Abstract
This study aimed to investigate the predictive values of central systolic blood pressure (cSBP) and peripheral systolic blood pressure (pSBP) for the progression of carotid intima-media thickness (cIMT). A total of 953 Chinese participants from an atherosclerosis cohort with complete information, including baseline cSBP, questionnaire information, biochemical examination, baseline, and follow-up carotid ultrasonography quantitative data, were included in this study. A multilinear regression model, adjusted for possible covariates, was used to investigate the predictive values of cSBP and pSBP for rate of cIMT change. The average age of all participants was (52.11 ± 4.74 years). The baseline levels of cSBP, pSBP, max cIMT, and mean cIMT were (132.55 ± 18.18)mmHg, (130.76 ± 15.40)mmHg, (813.52 ± 118.49)µm and (681.11 ± 99.90)µm, respectively Those with hypertension, diabetes and dyslipidemia accounted for 40.71% (388), 18.05% (172), and 70.41% (671), respectively. After 2.3 years of follow-up, the average rates of max and mean cIMT change were 8.70 (−0.49–19.43)% and 2.59 (−4.72–10.81)%, respectively. Per standard deviation increase of cSBP, but not pSBP, was associated with increases of max (for cSBP, β 1.07, 95%CI 0.18–1.96, p = 0.018; for pSBP, β 0.48, 95%CI −0.45–1.41, p = 0.315) and mean (for cSBP, β 0.84%, 95%CI 0.10−1.58, p = 0.027; for pSBP, β 0.59%, 95%CI −0.18–1.37, p = 0.135) cIMT change rate after adjusting for possible covariates. In conclusion, cSBP, but not pSBP, is independently associated with cIMT progression in our community-based Chinese population. cSBP should be considered for the purpose of CVD primary prevention.
Similar content being viewed by others
Introduction
Cardiovascular disease (CVD) represents the leading cause of mortality in developed and many developing countries [1]. One of the most important risk factors for CVD is atherosclerosis [2]. Carotid intima-media thickness (cIMT) has emerged as a noninvasive means to assess early atherosclerosis, nonatherosclerotic compensatory remodeling with largely medial hypertrophy and CVD risk [3, 4]. A consensus statement from the American Society of Echocardiography confirms that measurement of cIMT with B-mode ultrasound is a noninvasive, sensitive, and reproducible technique for identifying and quantifying atherosclerotic burden and CVD risk.
Hypertension is a traditional and major risk factor for atherosclerosis [5]. Brachial blood pressure has been used as the criterion for hypertension for centuries [6], and several studies have demonstrated a positive association between brachial blood pressure and subclinical atherosclerosis like cIMT [7,8,9]. An increasing body of evidence suggests that central blood pressure (cBP) is a meaningful supplement for peripheral blood pressure (pBP) in predicting the occurrence of atherosclerosis as well as CVD [10,11,12]. Normally, systolic blood pressure (SBP) amplifies as much as 10–14 mmHg when moving from the aorta to the brachial artery as a consequence of reflection of the pressure wave [13]. Central SBP (cSBP) reflects the pressure that the carotid arteries and heart are directly under and is associated with arterial stiffness. Therefore, it is important to explore whether noninvasive cSBP is meaningful in the assessment of cIMT progression [14,15,16].
Several cross-sectional investigations suggest that cBP is positively associated with cIMT [14, 17]; however, to our knowledge, no study has investigated the predictive value of cSBP for the progression of cIMT in a longitudinal way, and no longitudinal study has compared the predictive values of cSBP and pSBP for the progression of cIMT. Therefore, in this study, we compared the predictive value of cSBP and pSBP for the progression of cIMT using a cohort design.
Methods
Study design
A total of 9540 residents aged above 40 years were drawn from the Pingguoyuan and Gucheng communities of Shijingshan district in Beijing, China in 2011 and 2012. In 2014, 5962 of the participants with gene chip data were invited for a follow-up visit, and 64.1% (3823) of them came onsite. Those without carotid ultrasonography examination in a follow-up visit (38) or baseline (2678) and those without cBP examination (154) were excluded in the longitudinal analysis. Ultimately, 953 participants with baseline and follow-up quantitative carotid ultrasonography data were included in the analysis. The methods and primary results of this survey have been reported elsewhere [18, 19]. Written informed consent was obtained from all participants, and this study was approved by the ethics committee of Peking University First Hospital.
Baseline data collection
A standard questionnaire was used for the collection of lifestyle data, including current smoking and history of disease, medication information, sex and age. A venous blood sample was obtained from the forearm of each participant after a minimum 12-h overnight fast. Total cholesterol (TC), low-density lipoprotein (LDL), high-density lipoprotein (HDL), total triglyceride (TG), serum creatinine (Scr), fasting plasma glucose (FPG) and the standard 75 g oral glucose tolerance test (OGTT) were measured using a Roche C8000 Automatic Analyzer.
Seated brachial blood pressure was obtained after resting at least 5 min using an HEM-7117 electronic sphygmomanometer with appropriately sized cuffs. Triplicate measurements on the right arm were taken with ≥1 min between successive readings. The peripheral SBP (pSBP) and peripheral diastolic blood pressure (pDBP) readings used in the analysis were the mean of three successful measurements.
Noninvasive cSBP was then evaluated in the same position by mathematically transforming the radial artery pulse waveform to the aortic pulse waveform using an Omron HEM-9000AI device, which has proved to be an accurate method [20]. The radial artery pressure waveform was recorded for at least 10 s until the waveform was steady and was calibrated to the brachial blood pressure. Peaks of inflection points corresponding to early and late SBP were obtained by multidimensional derivatives of the original pulse waveforms. cSBP was calculated from the pressure at the late systolic shoulder of the radial pressure waveform. Linear regression was used for the calculation of cSBP [19, 21].
Carotid ultrasonography
Carotid ultrasonography was performed both in 2012 and 2014 using a GE Medical Systems (Milwaukee, WI, USA) ultrasound scanner equipped with an 8-MHz linear-array transducer. Participants were asked to lie on the scan bed in supine position with their head resting comfortably and to rotate their neck in the direction opposite to the probe using a 45-degree angle. Max and mean cIMTs were measured by certified sonographers with an MIA-Carotid Analyzer 6.0 from the far walls of the right and left common carotid artery (CCA) at end diastole (minimal lumen diameter) on multiple cycles of images. The measured segment of interest was 10 mm in length in the CCA near the bulb and was free of plaques. The max and mean cIMTs used in analysis were the mean of the bilateral max and the mean cIMT, respectively, and the rate of cIMT change was defined as (cIMT (follow-up visit) – cIMT (baseline)) / cIMT (baseline) × 100%.
Body mass index (BMI) was calculated as kilograms per meter squared. Current smoking was defined as smoking at least one cigarette for more than half a year. Hypertension was defined as pSBP ≥ 140 mmHg or/and pDBP ≥ 90 mmHg or/and the use of antihypertensive medications or/and a history of hypertension [22]. Diabetes was defined as FPG ≥ 7.0 mmol/l or/and OGTT ≥ 11.1 mmol/l or/and use of antidiabetes medications or/and a history of diabetes [23]. Dyslipidemia was defined as TC ≥ 5.18 mmol/l or/and TG ≥ 1.7 mmol/l or/and LDL ≥ 3.37 mmol/l or/and HDL <1.04 mmol/l or/and a disease history of dyslipidemia or/and using lipid-lowering drugs [24].
Statistical analysis
Statistical analysis was performed using Empower(R) (www.empowerstats.com, X&Y solutions, Inc. Boston MA) and R (http://www.R-project.org). Normally distributed continuous data including age, Scr, baseline cIMT, rate of cIMT change, cSBP and BMI are presented as the means ± SD, and differences between groups were examined using one-way ANOVA. Categorical data included sex, current smoking, use of lipid-lowering, antidiabetes, and antihypertensive medications, and prevalence rates of hypertension, diabetes, and dyslipidemia and are presented as numbers (percentages). Differences between groups were examined using the chi-square test. A multilinear regression model, adjusted for sex, age, BMI, Scr, current smoking, using of lipid-lowering drugs, antidiabetes and antihypertensive medications, diabetes, dyslipidemia and baseline cIMT, was used to investigate the predictive values of cSBP and pSBP for the progression of cIMT. A two-tailed P < 0.05 indicated statistical significance.
Results
Baseline characteristics of participants
Table 1 shows the characteristics of the participants stratified by cSBP groups. A total of 953 participants with an average age of 52.11 ± 4.74 years were included in this analysis, and 36.62% (349) of them were men. Those with hypertension, diabetes, and dyslipidemia accounted for 40.71% (388), 18.05% (172), and 70.41% (671), respectively. The baseline levels of cSBP and pSBP and the max and mean cIMT were (132.55 ± 18.18)mmHg, (132.55 ± 18.18)mmHg, and (813.52 ± 118.49)µm and (681.11 ± 99.90)µm, respectively. There were no differences in the percentages of current smoking, use of lipid-lowering and antidiabetes medications, percentages of male and Scr, in different baseline cSBP quartiles, but the average levels of baseline age, prevalence of hypertension, diabetes and dyslipidemia, BMI, cSBP and percentage using antihypertensive medications were significantly different at the level of 0.05 in different cSBP quartiles.
Predictive values of cSBP and pSBP in the progression of cIMT
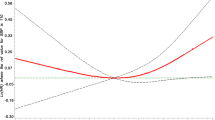
Table 2 shows the effects of cSBP and pSBP in the progression of cIMT in all 953 participants. After 2.3 years of follow-up, the rates of max and mean cIMT change were 8.70 (−0.49–19.43)% and 2.59 (−4.72–10.81)%, respectively. Per standard deviation increase of cSBP was associated with increases of 1.07% (95%CI 0.18–1.96, p = 0.018) and 0.84% (95%CI 0.10−1.58, p = 0.027) for the rates of max and mean cIMT change after adjusting for sex, age, BMI, current status of smoking, prevalence rates of diabetes and dyslipidemia, use of antidiabetes, antihypertensive and lipid-lowering drugs, Scr and baseline cIMT. Furthermore, compared with the first cSBP group (cSBP < 120 mmHg), there were increases of 2.80% (95%CI 0.39–5.21, p = 0.023) and 2.63% (95%CI 0.63–4.63, p = 0.010) for the rates of max and mean cIMT change in the third cSBP group (cSBP ≥ 140 mmHg). However, pSBP was not associated with cIMT progression in our cohort after adjusting for possible covariates (for max cIMT, β 0.48, 95%CI −0.45–1.41, p = 0.315; for mean cIMT, β 0.59, 95%CI −0.18–1.37, p = 0.135). Figure 1a, b shows the relationships of rates of max and mean cIMT change with cSBP, adjusted for possible covariates; data in the 2.5–97.5% range of the cSBP level were included in the figure. Figures 2 and 3 show interactions between covariates and cSBP; no interaction was found between covariates and cSBP in our cohort analysis. We further analyzed the effect of cSBP on the progression of cIMT in those without hypertension, and the result showed that even in those without hypertension, cSBP also predicted the progression of cIMT (for max cIMT, β 1.79, 95%CI 0.37−3.21, p = 0.014; for mean cIMT, β 1.10, 95%CI −0.05–2.25, p = 0.060).
Association between cSBP and progression of cIMT. a Association between cSBP and rate of max cIMT change; b Association between cSBP and rate of mean cIMT change. cSBP central systolic blood pressure (mmHg), cIMT carotid intima-media thickness; rate of cIMT change: (cIMT (follow-up visit) – cIMT (baseline)) / cIMT (baseline) × 100%. Adjusted for: sex, age, body mass index, baseline cIMT, serum creatinine, current smoking, diabetes, dyslipidemia, and the use of antihypertensive, antidiabetes and lipid-lowering drugs
Interaction analysis of potential effect modifiers for the association between cSBP and rate of max cIMT change. Adjusted for, if not stratified by, sex, age, body mass index (BMI), diabetes, dyslipidemia, antihypertensive, lipid-lowering and antidiabetes drugs, current smoking, serum creatinine, and baseline cIMT; cSBP central systolic blood pressure (mmHg), cIMT carotid intima-media thickness; Rate of cIMT change: (cIMT (follow-up) – cIMT (baseline)) / cIMT (baseline) × 100%
Interaction analysis of potential effect modifiers for the association between cSBP and rate of mean cIMT change. Adjusted for, if not stratified by, sex, age, body mass index (BMI), diabetes, dyslipidemia, antihypertensive, lipid-lowering and antidiabetes drugs, current smoking, serum creatinine, and baseline cIMT; cSBP central systolic blood pressure (mmHg), cIMT carotid intima-media thickness; rate of cIMT change: (cIMT (follow-up) – cIMT (baseline)) / cIMT (baseline) × 100%
Discussion
cSBP but not pSBP is independently associated with the progression of both max and mean cIMT in a longitudinal analysis in our community-based cohort. Moreover, cSBP still predicts cIMT progression in those subjects without hypertension after adjusting for possible covariates, which is consistent with many cross-sectional studies and supports trials using cBP as a treatment target [10]. To our knowledge, this is the first cohort study reporting the independent predictive value of cSBP for the progression of cIMT and is the first cohort study to investigate the predictive values of cSBP and pSBP for the progression of cIMT together.
cSBP has been found to be associated with targeted organ damages, such as left ventricular mass and cardiovascular death [2, 15, 25, 26], which indirectly supports our result since cIMT is associated with CVD and atherosclerosis. For example, a study with 1272 normotensive and untreated hypertensive participants in Taiwan found that cBP independently contributed to cardiovascular mortality and that both cPP and cSBP were confirmed to be associated with left ventricle hypertrophy and cardiovascular outcome in the Strong Heart Study [10]. Montezano noted that noninvasive cBP by pulse wave analysis was a valuable method in predicting TOD in untreated essential hypertension patients and that cSBP should be taken into account for the prevention of TOD in these patients [27].
In addition, clinical studies have concluded that cBP-lowering treatment properties in hypertensive patients alter hypertension-associated cardiac and vascular hypertrophy [28]. The ASCOT−BPLA study found that in recent ischemic stroke and transient ischemic attack patients, Allopurinol lowered cBP and reduced cIMT progression [28], which also indicated that cBP plays an important role in cIMT progression.
Furthermore, some cross-sectional studies also found a positive association between cSBP and cIMT, which is a support for our study [29]. For example, in a population-based cross-sectional study of 462 black South Africans, a positive relationship between central pulse pressure (cPP) and cIMT (partial r = 0.40, p < 0.01) was found [30]. Similarly, in the CRIC study at the University of Pennsylvania, both cPP (regression coefficient 0.0025, p < 0.001) and cSBP (regression coefficient 0.0017, p < 0.001) significantly associated with cIMT (n = 367) [31]. The most important study to date was a baseline cross-sectional survey in the same community-based population, which also highlighted a positive association between cBP and increase of cIMT (increase of cIMT was defined as cIMT > 0.9 mm; OR for an average increase of 10 mmHg cSBP: 1.069, p < 0.01) [32], but this study found that pSBP was also associated with cIMT. However, the study used data judged on site immediately by certified sonographers, and cIMT was used as a binary variable (increase of cIMT was defined as cIMT > 0.9 mm), while our analysis used data measured by certified sonographers with an MIA-Carotid Analyzer 6.0. More investigations are needed to determine the mechanisms of this discrepancy between longitudinal and cross-sectional analyses.
Two studies that did not demonstrate a positive association between cBP and cIMT were as follows: a part of the Latinos Using Cardio Actions to Reduce Risk (LUCHAR) program with 177 subjects found that cBP was associated with cIMT only in the univariate analysis; after adjusting for Framingham factors, this association was no longer significant [33]. However, as a cross-sectional study, the sample size was small. A study including 706 subjects (59 ± 10 years) who visited the hospital for a physical check-up found that cSBP had no relationship with cIMT, but in this study, the average age of participants was older than other studies, and no adjustment was made for antihypertensive medication, which may cause inaccuracy [34].
Regarding the predictive value of pSBP on cIMT progression, associated longitudinal studies are rare, and divergence existed in previous cohort studies. Kendrick et al. found that higher SBP at baseline was associated with a significantly greater yearly change in covariate-adjusted mean cIMT in 724 chronic kidney disease patients over a 4-year follow-up period [35]; however, Berg et al. found that pSBP was not associated with change in IMT in a population-based cohort study over a 5-year follow-up period [36]. Different disease status among participants may be possible reason for this discrepancy.
The mechanisms for the association between cSBP and progression of cIMT can be complex and are still under investigation. Nox-induced ROS production can be one possible reason, which is increased with elevated blood pressure, influencing mitogenic signaling and cell cycle regulation and leading to altered cell growth and vascular remodeling [37]. Matrix metalloproteinases also play a role in the progression of cIMT since they contribute to extracellular matrix proteolysis and vascular smooth muscle cell migration [38]. In addition, because of the existence of pulse amplification and central wave reflections [13], SBP is normally amplified as much as 10–14 mmHg when moving from the aorta to the brachial artery. Therefore, cBP is a more accurate representation of loading conditions on the heart and carotid artery. Moreover, the pulse wave travels faster in stiff arteries than healthy and compliant arteries; as a result, in stiff arteries, the reflection of the pulse wave merges earlier with the incident wave, making the elevation of cSBP more apparent in stiff arteries [39], suggesting that cSBP can reflect the stiffness of arteries, which is associated with carotid artery damage. Furthermore, arteries are under steady and pulsatile stretch [40]; steady stretch implicates FAK (focal adhesion kinase), and pulsatile stretch frees radicals derived from oxidative stress and the presence of inflammatory factors. cSBP is associated with both steady and pulsatile stretches, meaning that elevated cSBP may damage arteries through both of the above pathways [41, 42].
Limitations
There are several limitations in this study. First, this was a single-center study with participants derived from only one community-based cohort; therefore, more studies are needed for a more exact relationship between cSBP and the progression of cIMT. However, while no longitudinal observational study has been designed to investigate the predictive value of cSBP on the progression of cIMT, our study filled this gap. Second, diastolic blood pressure measurement is not available for the Omron device; therefore, cDBP and cPP were not available in this study. More studies on these measurements are still needed. However, cSBP was found to have a predictive value for the progression of cIMT even after adjusting for other covariates in this study, which is also meaningful. Third, in the present study, the duration of follow-up was slightly shorter. Replication analyses are still needed to fully determine the predictive value of cSBP for cardiovascular events, but we did identify a predictive value of cSBP for the progression of cIMT, meaning that cSBP plays an important role in the progression of cIMT. Previous studies that focused on the progression of cIMT have successfully identified risk factors of cIMT progression with a follow-up period of no longer than 3 years [43, 44].
Conclusion
cSBP, but not pSBP, was independently associated with cIMT progression risk in our Chinese community-based population. Assessment and treatment of cBP may help prevent atherosclerosis at an early stage.
References
Wong ND. Epidemiological studies of CHD and the evolution of preventive cardiology. Nat Rev Cardiol. 2014;11:276−89.
Bots ML, Dijk JM, Oren A, Grobbee DE. Carotid intima-media thickness, arterial stiffness and risk of cardiovascular disease: current evidence. J Hypertens. 2002;20:2317–25.
Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N. et al. Mannheim Carotid intima-media thickness and plaque consensus (2004–2006–2011). Cerebrovasc Dis. 2012;34:290–6.
Lorenz MW, Markus HS, Bots ML, Rosvall M, Sitzer M. Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation. 2007;115:459–67.
Aronson S, Fontes ML. Hypertension: a new look at an old problem. Curr Opin Anaesthesiol. 2006;19:59–64.
Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, et al. 2007 guidelines for the management of arterial hypertension. J Hypertens. 2007;25:1105–87.
Raitakari O, Juonala M, Kahonen M, Taittonen L, Laitinen T, Maki-Torkko N, et al. Cardiovascular risk factors in childhood and carotid artery intima-media thickness in adulthood. JAMA. 2003:2277−83.
Zachariah JP. Improving blood pressure in children is protective over the long term. Circulation. 2013;3:198–9.
Herder M, Johnsen SH, Arntzen KA, Mathiesen EB. Risk factors for progression of carotid intima-media thickness and total plaque area: a 13-year follow-up study: The Tromso Study. Stroke. 2012;43:1818–23.
Roman MJ, Devereux RB, Kizer JR, Lee ET, Galloway JM, Ali T et al. Central pressure more strongly relates to vascular disease and outcome than does brachial pressure: The Strong Heart Study. Hypertension. 2007;50:197–203.
Salvi P, Bellasi A, Di Iorio B. Does it make sense to measure only the brachial blood pressure? Blood Purif. 2013;36:21–5.
Woodman RJ, Kingwell BA, Beilin LJ, Hamilton SE, Dart AM, Watts GF. Assessment of central and peripheral arterial stiffness: studies indicating the need to use a combination of techniques. Am J Hypertens. 2005;18:249–60.
Safar ME, Jankowski P. Central blood pressure and hypertension: role in cardiovascular risk assessment. Clin Sci. 2009;116:273–82.
Pierre Boutouyrie MP, Caroline Bussy M, Patrick Lacolley MP, Xavier Girerd M, Laloux B, Ste Phane Laurent MP. Association between local pulse pressure, mean blood pressure, and large-artery remodeling. Circulation. 1999:1387−93.
Roman MJ, Devereux RB. Association of central and peripheral blood pressures with intermediate cardiovascular phenotypes. Hypertension. 2014;63:1148–53.
Omboni S, Posokhov IN, Rogoza AN. Relationships between 24-h blood pressure variability and 24-h central arterial pressure, pulse wave velocity and augmentation index in hypertensive patients. Hypertens Res. 2017;40:385–91.
Roman MJ. Association of central and peripheral pulse pressure with intermediate cardiovascular phenotypes. J Hum Hypertens. 2012:67–74.
Fan F, Qi L, Jia J, Xu X, Liu Y, Yang Y. et al. Noninvasive central systolic blood pressure is more strongly related to kidney function decline than peripheral systolic blood pressure in a Chinese community-based population novelty and significance. Hypertension. 2016;67:1166–72.
Cheng G, Fan F, Zhang Y, Qi L, Jia J. Different associations between blood pressure indices and carotid artery damages in a community-based population of China. J Hum Hypertens. 2016;30:750–4
Miyashita H. Clinical assessment of central blood pressure. Curr Hypertens Rev. 2012;8:80–90.
Odaira M, Tomiyama H, Hashimoto H, Kojima I, Matsumoto C, Yoshida M. et al. Increased arterial stiffness weakens the relationship between wave reflection and the central pressure indexes in men younger than 60 years of age. Am J Hypertens. 2011;24:881–6.
Giuseppe Mancia CI. et al.Guy De Backer CBAD, Czech Republic RFBG, Italy AMHU, France KNPL, Poland RESG, Italy AZ. 2007 guidelines for the management of arterial hypertension. The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hum Hypertens. 2007;25:1105–87.
Handelsman Y, Bloomgarden ZT, Grunberger G, Umpierrez G, Zimmerman RS, Bailey TS, et al. American Association of Clinical Endocrinologists and American College of Endocrinology—clinical practice guidelines for developing a diabetes mellitus comprehensive care plan—2015. Endocr Pract. 2015;(suppl 1):1−87.
The joint committee on the revision of guidelines for the prevention and treatment of dyslipidemia in adults in China. Guidelines for the prevention and treatment of dyslipidemia in adults in China (2016 revision) Chin. J Cardiol. 2016;44:833–53.
Wang KL, Cheng HM, Chuang SY, Spurgeon HA, Ting CT, Lakatta EG, et al. Central or peripheral systolic or pulse pressure: which best related to target-organs and future mortality. J Hum Hypertens. 2009;3:461–7.
Han W, Han X, Sun N, Chen Y, Jiang S, Li M. Relationships between urinary electrolytes excretion and central hemodynamics, and arterial stiffness in hypertensive patients. Hypertens Res. 2017;40:746–51.
Lekakis JP, Zakopoulos NA, Protogerou AD, Kotsis VT, Papaioannou TG, Stamatelopoulos KS. et al. Cardiac hypertrophy in hypertension: relation to 24-h blood pressure profile and arterial stiffness. Int J Cardiol. 2004;97:29–33.
Williams B, Lacy PS, Thom SM, Cruickshank K, Stanton A, Collier D. et al. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes—principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation. 2006;113:1213–25.
Kollias A, Lagou S, Zeniodi ME, Boubouchairopoulou N, Stergiou GS. Association of central versus brachial blood pressure with target-organ damage novelty and significance. Hypertension. 2015;67:183–90.
Norton GR, Majane OH, Maseko MJ, Libhaber C, Redelinghuys M, Kruger D. et al. Brachial blood pressure-independent relations between radial late systolic shoulder-derived aortic pressures and target organ changes. Hypertension. 2012;59:885–92.
DeLoach SS, Appel LJ, Chen J, Joffe MM, Gadegbeku CA, Mohler ER. et al. Aortic pulse pressure is associated with carotid IMT in chronic kidney disease: report from Chronic Renal Insufficiency Cohort. Am J Hypertens. 2009;22:1235–41.
Cheng G, Fan F, Zhang Y, Qi L, Jia J. Different associations between blood pressure indices and carotid artery damages in a community-based population of China. J Hum Hypertens. 2016;36:1–5.
Krantz MJ, Long CS, Hosokawa P, Karimkahani E, Dickinson M, Estacio RO, et al. Pulse wave velocity and carotid atherosclerosis in White and Latino patients with hypertension. BMC 2011;11:15.
Yamashita S, Dohi Y, Takase H, Sugiura T, Ohte N. Central blood pressure reflects left ventricular load, while brachial blood pressure reflects arterial damage. Blood Press. 2014;23:356–62.
Kendrick J, Chonchol M, Gnahn H, Sander D. Higher systolic blood pressure is associated with progression of carotid intima–media thickness in patients with chronic kidney disease. Kidney Int. 2010;77:794–800.
Berg EVD, Biessels GJ, Stehouwer CDA, Kappelle LJ, Heine RJ, Nijpels G. et al. Ten-year time course of risk factors for increased carotid intima–media thickness: the Hoorn Study. Eur J Cardiov Prev R. 2010;17:168–74.
Montezano AC, Tsiropoulou S, Dulak-Lis M, Harvey A, Camargo LDL, Touyz RM. Redox signaling, Nox5 and vascular remodeling in hypertension. Curr Opin Nephrol Hy. 2015;24:425–33.
Belo VA, Guimarães DA, Castro MM. Matrix metalloproteinase 2 as a potential mediator of vascular smooth muscle cell migration and chronic vascular remodeling in hypertension. J Vasc Res. 2016;52:221–31.
Staessen JA, Wang J, Bianchi G, Birkenhäger WH. Essential hypertension. Lancet. 2003:1629−41.
Safar ME. Systolic blood pressure, pulse pressure and arterial stiffness as cardiovascular risk factors. Curr Opin Nephrol Hy. 2001;10:257–61.
Lehoux S, Esposito B, Merval R, Loufrani L, Tedgui A. Pulsatile stretch-induced extracellular signal-regulated kinase 1/2 activation in organ culture of rabbit aorta involves reactive oxygen species. Arterioscl Throm Vas. 2000;20:2366–72.
Lehoux S, Esposito B, Merval R, Tedgui A. Differential regulation of vascular focal adhesion kinase by steady stretch and pulsatility. Circulation. 2005;111:643–9.
Furuhashi M, Yuda S, Muranaka A, Kawamukai M, Matsumoto M, Tanaka M. et al. Circulating fatty acid-binding protein 4 concentration predicts the progression of carotid atherosclerosis in a general population without medication. Circ J. 2018;82:1121–9.
Kim J, Park S, Yan P, Jeffers BW, Cerezo C. Effect of inter-individual blood pressure variability on the progression of atherosclerosis in carotid and coronary arteries: apost hoc analysis of the NORMALISE and PREVENT studies. Eur Heart J—Cardiovasc Pharmacother. 2016:pvw019.
Acknowledgements
We thank the staff members of the Gucheng and Pingguoyuan Community Health Centers and the research coordinators who participated in this cohort survey.
Funding
This study was supported by grants from the UM-PUHSC Joint Institute for Translational and Clinical Research (grant nos: BMU20110177 and BMU20160530).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sun, P., Yang, Y., Cheng, G. et al. Noninvasive central systolic blood pressure, not peripheral systolic blood pressure, independently predicts the progression of carotid intima-media thickness in a Chinese community-based population. Hypertens Res 42, 392–399 (2019). https://doi.org/10.1038/s41440-018-0175-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-018-0175-5
Keywords
This article is cited by
-
Association of central blood pressure with an exaggerated blood pressure response to exercise among elite athletes
European Journal of Applied Physiology (2024)
-
Comparison of Brachial Blood Pressure and Central Blood Pressure in Attended, Unattended, and Unattended Standing Situations
Hypertension Research (2021)
-
Reference values of office central blood pressure, pulse wave velocity, and augmentation index recorded by means of the Mobil‐O‐Graph PWA monitor
Hypertension Research (2020)
-
Deterioration of sexual function is associated with central hemodynamics in adult Japanese men
Hypertension Research (2020)