Abstract
Objective
To examine the relationship of visual function as assessed by visual acuity, contrast sensitivity, and multifocal electroretinography (mfERG) to macular structural and microvascular measures on optical coherence tomography (OCT) and angiography (OCTA) in individuals with diabetes.
Methods
This is a prospective observational study conducted at a tertiary eye care centre in India. Right eyes of 121 adults with type 2 diabetes with no diabetic retinopathy (DR), mild or moderate nonproliferative DR (NPDR) were examined. Severe NPDR, proliferative DR and diabetic macular oedema were excluded. Participants underwent assessment of glycated haemoglobin (HbA1C), blood pressure, best corrected visual acuity (LogMAR), contrast sensitivity (CS), mfERG, ultrawide field fundus photography, OCT and OCTA. Correlations were assessed by Spearman’s rank correlation (rho).
Results
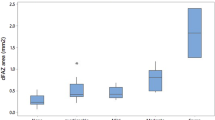
Of the total of 121 eyes, 89 had No DR, 32 had mild to moderate NPDR. In the No DR group, the LogMAR acuity was significantly and negatively correlated to central subfoveal thickness (CST) (rho = −0.420), macular vessel density (rho = −0.270) and perfusion (rho = −0.270). (ii) Contrast sensitivity correlated to foveal avascular zone circularity (rho = 0.297); (iii) mfERG P1 response densities were better with higher macular perfusion index (rho = 0.240). In the NPDR group, the LogMAR acuity also showed a significant negative correlation to CST (rho = −0.379). Other correlations were not significant.
Conclusion
Retinal and visual functional changes are evident in diabetic patients with No DR and are correlated to subclinical retinal structural changes detectable using multimodal imaging.
Similar content being viewed by others
Introduction
Diabetic retinopathy (DR) poses a major challenge to public health globally [1] and is identified by the presence of typical microvascular lesions in individuals with diabetes. Individuals with diabetes experience visual dysfunction in terms of reduced colour vision, contrast sensitivity (CS), and dark adaptation [2], which can affect the quality of life [3], especially with increasing severity of DR [4,5,6,7]. The exact pathology underlying this early functional compromise in diabetes is unclear although DR has been attributed to two pathophysiological alterations: vascular and neuroretinal degeneration [4,5,6,7].
Advances in ophthalmic imaging techniques such as optical coherence tomography angiography (OCTA), and multifocal electroretinogram (mfERG) have made it possible to examine for subclinical vascular changes in the retina as well as in the identification of biomarkers in the monitoring of the disease progression, and response to therapy progression and treatment [7]. Several visual functional attributes assessed by colour vision, contrast sensitivity [3], dark adaptation [4], visual fields and electroretinography [5] are reported to be compromised in individuals with diabetes, indicating abnormal inner retinal function and visual disability [6, 7]. However, the correlation of visual function and retinal function with the retinal vascular changes remains unclear. In this study, we investigated whether visual functions assessed by visual acuity, contrast sensitivity and retinal function assessed by multifocal electroretinography (mfERG) are related to each other and to retinal vascular changes in diabetes in eyes with No DR and mild or moderate nonproliferative DR (NPDR). The OCT angiometric measures and their correspondence with mfERG in eyes with No DR and NPDR were explored.
Subjects and methods
This was a prospective study conducted at a tertiary eye hospital in Chennai, South India. The study was approved by the Institutional Review Board and followed the tenets of the Declaration of Helsinki.
The current study is an analysis of baseline data of a 4-year longitudinal study in individuals with diabetes, who are followed-up annually for 4 years. For the original longitudinal study, the sample size calculated was 258 individuals with type 2 diabetes (129 with DR and 129 with DM and No DR) deduced after considering attrition rate per year, and the sensitivity and specificity in detecting development and progression of DR in each group over four years. The current report is a sub-analysis of the baseline data of participants recruited from March 2018 to January 2019. Figure 1 in the manuscript text is a flow chart demonstrating the initial number of patients included and the final number of participants after applying the following eligibility criteria. (Out of the 80 participants with NPDR, one participant did not fixate well despite good visual acuity, so excluded, thus NPDR was n = 79.)
Study participants
Eligibility criteria
Participants were consecutively recruited from the vitreoretinal outpatient department of the hospital. Individuals with type 2 diabetes with at least 1-year duration with evidence of No DR, mild or moderate NPDR were examined. Those who could not give informed consent or indicated that they are unable to attend for annual study visits were excluded. Individuals with media haze that compromised visual acuity or quality of imaging, had inability to maintain fixation on mfERG, coexisting ocular infection or inflammation, spherical refractive error greater than ±6 D, astigmatism greater than ±3 D, IOP > 22 mmHg, individuals with a vertical and horizontal cup-disc ratio >0.6 or reasonable suspicion of glaucoma from optic nerve head appearance, those who had undergone or were planned for vitreoretinal surgery in at least one eye, retinal vascular occlusions, untreated or treated proliferative DR and, those participating in any interventional studies were excluded.
Eligible individuals underwent assessment of visual acuity, objective and subjective refraction, intraocular pressure, systolic and diastolic blood pressure, cataract grading after pupillary dilatation, indirect ophthalmoscopy, ultrawide field fundus photography (OptosUWF™, OptosInc, UK) and HbA1c testing. Best-corrected visual acuity (BCVA) was recorded on Snellen’s chart and converted to LogMAR acuity. Participants were required to have a minimum BCVA of 6/24 (20/60) for fixation on mfERG and for contrast sensitivity evaluation. Cataract grading was undertaken after pupillary dilatation by one observer (SS) using a slit-lamp (SL-120; Carl Zeiss Meditec, Jena, Germany) per LOCS III standard photographs (LOCS III; LOCS chart III; Leo T Chylack, Harvard Medical School, Boston, MA, USA) [8]. Lens opacities were graded as nuclear opalescence (NO grades 1–6), nuclear colour (NC grades 1–6), cortical cataract (CC grades 1–5) and posterior subcapsular cataract (PSC grades 1–5). Two participants who were aphakic in both eyes were excluded. Figure 1 shows initially recruited participants and those who underwent mfERG for this study.
Diabetic retinopathy and macular oedema
DR grading was performed by one experienced observer (SS) based on the International Clinical Diabetic Retinopathy Disease Severity Scale and macular oedema [9] No DR was defined as having no apparent retinopathy; mild NPDR was defined by the presence of microaneurysms only; moderate NPDR was defined by more than just microaneurysms but less than severe NPDR; severe NPDR was defined by either presence of severe intraretinal haemorrhages and microaneurysms in each of the four quadrants, or definite venous beading in two or more quadrants, or intraretinal microvascular abnormalities in one or more quadrants. Proliferative DR was defined as the presence of either neovascularization or vitreous/preretinal haemorrhage. The presence of diabetic macular oedema (DMO) was identified by the same ophthalmologist (RR) on indirect ophthalmoscopy using the international classification [9]. DMO was considered to be present if there was some retinal thickening or hard exudates in posterior pole, as observed on dilated ophthalmoscopy. Those with severe NPDR, proliferative DR or DMO were excluded.
Optical coherence tomography angiography
Participants underwent an OCTA using the Zeiss Angioplex™ OCTA (Cirrus HD-OCT 5000 with Angioplex OCTA, Carl Zeiss meditec, USA) built on the previous Cirrus™ HD-OCT Model 5000 instrument. The technology generates high-resolution 3-D maps of the retinal and choroidal microvasculature from 68,000 A-scans per second from the central 6 mm. Macular thickness was assessed from the internal limiting membrane to the retinal pigment epithelium using the macular cube 512 × 128 protocol, quantitative angiometric measures were assessed using the angiography 6 × 6 mm protocol. The quantitative vascular and perfusion measures from the superficial capillary plexus layer were examined from the 6 × 6 scan protocol. Images with signal strength <6.0 were excluded. Automated foveal avascular zone (FAZ) measures such as area, perimeter, circularity, vessel density and perfusion in the superficial vascular plexus in the angiography 6 mm × 6 mm scans were examined and recorded by a single operator (SS). The above examination was repeated until no segmentation error was seen. Nevertheless, there were nine eyes/participants with poorly defined FAZ borders and therefore were excluded during the initial examination.
Multifocal electroretinography
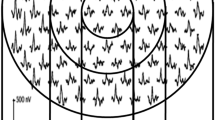
Participants also underwent mfERG (VerisTM Science 6.4.8 app, California, USA) based on the International Society for Clinical Electrophysiology of Vision guidelines for basic mfERG [10] Testing was done uniocularly using a Burian Allen Electrode, with refractive correction in place, and the other eye was patched. A gold cup electrode attached to the earlobe served as a ground electrode. The stimulus for mfERG consisted of an array of 103 hexagons presented on a monitor at a frame rate of 75 Hz, subtending an angle of 35° horizontally and 31° vertically at a viewing distance of 53 cm, flickering according to a pseudorandom m-sequence at a mean luminance of 64 cd/m2. The luminance of the bright and the dark hexagons were 128 cd/m2 and 1 cd/m2, respectively. For fixation, a red cross of 2 mm diameter was used and an in-built camera enabled the operator to monitor fixation throughout the recording. An internal Grass amplifier (Grass Technologies, An Astro-Med, Inc, West Warwick, R.I.) amplified the recordings (6100,000) which were then band-pass filtered (10–100 Hz). The actual mfERG recording time was 7 min and 17 s per eye. The mfERG responses were analysed using the Veris software and the first-order kernels were recorded and displayed in the form of a trace array of 103 local retinal responses, a 3-dimensional topographical chart, and as group trace averages of six concentric rings [11] The first-order P1 implicit times (expressed in ms) and response densities (expressed in nV/deg2) were analysed.
Contrast sensitivity
The Pelli-Robson contrast sensitivity chart (Metropia Ltd, Cambridge, UK distributed by Clement Clarke International, Essex, UK) was used for assessing CS, at a recommended testing of 1 m. The letter size at 1 m distance would ~6/200 or represent 20/688 and would subtend an angular width of 2.9° (171.9 min of arc), which is about three times larger than the largest letters on a normal visual acuity chart. The chart has six letters in each row arranged in groups of three, each group having an individual contrast. Participants were asked to read the letters from left to right and to next line, starting with the highest contrast, until they are unable to read at least two of the three letters in a group. A numerical score was then assigned based on a minimum of two letters out of a triplet with the same photometric contrast and was recorded as the lowest contrast correctly read (indication of having reached threshold) expressed in log units. The score is a single number which is a measure of the log contrast sensitivity and it represents 1/contrast. For instance, a CS score of 0.6 log units on the Pelli-Robson chart represents 1/(100.6) = 0.25 or 25% contrast.
Statistical analysis
All measures in the right eyes of participants were included in the analysis. Continuous variables were assessed for normality of distribution. Since the data were not normally distributed, two-group comparisons were made with the Mann-Whitney U test, and univariate correlations were assessed using the Spearman’s rank correlation (rho). Correlations were assessed between CS scores, LogMAR acuity, P1 implicit times and response densities of mfERG, with that of OCT central subfield thickness (CST), OCT angiometric measures (foveal avascular zone (FAZ) metrics, macular vessel density and perfusion).
Results
Figure 1 shows the recruitment to the whole study and the final number of participants who underwent mfERG. In total, 89 had diabetes with No DR and 32 had mild or moderate NPDR. 74% of eyes in the diabetic group (75% in No DR and 72% in NPDR) were phakic.
Baseline clinical characteristics in diabetes with No DR and in NPDR
Table 1 shows baseline clinical characteristics in diabetes with No DR and in NPDR.
Retinal functional measures in diabetes with No DR versus diabetes with NPDR
Retinal functional measures assessed by best corrected visual acuity (BCVA), CS and mfERG P1 measures in diabetes with No DR versus diabetes with NPDR are summarised in Table 2. The BCVA was not different between the groups (p = 0.7); however, the contrast sensitivity (CS) was significantly lower in the NPDR groups (p = 0.045), although numerically these represented small changes. The P1 implicit times in all rings were slightly but significantly delayed in the NPDR group compared to the No DR group. The P1 response densities in rings 5 and 6 were slightly but significantly lower in the NPDR group (p = 0.044 for both rings).
OCT and OCT angiometrics in No DR versus NPDR groups
The central subfoveal thickness (CST) and OCT angiometric measures in No DR versus NPDR groups are summarised in Table 3. The central subfield thickness was higher in the NPDR group compared to the No DR group and it approached statistical significance (p = 0.05), but the FAZ metrics and macular vessel density and perfusion were not significantly different between the groups.
Structure–function correlation in diabetes, No DR and NPDR groups
Best-corrected visual acuity (BCVA)
Table 4 provides the Spearman’s correlations between retinal structural and functional measures.
In the entire diabetes group (with and without NPDR), BCVA correlated negatively with the CST (rho = −0.443), FAZ circularity (rho = −0.207), superficial vessel density (SVD) (rho = −0.282), and perfusion index (rho = −0.271). In the No DR group, BCVA was correlated to CST (rho = −0.420), SVD (rho = −0.270) and perfusion (rho = −0.270). In the NPDR group, BCVA showed a similar negative correlation with that of CST (rho = −0.379).
Contrast sensitivity (CS)
In the entire diabetes group (with NPDR and No DR), CS showed a positive correlation to FAZ circularity in the diabetes group (rho = 0.268) and in the No DR group (rho = 0.297), but not in the NPDR group.
mfERG P1 measures
In the entire group with diabetes (having No DR and NPDR), the mfERG P1 response densities were significantly correlated to SVD (rho = 0.223) and perfusion (rho = 0.228).
In the No DR group, the P1 response densities were significantly correlated to the perfusion (rho = 0.240). The P1 implicit times did not show any significant correlations to any of the retinal structural measures assessed.
In the NPDR group, the mfERG measures showed no significant correlations with the retinal structural measures assessed in the study.
After applying Bonferroni’s correction for multiple comparisons, the Bonferroni adjusted p value was 0.002 for the Spearman’s correlation. In the entire group with diabetes, the correlation between the LogMAR acuity with the macular vessel density (p = 0.003) and perfusion (p = 0.004) approached this adjusted value of p = 0.002.
In those with No DR, and in those with NPDR, the correlation between retinal structure and function was not significant after Bonferroni’s correction.
Discussion
We examined if functional measures such as contrast sensitivity, visual acuity and mfERG correlate with OCT and OCT angiometric measures in diabetes, with No DR and in those with mild or moderate NPDR. Some of the visual functional measures in our study correlate with retinal OCT angiometric measures even before the occurrence of clinical signs of DR.
Visual acuity (VA) is an important measure of visual function and is a standard test in routine eye exams and clinical trials. In our study, VA is correlated to CST in individuals with diabetes with No DR and in those with NPDR. This is consistent with a recent population-based studies in healthy normal eyes, wherein, a thicker CST was significantly associated with better vision- and vision-specific functions [12, 13]. The observation that BCVA is correlated to SVD, perfusion index and higher FAZ circularity suggests the central macular function is influenced not only by CST but also by macular vascular indices on OCTA, in those with no clinical signs of DR. Similar correlations were also observed in the NPDR group but without statistical significance, which may be explained by a relatively smaller sample size in the NPDR group compared to the No DR group. Similar observation has also been reported in other disease conditions. For instance, a negative correlation was observed between BCVA and perifoveal capillary blood flow velocity, and a more stronger positive correlation between retinal thickness at the central fovea and BCVA in patients with branch retinal vein occlusion [14, 15]. In subjects with glaucoma, a negative correlation was observed between SVD and visual field sensitivity [16]. It was proposed that a reduction in perifoveal capillary blood flow velocity due to perifoveal capillary occlusion could lead to an increase in retinal thickness and therefore, may play an important role in the development and progression of DMO.
In contrast, the FAZ area and perimeter failed to show significant correlations to BCVA possibly due to the well-recognised larger inter-subject variations, suggesting that visual function is related to the overall macular perfusion rather than to FAZ area or perimeter. Although BCVA is a foveal function and the fovea is supplied by choriocapillaris, our study shows that BCVA can be influenced by macular perfusion. Our findings also show that the OCTA angiometrics are a more reliable marker of visual function changes than the visible mild to moderate DR changes.
We observed a maximum of 20% or a fourth of variance in BCVA is related to CST. Although, macular oedema was excluded, we found that subclinical thickening of CST in eyes with No DR is associated with a lower BCVA.
CS showed a positive correlation to FAZ circularity only in eyes with No DR (rho = 0.297). Early studies in the 1980s [17,18,19] and the later studies [20,21,22,23,24] observed that CS is reduced in patients with diabetes with No DR in comparison to those without diabetes.
Later, Harris et al. in 1996 [25], demonstrated a drastic recovery of contrast sensitivity to normal levels in individuals with minimal DR when administered with 100% oxygen. A similar observation was not seen in the healthy control group. This demonstrated that contrast sensitivity and retinal tissue oxygenation are related to each other, and that some of the early deficits in CS may be reversed by oxygenation levels. The authors concluded hypoxia as a mechanism in the pathogenesis of visual dysfunction and vascular compromise in DR. Our study shows that CS changes are best seen in eyes with No DR, suggesting that CS may be one of the earliest visual function changes in diabetes but does not correlate with visible vascular changes. Altered CS in diabetes may result from changes in blood flow and ischaemia [23]. We also show that CS correlates with foveal vascular changes rather than global macular perfusion indices. Similar observations were made by Arend et al. in 1997 [26] wherein CS is lower in eyes with a larger FAZ area in individuals with early diabetes mellitus.
These findings indicate that the alterations to the perifoveal network may influence selective parameters of visual function namely, contrast sensitivity and may therefore be regarded a clinical indicator of subtle ischaemia, while visual acuity changes seem to be related more to macular perfusion.
With regards to structure–function correlation, the P1 response densities in ring 1 showed a positive correlation to SVD (rho = 0.223) and perfusion (rho = 0.228) in the entire group and with perfusion in the No DR group (rho = 0.240), suggesting that the P1 response densities (a measure of bipolar cell function, photoreceptor function and/or glial activation) are influenced by macular vessel density and perfusion (vascular parameters) even before the appearance of clinical signs of DR [27].
A maximum of 6% of variance in P1 response densities in the central ring is explained by changes in macular vessel density and perfusion. Nonetheless, there is still a vascular compromise that is associated with retinal functional compromise in the absence of clinical signs of DR.
The mfERG provides a measure of retinal and macular integrity especially when the changes are minimal and dysfunction is localised to a small area. Studies on primates reported that the depolarisation of the ON-bipolar cells contributes to P1 implicit times while the set of weak mfERG response densities (10−9 V) are mainly elicited by the photoreceptor layer and outer plexiform-bipolar layer [28]. We observed that the mfERG implicit times were delayed in those with NPDR compared to those with diabetes but No DR, which is in accordance with previous reports [28,29,30]. With regards to the response density, the P1 response densities in NPDR were reduced only in rings 5 and 6 compared to the No DR group. The rings 5 and 6 roughly correspond to the perifoveal (the outer macular) region. One explanation for the above observation could be that the implicit times rather than response densities may be more sensitive to vascular insult or ischaemia in those with NPDR [29, 30] compared to those with No DR. Another explanation could be that the mfERG response densities are subject to wide variations, and therefore, may not be as reliable indicators as are the implicit times [29, 30].
In addition, the mfERG changes in NPDR in the absence of OCTA changes indicate that retinal functional alterations may occur much earlier than vascular impairment detectable on OCT angiometric parameters. This finding was observed in the absence of significant differences in the OCT angiometric measures between the two groups. This suggests that the delayed P1 implicit times are not related to the OCT vascular indices examined here. An interesting observation was that when examining the structure–function correlation, the mfERG response densities (rather than the implicit times) in ring 1 showed significant correlation with the OCT angiometric measures (SVD and perfusion indices) in the entire group with diabetes, and correlated with perfusion index in the No DR group but demonstrated no significant correlations in the NPDR group. It is likely that the delayed implicit times in the NPDR group in comparison to No DR group may suggest mfERG implicit times may have other retinal structural correlates that are outside of what is assessed in this study.
One other explanation could be the existence of two different phenotypes of NPDR as reported in the European Consortium for the Early Treatment of Diabetic Retinopathy (EUROCONDOR) study; one wherein, patients with no clinical signs of DR had mfERG alterations and the other where, patients with early microvascular impairment had no mfERG alterations. It has also been proposed that one phenotype may be more prone to develop microvascular alterations and the other to neurodegenerative abnormalities [31]. This could also be a likely explanation for observing lack of significant correlations between OCTA and mfERG measures in the NPDR group. Our data confirm the presence of different vascular and functional characteristics in patients with and without clinical signs of DR, and in addition, shows a differential correlation between the retinal structure and function.
The reduced macular vessel density and/or perfusion observed in our study may suggest occlusion or destruction of parafoveal retinal capillaries. Changes to circularity may be one of the earliest changes seen in this regard. These vascular changes influence the retinal and central visual function, even in the absence of DR.
We defined DMO based on retinal thickening or hard exudates in the posterior pole on indirect ophthalmoscopy rather than OCT-based definition of CST ≥ 300 μm. The reason is that we aimed to correlate both foveal and parafoveal changes on OCTA with mfERG. Restricting to CST ≥ 300 μm is representative of only the foveal integrity, and CST has a poor correlation even with visual acuity (a measure of foveal function). However, we explored CST criteria of DMO on OCT and observed that eight eyes in the NPDR group had CST ≥ 300 μm, indicating that these eyes were not entirely free from DMO. This factor may be taken into consideration while interpreting the results.
Strengths of this study include a large sample size of eyes with No DR, the use of standardised grading and the use of multiple visual function tests to assess various aspects of retinal changes. Nevertheless, the study has certain limitations. We did not examine axial length for the participants but excluded refractive errors greater than 6 D sphere and astigmatism greater than 3 D cylinder. Therefore, our OCT angiometric findings were not corrected for axial length [32] in both the groups. Our study focused largely on eyes with No DR. Our results need to be validated in larger cohorts in various degrees of DR. In addition, a comprehensive method to categorise visual function may provide more valuable information. Since 6/24 was considered as the minimum required visual acuity, we did not have a wider range of visual acuity to categorise visual function in a comprehensive manner for further analyses. For the same reason, we could not explore the CS changes with respect to a wider range of BCVA. The NPDR group size further limited our ability to analyse the visual function data in a more comprehensive manner.
In summary, our study observed that lower macular vessel density and macular perfusion influence the retinal function assessed by BCVA and mfERG measures while contrast sensitivity seems to be affected by more focal changes in the foveal avascular zone, even before the clinical signs of DR. Future studies may be required to assess the predictive capabilities of visual function parameters in the development of DR and to validate these findings in a larger sample of patients with mild and moderate NPDR.
Summary
What was known before:
-
It is known that several retinal structural and functional changes are evident in individuals with diabetes even before the appearance of diabetic retinopathy.
What this study adds:
-
The study observed subclinical retinal changes and the correlation of visual function and retinal function with that of retinal vascular changes in eyes with and without diabetic retinopathy.
References
Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes, estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047–53.
Cusick M, SanGiovanni JP, Chew EY, Csaky KG, Hall-Shimel K, Reed GF, et al. Central visual function and the NEI-VFQ-25 near and distance activities subscale scores in people with type 1 and 2 diabetes. Am J Ophthalmol. 2005;139:1042–1050.
Roh M, Selivanova A, Shin HJ, Miller JW, Jackson ML. Visual acuity and contrast sensitivity are two important factors affecting vision-related quality of life in advanced age-related macular degeneration. PLoS One. 2018;13:e0196481 https://doi.org/10.1371/journal.pone.0196481
Antonetti DA, Barber AJ, Bronson SK, Freeman WM, Gardner TW, Jefferson LS, et al. Diabetic retinopathy, seeing beyond glucose-induced microvascular disease. Diabetes. 2006;55:2401–11.
Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest. 1998;102:783–91.
Han Y, Bearse MA, Schneck ME, Barez S, Jacobsen CH, Adams AJ. Multifocal electroretinogram delays predict sites of subsequent diabetic retinopathy. Invest Ophthalmol Vis Sci. 2004;45:948–54.
Shapley R, Enroth-Cugell C. Visual adaptation and retinal gain controls. Prog Retinal Res. 1984;3:263–346.
Chylack LT, Wolfe JK, Singer DM, Leske MC, Bullimore MA, Bailey IL, et al. The lens opacities classification system III. The Longitudinal Study of Cataract Study Group. Arch Ophthalmol. 1993;111:831–6.
Wilkinson CP, Ferris FL, Klein RE, Lee PP, Agardh CD, Davis M, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology. 2003;110:1677–82.
Hood DC, Bach M, Brigell M, Keating D, Kondo M, Lyons JS, et al. ISCEV standard for clinical multifocal electroretinography (mfERG) (2011 edition). Doc Ophthalmol. 2012;124:1–13.
Sen P, Roy R, Maru S Ravi. Evaluation of focal retinal function using multifocal electroretinography in patients with X-linked retinoschisis. Can J Ophthalmol. 2010;45:509–13.
Poh S, Tham YC, Chee ML, Dai W, Majithia S, Soh ZD, et al. Association between macular thickness profiles and visual function in healthy eyes: The Singapore Epidemiology of Eye Diseases (SEED) Study. Sci Rep. 2020;10:1–7.
Bressler SB, Qin H, Beck RW, Chalam KV, Kim JE, Melia M.Diabetic Retinopathy Clinical Research Network et al. Factors associated with changes in visual acuity and central subfield thickness at 1 year after treatment for diabetic macular edema with ranibizumab. Arch Ophthalmol.2012;130:1153–61.
Noma H, Funatsu H, Harino S, Nagaoka T, Mimura T, Hori S. Influence of macular microcirculation and retinal thickness on visual acuity in patients with branch retinal vein occlusion and macular edema. Jpn J Ophthalmol. 2010;54:430–4.
Samara WA, Shahlaee A, Sridhar J, Khan MA, Ho AC, Hsu J. Quantitative optical coherence tomography angiography features and visual function in eyes with branch retinal vein occlusion. Am J Ophthalmol. 2016;166:76–83.
Jeon SJ, Park HYL, Park CK. Effect of macular vascular density on central visual function and macular structure in glaucoma patients. Sci Rep. 2018;8:16009 https://doi.org/10.1038/s41598-018-34417-4
Hyvärinen L, Laurinen P, Rovamo J. Contrast sensitivity in evaluation of visual impairment due to macular degeneration and optic nerve lesions. Acta Ophthalmol. 1983;61:161–170.
Sokol S, Moskowitz A, Skarf B, Evans R, Molich M, Senior B. Contrast sensitivity in diabetics with and without background retinopathy. Arch Ophthalmol. 1985;103:51–54.
Marmor MF. Contrast sensitivity versus visual acuity in retinal disease. Br J Ophthalmol. 1986;70:553–9.
Bursell SE, Clermont AC, Kinsley BT, Simonson DC, Aiello LM, Wolpert HA. Retinal blood flow changes in patients with insulin-dependent diabetes mellitus and no diabetic retinopathy. Invest Ophthalmol Vis Sci. 1996;37:886–97.
Ismail GM, Whitaker D. Early detection of changes in visual function in diabetes mellitus. Ophthalmic Physiol Opt. 1998;18:3–12.
Heravian J, Shoeibi N, Azimi A, Yasini S, Ostadimoghaddam H, Yekta AA, et al. Evaluation of contrast sensitivity, color vision and visual acuity in patients with and without diabetes. Iran J Ophthalmol. 2010;22:33–40.
Shoshani YZ, Harris A, Rusia D, Spaeth GL, Siesky B, Pollack A, et al. Contrast sensitivity, ocular blood flow and their potential role in assessing ischaemic retinal disease. Acta Ophthalmol. 2011;89:e382–395.
Gella L, Raman R, Pal SS, Ganesan S, Sharma T. Contrast sensitivity and its determinants in people with diabetes, SN-DREAMS-II, Report No 6. Eye. 2017;31:460–466.
Harris A, Arend O, Danis RP, Evans D, Wolf S, Reim M. Hyperoxia improves contrast sensitivity in early diabetic retinopathy. Br J Ophthalmol. 1996;80:209–13.
Arend A, Remky D, Evans R, Stüber A. Harris. Contrast sensitivity loss is coupled with capillary dropout in patients with diabetes. Invest Ophthalmol Vis Sci. 1997;38:1819–24.
Liu L, Gao J, Bao W, Hu C, Xu Y, Zhao B, et al. Analysis of foveal microvascular abnormalities in diabetic retinopathy using optical coherence tomography angiography with projection artifact removal. J Ophthalmol. 2018. https://doi.org/10.1155/2018/3926745
Pescosolido N, Barbato A, Stefanucci A, Buomprisco G. Role of electrophysiology in the early diagnosis and follow-up of diabetic retinopathy. J Diabetes Res. 2015. https://doi.org/10.1155/2015/319692
Bearse MA Jr, Adams AJ, Han Y, Schneck ME, Ng J, Bronson-Castain K, et al. A multifocal electroretinogram model predicting the development of diabetic retinopathy. Retinal Eye Res. 2006;25:425–48.
Fortune B, Schneck ME, Adams AJ. Multifocal electroretinogram delays reveal local retinal dysfunction in early diabetic retinopathy. Invest Ophthalmol Vis Sci. 1999;40:2638–51.
Frydkjaer-Olsen U, Soegaard Hansen R, Simó R, Cunha-Vaz J, Peto T, Grauslund J.EUROCONDOR Correlation between retinal vessel calibre and neurodegeneration in patients with type 2 diabetes mellitus in the European Consortium for the Early Treatment of Diabetic Retinopathy (EUROCONDOR). Ophthalmic Res. 2016;56:10–16.
Sampson DM, Gong P, An D, Menghini M, Hansen A, Mackey DA, et al. Axial length variation impacts on superficial retinal vessel density and foveal avascular zone area measurements using optical coherence tomography angiography. Invest Ophthalmol Vis Sci. 2017;58:3065–72.
Funding
This work was supported by the DBT/Wellcome Trust India Alliance Fellowship [grant number IA/CPHE/16/1/502670] awarded to Dr Sangeetha Srinivasan.
Author information
Authors and Affiliations
Contributions
SS and RRN were responsible for designing the study; SS obtained funding, screened, examined patients, performed data analysis, interpreted results, wrote the paper; SS, SSD, MB interpreted the results, critically reviewed the manuscript. SS, SSD, RRN, RR, AM, RAM, VK, MB approved the content of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Srinivasan, S., Sobha Sivaprasad, Rajalakshmi, R. et al. Retinal structure–function correlation in type 2 diabetes. Eye 36, 1865–1871 (2022). https://doi.org/10.1038/s41433-021-01761-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01761-1
This article is cited by
-
Structure–function associations between contrast sensitivity and widefield swept–source optical coherence tomography angiography in diabetic macular edema
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)
-
Associations of quantitative contrast sensitivity with wide-field swept-source optical coherence tomography angiography in retinal vein occlusion
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)