regression in the right proptosis, hypoglobus and brow lesion by >50–75%. i–l Photograph of patient 3 with fair regression. I, J Pre-bleomycin standard and bird’s eye view demonstrating right proptosis, K, L Post-bleomycin standard and bird’s eye view demonstrating regression in the right proptosis by 25–50%. M–P Photograph of patient 4 with poor regression. M, N Pre-bleomycin standard and bird’s eye view demonstrating right proptosis with eyelid, brow and conjunctival involvement, O and P post-bleomycin standard and bird’s eye view demonstrating disappearance of the conjunctival component with <25% regression in the eyelid and brow components.
Statistical analysis
The data were arranged on an excel spreadsheet. Relevant statistical analysis was done using MedCalc version 12.2.1.0 (Ostend, Belgium). Continuous parametric data were reported using the mean (± standard deviation) and nonparametric data were reported as median along with the range. Variables of the comparative groups were compared using paired t test for parametric distribution and Mann–Whitney U test for non-parametric distribution. Multivariate logistic regression analysis and chi-square test was performed to assess the effect of multiple factors that might have influenced the outcome. A p value of <0.05 was assigned as statistically significant.
Results
There were 81 suspected OA-LM during the study period. Of these, 69 cases were included in this study, and 12 cases were excluded due to the presence of an associated venous component on imaging. The mean age at presentation was 20 ± 16 years (median 16 years; range 1 month to 70 years). The mean age at onset of the disease was 12 ± 15 years (median 6 years; range 0–70 years). The mean duration of symptoms was for 6 ± 10 years (median 2 years; range 0–50 years). Forty-one (59%) patients were female and 28 (41%) were male. Fourteen (20%) patients presented with an acute onset of symptoms (within a week of onset of symptoms), 4 (6%) with subacute (between 1 and 6 weeks) and 51 (74%) with a chronic (beyond 6 weeks) onset of symptoms. The most common symptom was presence of a peri-orbital mass in 56 (81%) patients. The most common sign was proptosis in 46 (67%) patients. The patients in this series had eyelid, orbit and conjunctival involvement of varying degree (Fig. 2). The most common presentation was that of the orbital-palpebral variant in 29 (42%) patients. There were 7 (10%) patients with involvement of the temporal fossa, frontal region or the cheek in addition to the OA-LM. The demographics and clinical features of patients with orbital and/or adnexal lymphatic malformations are presented in Table 2.
A Fisolated eyelid involvement, G–L eyelid and conjunctiva involvement, M–R isolated orbital involvement, S–X combined conjunctiva, eyelid and orbital involvement. Patient 1: (A) pre-operative photograph demonstrating a right eyelid cyst, (B) on eversion of upper eyelid, (C) in downgaze, (D) post 2 IU bleomycin sclerotherapy with regression of cyst, (E) on eversion of upper eyelid and F on downgaze. Patient 2: (G) pre-operative photograph demonstrating a right lower eyelid and conjunctival lesion, (H) on upgaze with lower eyelid eversion demonstrating caruncular and conjunctival involvement, (I) in downgaze demonstrating conjunctival involvement, (J) post 8 IU cumulative bleomycin sclerotherapy with regression eyelid lesion, (K) on eversion of lower eyelid regression of the caruncular component and (F) regression of the superior conjunctival component. Patient 3: (M and N) pre-operative photograph demonstrating left lower eyelid fullness and proptosis, (O) CT orbit suggestive of a microcystic lesion in the left intraconal space with left proptosis. P, Q Post 8 IU cumulative bleomycin sclerotherapy with regression eyelid fullness and proptosis and (R) decrease in proptosis on CT scan with reduction in tumour size. Patient 4: (S) Pre-operative photograph demonstrating left eyelid fullness and proptosis, (T) left eye with conjunctival involvement, (U) Ct orbit suggestive of eyelid and intra as well as extraconal orbital involvement. (V) post 24 units of cumulative bleomycin sclerotherapy with regression in the eyelid component and the proptosis, (W) regression of the conjunctival component and (X) CT orbit suggestive of regression of the intraconal component with reduction in the extraconal and eyelid components.
On imaging, 34 (49%) patients had microcytic lesions, 22 (32%) had macrocystic lesions and 13 (19%) had mixed micro and macrocystic lesions (Fig. 3). Mean units of bleomycin injected per patient per session were 4 ± 2 IU (median, 4 IU, range 0.3–12 IU). Mean number of treatment sessions required were 2 ± 1 (median,2; range 1–6). Mean total units of bleomycin injected per patient were 9 IU ± 8 IU (median 5.5 IU, range 1–38 IU). The mean reduction in proptosis was of 4 mm (median 2.4, range 1–9 mm) at the last follow-up visit. The mean pre-procedure Hertel’s value was 29 ± 5 mm. The mean post-procedure Hertel’s value was 20 ± 2 mm (p < 0.0001, 95% CI −11.2 to −6.8). The mean pre-procedure lesion volume was calculated to 7 ± 4 cm3 (7 cm3; 4–14 cm3). The mean post-procedure lesion volume was 0.8 ± 1.2 cm3 (median 1; range 0.2–2.4 cm3). This change was statistically and clinically significant (p < 0.0001, 95% CI −7.89 to −4.51). The response to sclerotherapy was excellent in 43 (62%) patients, good in 12 (17%) patients, fair in 9 (13%) patients and poor in 5 (7%) patients. The management and treatment outcomes of OA-LM are presented in Table 3. On subgroup analysis, excellent or good outcome was seen in 100% of the patients with macrocystic lesions, 80% of the patients with microcystic lesions and 46% of patients with mixed disease phenotypes (Fig. 3) (Table 4). Fourteen (20%) patients had fair to poor outcome (Table 4). These responses were comparable across the morphological subgroups (p = 0.24, chi-square test). Nine (56%) of these patients had a prior history of sclerotherapy with sodium tetradecyl sulphate and 7 (44%) had a history of prior debulking. There were two patients who had undergone both sclerotherapy and debulking. Multivariate logistic regression analysis was done to assess the effect of age, chronicity of presentation, previous interventions, orbital vs adnexal disease, quadrant of orbital involvement, cyst architecture, per sitting and cumulative dose of bleomycin as well as the number of sittings of bleomycin sclerotherapy with tumour response to sclerotherapy (see Supplementary material). The only significant variable affecting the response to sclerotherapy was the duration of disease (p = 0.04). A better response was seen in acute presentations.
Clinical and radiological features of macrocyst (A–F), microcyst (G–L) and mixed (M–R) architecture with response to bleomycin sclerotherapy. Patient 1 (A–F): A and B pre-operative clinical photographs demonstrating a left eyelid fullness with inferior globe dystopia, C Preoperative CT orbit s/o macrocysts in left upper eyelid, D and E post 4 IU bleomycin sclerotherapy clinical photographs with regression of cyst, E post-operative CT orbit with regression of the cyst. Patient 2 (G–L): G and H pre-operative clinical photographs demonstrating a right eyelid fullness with superior globe dystopia, I Preoperative CT orbit s/o inferior eyelid and orbital microcysts, J and K post 36 cumulative IU bleomycin sclerotherapy clinical photographs with regression of the globe dystopia and the eyelid fullness, L post-operative CT orbit demonstrating decrease in the of the volume of the cyst. Patient 3 (M–R): M and N pre-operative clinical photographs demonstrating a right proptosis, O Preoperative MRI orbit demonstrating orbital intra and extraconal macro and microcysts, P and O post 30 cumulative IU bleomycin sclerotherapy clinical photographs with regression of the proptosis, P post-operative MRI orbit demonstrating regression of the cysts.
Adverse reactions included inflammation in 11 (16%) patients, lasting a mean duration of 1 day (median 2; range 1–5 days), resolving with oral steroids in 4/11 (33%) and without any medications in 7/11 (67%). Peri-ocular pigmentation was seen in 15 (22%) patients. It gradually resolved over 3 months (median 3; range 1–9 months). In this series, bleomycin injection was not associated with any long-term side-effects, globe perforations, injection-related intraocular injuries or damage to orbital structures. Tumour regression persisted without evidence of recurrence over an average follow-up period of 3.5 years (median 3 years; range 1.5–5 years).
Discussion
This study discusses the efficacy and the long-term outcomes of non-image guided bleomycin sclerotherapy in OA-LM. The results demonstrate that the protocol of monthly transcutaneous or transconjunctival non-image guided injections continued till clinical regression leads to excellent or good outcome in 79% of all LM and in 100% of the macrocyst, 80% of the microcyst and 46% of mixed cyst LM. In this series, half of the patients with the mixed disease type and those with a history of prior debulking, responded poorly to bleomycin injection. The adverse event profile for all treated patients was limited to a short duration of peri-ocular inflammation and hyperpigmentation. Tumours responding to bleomycin sclerotherapy maintained this response over a mean duration of 3.5 years.
In the medical literature, good outcomes are reported describing the use of sclerosants for the management of macrocystic LM [14,15,16,17]. Barnacle et al. [18] used sodium tetradecyl sulphate sclerotherapy under imaging control in macrocystic disease and reported that all of the 29 patients achieved ≥ 50% reduction in maximal lesion diameter, with complete radiological regression in 51.7% (n = 15). Cheng et al. reported > 80% success rate of doxycycline sclerotherapy in macrocystic and mixed disease. In contrast, sclerotherapy has been found to be less effective in microcystic disease [20]. Chaudry et al. [25] reported the efficacy of bleomycin sclerotherapy for microcystic lymphatic malformations in the head and neck region. There was complete response in 38% patients (n = 12), partial response in 58% (n = 18), and no response in 3% (n = 1). In our study, good to excellent response was seen in 80% (27 of 34) of microcystic disease, in 100% (22 of 22) of macrocystic disease and 46% (6 of 13) of mixed lesions over a mean follow up of 3.5 years.
Following sclerotherapy, significant regression of lymphatic malformations has been reported in the 82–92% range [26,27,28,29,30]. The rate of poor response with sclerotherapy has been found to be 12–18% [26,27,28,29,30]. In our study, 93% of lymphatic malformations demonstrated a significant reduction in volume (fair to excellent response) following bleomycin sclerotherapy and 7% responded poorly. While 100% (22) patients with macrocystic morphology showed a good to excellent response to sclerotherapy, patients with microcystic and mixed lesions showed a lower response rate. On multivariate analysis, the acuteness of presentation (patient reporting within a week of onset of symptoms) was the only factor correlating with a better response to sclerotherapy (p = 0.04). The relatively unfavourable response to sclerotherapy (though not statistically significant) in patients with microcystic lesions could be due to the fact that microcysts are densely compacted smaller cystic structures and collapse of the cyst wall of microcystic lesions might not account for a significant decrease in the volume of the lesion. However, the most important role in sclerotherapy for microcystic lesions is played by lidocaine. Local anaesthetics such as lidocaine interact with cell membrane phospholipids and increase the permeability as well as sensitize these membranes to cytotoxic agents such as bleomycin [31]. This property is due to lidocaine-related increase in influx of calcium ions, thereby favouring more transport of bleomycin into the cells [31]. We believe that the addition of lidocaine (in a 4:1 dilution with bleomycin, 4 parts bleomycin and 1 part lidocaine) is crucial in the management of lymphatic malformations.
We attempted to assess the technique of bleomycin injection and the role of image guidance on the outcomes. Da Ros et al. [32] reported “lymphographic-like technique” of sclerotherapy where the position of the 22 G needle inserted into the lymphatic malformation was checked by fluoroscopic and sonographic guidance and an electronic syringe pump administered the sclerosant at a flow rate of 0.7 mL/h. They observed improvement in clinical symptoms in 93% patients. In the current study of non-image guided sclerotherapy, 93% of our patients showed fair to excellent clinical regression and in 79% the regression was graded as good or excellent. Harmoush et al. [33] used multimodal imaging in the form of intraoperative fluoroscopy combined with intra-operative cone beam CT for the management of orbital LM. The average number of sclerotherapy sessions per patient were 1.4 (range, 1–3). In comparison, the mean number of injections in our subset were 1.9 (range, 1–6). While the lesion types in their series were not documented, the published figures indicate that all of their lesions were macrocysts. In contrast, in our series, two-thirds of our patients had microcystic or mixed presentation. The prominent differences between the two studies, however, were that sclerotherapy was performed by interventional radiologists in the Harmoush et al. [33] study while in our series, it was performed by oculoplastic surgeons. We propose that good pre-operative imaging gives an idea of the 3D configuration of the tumour and may pre-empt the need for image guidance, especially for an ophthalmologist who is trained in routine retrobulbar and peribulbar injections.
Hill et al. [34] reported the average number of sessions needed for complete ablation of the LM were 1.7 in 20 patients with orbital lymphatic malformations. Paramasivam et al. [35] reported that 65% of patients required less than three procedures, whereas 35% required 3–5 procedures for bleomycin sclerotherapy. Raichura [36] et al. reported that though the number of bleomycin injections required were 1–6, satisfactory outcome was seen with 2 or less. In the present study, the average number of treatment sessions required were 2 ± 1 (median 2, range 1–6). Hill et al. [34] reported the mean reduction in proptosis of 2.4 mm, whereas Barnacle et al. [18] reported subjective improvement of proptosis in all patients. In the present study, the mean reduction in proptosis was noted to be 4 mm.
The adverse effects reported with bleomycin sclerotherapy have been transient mydriasis which resolves eventually in a few months [32, 35], intralesional haemorrhage which may require immediate lateral canthotomy with cantholysis of the inferior or superior limb of the lateral canthal tendon, lid oedema, ecchymosis, erythema, transient increase in proptosis, mild pain and transient hyperpigmentation of skin which are short term [34]. No long-term side-effects have been noted [34,35,36,37]. There has been one report of fatal lung toxicity in a 15-month-old girl, 1 week after injection of 7 units of bleomycin into a cheek macrocystic lymphatic malformation [38]. In the present study, short term side-effects of inflammation in 16% and transient hyperpigmentation in 22% were noted. No long-term side effects occurred in any of the patients. Hill et al. [34] and Raichura et al. [36] reported no recurrences in patients who completed the course of sclerotherapy. In our experience as well, no recurrences were seen over a mean follow-up period of 3.5 years.
One of the limitations of our study was the retrospective nature of the study design. Another limitation was the clinical grading of the regression being associated with subjective errors. A prospective study with radiologic regression following each session of sclerotherapy and at treatment conclusion would have helped to accurately quantify the percentage of radiological regression. Our study has demonstrated that using a well-conceived protocol, non-image guided bleomycin sclerotherapy can be a safe and effective management option for lymphatic malformations. Sclerotherapy being performed by different physicians across the eye institute network proved advantageous in compiling this series of patients. Macrocystic malformations tend to have a better response to sclerotherapy than microcystic malformations. Lesions that have undergone previous debulking tend to have a poorer outcome. Tumour regression is long lasting without evidence of recurrence. We recommend bleomycin sclerotherapy as the first line management of lymphatic malformations with surgery being reserved for those malformations unresponsive to sclerotherapy.
Summary
What was known before
-
1.
Bleomycin is an effective sclerosant for macrocystic orbital and adnexal malformations.
-
2.
Bleomycin has a good safety profile.
-
3.
Orbital infections of bleomycin are image guided.
What this study adds
-
1.
Bleomycin sclerotherapy is effective in 77% of orbital and adnexal lymphatic malformations.
-
2.
Excellent to good response is seen in 80% OA-LMs with microcystic architecture.
-
3.
Image guidance is not essential for superficial as well as deep orbital LM.
References
Nassiri N, Rootman J, Rootman DB, Goldberg RA. Orbital lymphaticovenous malformations: current and future treatments. Surv Ophthalmol. 2015;60:383–405.
Pires AM, Reinehr C, Boff F, Nazar FL. Intralesional bleomycin sclerotherapy of lymphangioma: partial response to treatment. Dermatol Surg. 2015;41:175–7.
Bothra N, Panda L, Sheth J, Tripathy D. Role of intralesional bleomycin sclerotherapy as the sole or adjunct treatment of superficial ocular adnexal lymphatic malformations. Eye 2018;32:152–5.
Hornblass A. The use of YAG-tipped contact laser in removing orbital lymphangiomas. Plast Reconstr Surg. 1989;83:397.
Lam SC, Yuen HKL. Medical and sclerosing agents in the treatment of orbital lymphatic malformations: what’s new? Curr Opin Ophthalmol. 2019;30:380–5.
Patel SR, Rosenberg JB, Barmettler A. Interventions for orbital lymphangioma. Cochrane Database Syst Rev. 2019;5(5):CD013000.
Guinto G, Guinto-Nishimura Y. Orbital lymphangiomas. World Neurosurg. 2014;81:708–9.
Harris GJ. Orbital vascular malformations: a consensus statement on terminology and its clinical implications. Orbital Society. Am J Ophthalmol. 1999;127:453–5.
Yetiser S, Karaman K Treatment of lymphangioma of the face with intralesional bleomycin: case discussion and literature review. J Maxillofac Oral Surg. 2011;10:152–4.
Patel KC, Kalantzis G, El-Hindy N, Chang BY. Sclerotherapy for orbital lymphangioma—case series and literature review. Vivo. 2017;31:263–266.
Woo YJ, Kim CY, Sgrignoli B, Yoon JS. Orbital lymphangioma: characteristics and treatment outcomes of 12 cases. Korean J Ophthalmol. 2017;31:194–201.
Lally SE. Update on orbital lymphatic malformations. Curr Opin Ophthalmol. 2016;27:413–5.
Berthout A, Jacomet PV, Putterman M, Galatoire O, Morax S. Surgical treatment of diffuse adult orbital lymphangioma: two case studies. J Fr Ophtalmol. 2008;31:1006–17.
Perkins JA, Manning SC, Tempero RM, Cunningham MJ, Edmonds JL Jr, Hoffer FA, et al. Lymphatic malformations: review of current treatment. Otolaryngol Head Neck Surg. 2010;142:795–803.
Nehra D, Jacobson L, Barnes P, Mallory B, Albanese CT, Sylvester KG. Doxycycline sclerotherapy as primary treatment of head and neck lymphatic malformations in children. J Pediatr Surg. 2008;43:451e460.
Smith MC, Zimmerman MB, Burke DK, Bauman NM, Sato Y, Smith RJ. Efficacy and safety of OK-432 immunotherapy of lymphatic malformations. Laryngoscope 2009;119:107–15.
Alomari AI, Karian VE, Lord DJ, Padua HM, Burrows PE. Percutaneous sclerotherapy for lymphatic malformations: a retrospective analysis of patient-evaluated improvement. J Vasc Inter Radio. 2006;17:1639–48.
Barnacle AM, Theodorou M, Maling SJ, Abou-Rayyah Y. Sclerotherapy treatment of orbital lymphatic malformations: a large single-centre experience. Br J Ophthalmol. 2016;100:204–8.
Cheng J. Doxycycline sclerotherapy in children with head and neck lymphatic malformations. J Pediatr Surg. 2015;50:2143–6.
Yura J, Hashimoto T, Tsuruga N, Shibata K. Bleomycin treatment for cystic hygroma in children. Nihon Geka Hokan. 1977;46:607–14.
Lee KH, Han SH, Yoon JS. Successful treatment of orbital lymphangioma with intralesional bleomycin and application of continuous negative pressure. Korean J Ophthalmol. 2015;29:70–2.
Dave TV, Javed Ali M, Jyothi AH. Peri-punctal lymphangioma treated with bleomycin. Ophthalmic Plast Reconstr Surg. 2018;34:e140.
Kumar V, Kumar P, Pandey A, Gupta DK, Shukla RC, Sharma SP, et al. Intralesional bleomycin in lymphangioma: an effective and safe non-operative modality of treatment. Cutan Aesthet Surg. 2012;5:133–136.
Dave T, Taneja S, Tiple S, Basu S, Naik MN. Conjunctival retention cysts: outcomes of aspiration and sclerotherapy with sodium tetradecyl sulfate. Ophthalmic Plast Reconstr Surg. 2019;35:165–169.
Chaudry G, Guevara CJ, Rialon KL, Kerr C, Mulliken JB, Greene AK, et al. Safety and efficacy of bleomycin sclerotherapy for microcystic lymphatic malformation. Cardiovasc Interv Radio. 2014;37:1476–81.
Tanigawa N, Shimomatsuya T, Takahashi K, Inomata Y, Tanaka K, Satomura K, et al. Treatment of cystic hygroma and lymphangioma with the use of bleomycin fat emulsion. Cancer 1987;60:741–749.
Zulfiqar MA, Zaleha AM, Zakaria Z, Amin T. The treatment of neck lymphangioma with intralesional injection of bleomycin. Med J Malays. 1999;54:478–481.
Rozman Z, Thambidorai RR, Zaleha AM, Zakaria Z, Zulfiqar MA. Lymphangioma: is intralesional bleomycin sclerotherapy effective? Biomed Imaging Inter J 2011;7:e18.
Porwal PK, Dubey KP, Morey A, Singh H, Pooja S, Bose A. Bleomycin Sclerotherapy in Lymphangiomas of Head and Neck: Prospective Study of 8 Cases. Indian J Otolaryngol Head Neck Surg. 2018;70:145–8.
Hanif AM, Saunders JA, Hawkins CM, Wojno TH, Kim HJ. Use of percutaneous bleomycin sclerotherapy for orbital lymphatic malformations. Orbit 2019;38:30–36.
Mizuno S, Ishida A. Selective enhancement of bleomycin cytotoxicity by local anesthetics. Biochem Biophys Res Commun. 1982;105:425–31.
Da Ros V, Iacobucci M, Puccinelli F, Spelle L, Saliou G. Lymphographic-like technique for the treatment of microcystic lymphatic malformation components of <3 mm. Am J Neuroradiol. 2018;39:350–354.
Harmoush S, Chinnadurai P, El Salek K, Metwalli Z, Herce H, Bhatt A, et al. Multimodality image-guided sclerotherapy of low-flow orbital vascular malformations: report of single-center experience. J Vasc Inter Radio. 2016;27:987–995.
Hill RH 3rd, Shiels WE 2nd, Foster JA, Czyz CN, Stacey A, Everman KR, et al. Percutaneous drainage and ablation as first line therapy for macrocystic and microcystic orbital lymphatic malformations. Ophthalmic Plast Reconstr Surg. 2012;28:119–25.
Paramasivam S, Fay A, FifiJ, Berenstein A. O-015 image guided bleomycin sclerotherapy for orbital lymphatic malformation. J Neuro Interv Surg. 2014;6:A8–9.
Raichura ND, Alam MS, Noronha VO, Mukherjee B. A prospective study of the role of intralesional bleomycin in orbital lymphangioma. J AAPOS. 2017;21:146–151.
MacIntosh PW, Yoon MK, Fay A. Complications of intralesional bleomycin in the treatment of orbital lymphatic malformations. Semin Ophthalmol. 2014;29:450–5.
Cho AL, Kiang SC, Lodenkamp J, Tritch WTH, Tomihama RT. Fatal lung toxicity after intralesional bleomycin sclerotherapy of a vascular malformation. Cardiovasc Interv Radio. 2020;43:648–651.
Acknowledgements
We acknowledge the support by Shobha Mocherla, PhD, Audio-Visual Producer, Communications, L V Prasad Eye Institute for proof reading of language and grammar throughout the text, tables and figures.
Funding
This study was funded by the Hyderabad Eye Research Foundation.
Author information
Authors and Affiliations
Contributions
TVD: conceptualization of the manuscript, data collection, data analysis and interpretation, manuscript drafting, critical revision and manuscript finalization. BKM: Data collection, analysis, and interpretation. SL: data collection, analysis and interpretation. DT, SK, SR, SM, AD, and AGK: review of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Dave, T.V., Madhuri, B.K., Laghmisetty, S. et al. Long term outcomes of transcutaneous non-image guided bleomycin sclerotherapy in orbital/adnexal lymphatic malformations: a protocol-based management in 69 eyes. Eye 36, 789–799 (2022). https://doi.org/10.1038/s41433-021-01527-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01527-9