Abstract
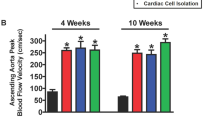
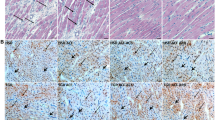
Coronary flow velocity (CFV) is reduced in pathologic cardiac hypertrophy. This functional reduction is linked to adverse cardiac remodeling, hypertension and fibrosis, and angiotensin II (AngII) is a key molecular player. Angiotensin receptor blockers (ARBs) are known to attenuate adverse cardiac remodeling and fibrosis following increased afterload, while the mechanism by which these drugs offer clinical benefits and regulate hemodynamics remains unknown. To establish a direct connection between coronary flow changes and angiotensin-induced hypertension, we used a Doppler echocardiographic method in two distinct disease models. First, we performed serial echocardiography to visualize coronary flow and assess heart function in patients newly diagnosed with hypertension and currently on ARBs or calcium channel blockers (CCBs). CFV improved significantly in the hypertensive patients after 12 weeks of ARB treatment but not in those treated with CCBs. Second, using murine models of pressure overload, including Ang II infusion and aortic banding, we mimicked the clinical conditions of Ang II- and mechanical stress-induced hypertension, respectively. Both Ang II infusion and aortic banding increased the end-systolic pressure-volume relationship and cardiac fibrosis, but interestingly, only Ang II infusion resulted in a significant reduction in CFV and corresponding activation of pressure-sensitive proteins, including connective tissue growth factor, hypoxia-inducible factor 1α and signal transducer and activator of transcription 3. These data support the existence of a molecular and functional link between AngII-induced hemodynamic remodeling and alterations in coronary vasculature, which, in part, can explain the clinical benefit of ARB treatment in hypertensive patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chilian WM. Coronary microcirculation in health and disease. Summary of an NHLBI workshop. Circulation. 1997;95:522–8.
Levy BI, Ambrosio G, Pries AR, Struijker-Boudier HA. Microcirculation in hypertension: a new target for treatment? Circulation. 2001;104:735–40.
Camici PG, Olivotto I, Rimoldi OE. The coronary circulation and blood flow in left ventricular hypertrophy. J Mol Cell Cardiol. 2012;52:857–64. https://doi.org/10.1016/j.yjmcc.2011.08.028.
Kawai M, Hongo K, Komukai K, Morimoto S, Nagai M, Seki S, et al. Telmisartan predominantly suppresses cardiac fibrosis, rather than hypertrophy, in renovascular hypertensive rats. Hypertens Res. 2009;32:604–10. https://doi.org/10.1038/hr.2009.61.
Tsutsui H, Matsushima S, Kinugawa S, Ide T, Inoue N, Ohta Y, et al. Angiotensin II type 1 receptor blocker attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Hypertens Res. 2007;30:439–49. https://doi.org/10.1291/hypres.30.439.
Schmermund A, Lerman LO, Ritman EL, Rumberger JA. Cardiac production of angiotensin II and its pharmacologic inhibition: effects on the coronary circulation. Mayo Clin Proc. 1999;74:503–13. https://doi.org/10.4065/74.5.503.
Kamezaki F, Tasaki H, Yamashita K, Shibata K, Hirakawa N, Tsutsui M, et al. Angiotensin receptor blocker improves coronary flow velocity reserve in hypertensive patients: comparison with calcium channel blocker. Hypertens Res. 2007;30:699–706. https://doi.org/10.1291/hypres.30.699.
Hinoi T, Tomohiro Y, Kajiwara S, Matsuo S, Fujimoto Y, Yamamoto S, et al. Telmisartan, an angiotensin II type 1 receptor blocker, improves coronary microcirculation and insulin resistance among essential hypertensive patients without left ventricular hypertrophy. Hypertens Res. 2008;31:615–22. https://doi.org/10.1291/hypres.31.615.
Packer M. Pathophysiological mechanisms underlying the adverse effects of calcium channel-blocking drugs in patients with chronic heart failure. Circulation. 1989;80:IV59–67.
Unno K, Oikonomopoulos A, Fujikawa Y, Okuno Y, Narita S, Kato T, et al. Alteration in ventricular pressure stimulates cardiac repair and remodeling. J Mol Cell Cardiol. 2019;133:174–87. https://doi.org/10.1016/j.yjmcc.2019.06.006.
Goyal J, Khan ZY, Upadhyaya P, Goyal B, Jain S. Comparative study of high dose mono-therapy of amlodipine or telmisartan, and their low dose combination in mild to moderate hypertension. J Clin Diagn Res. 2014;8:HC08–11. https://doi.org/10.7860/JCDR/2014/9352.4500.
Shimizu Y, Yamasaki F, Furuno T, Kubo T, Sato T, Doi Y, et al. Metabolic effect of combined telmisartan and nifedipine CR therapy in patients with essential hypertension. Int J Gen Med. 2012;5:753–8. https://doi.org/10.2147/IJGM.S28890.
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28:1–39 e14. https://doi.org/10.1016/j.echo.2014.10.003.
Wada T, Hirata K, Shiono Y, Orii M, Shimamura K, Ishibashi K, et al. Coronary flow velocity reserve in three major coronary arteries by transthoracic echocardiography for the functional assessment of coronary artery disease: a comparison with fractional flow reserve. Eur Heart J Cardiovasc Imaging. 2014;15:399–408. https://doi.org/10.1093/ehjci/jet168.
deAlmeida AC, van Oort RJ, Wehrens XH. Transverse aortic constriction in mice. Journal of visualized experiments: JoVE. 2010;. https://doi.org/10.3791/1729.
Lu H, Howatt DA, Balakrishnan A, Moorleghen JJ, Rateri DL, Cassis LA, et al. Subcutaneous angiotensin II infusion using osmotic pumps induces aortic aneurysms in mice. J Visual Exp. 2015;. https://doi.org/10.3791/53191.
Krege JH, Hodgin JB, Hagaman JR, Smithies O. A noninvasive computerized tail-cuff system for measuring blood pressure in mice. Hypertension 1995;25:1111–5. https://doi.org/10.1161/01.hyp.25.5.1111.
Townsend D. Measuring pressure volume loops in the mouse. J Visual Exp. 2016. https://doi.org/10.3791/53810.
Chang WT, Fisch S, Chen M, Qiu Y, Cheng S, Liao R. Ultrasound based assessment of coronary artery flow and coronary flow reserve using the pressure overload model in mice. J Visual Exp. 2015:e52598. https://doi.org/10.3791/52598.
Haghikia A, Stapel B, Hoch M, Hilfiker-Kleiner D. STAT3 and cardiac remodeling. Heart Fail Rev. 2011;16:35–47. https://doi.org/10.1007/s10741-010-9170-x.
Szabo Z, Magga J, Alakoski T, Ulvila J, Piuhola J, Vainio L, et al. Connective tissue growth factor inhibition attenuates left ventricular remodeling and dysfunction in pressure overload-induced heart failure. Hypertension 2014;63:1235–40. https://doi.org/10.1161/HYPERTENSIONAHA.114.03279.
Higgins DF, Biju MP, Akai Y, Wutz A, Johnson RS, Haase VH. Hypoxic induction of Ctgf is directly mediated by Hif-1. Am J Physiol Ren Physiol. 2004;287:F1223–32. https://doi.org/10.1152/ajprenal.00245.2004.
Ikonomidis I, Tzortzis S, Paraskevaidis I, Triantafyllidi H, Papadopoulos C, Papadakis I, et al. Association of abnormal coronary microcirculatory function with impaired response of longitudinal left ventricular function during adenosine stress echocardiography in untreated hypertensive patients. Eur Heart J Cardiovasc Imaging. 2012;13:1030–40. https://doi.org/10.1093/ehjci/jes071.
Daniels JT, Schultz GS, Blalock TD, Garrett Q, Grotendorst GR, Dean NM, et al. Mediation of transforming growth factor-beta(1)-stimulated matrix contraction by fibroblasts: a role for connective tissue growth factor in contractile scarring. Am J Pathol. 2003;163:2043–52. https://doi.org/10.1016/s0002-9440(10)63562-6.
Han J, Ye S, Zou C, Chen T, Wang J, Li J, et al. Angiotensin II causes biphasic STAT3 activation through TLR4 to initiate cardiac remodeling. Hypertension. 2018;72:1301–11. https://doi.org/10.1161/HYPERTENSIONAHA.118.11860.
Xiong A, Liu Y. Targeting hypoxia inducible factors-1alpha as a novel therapy in fibrosis. Front Pharm. 2017;8:326 https://doi.org/10.3389/fphar.2017.00326.
Silter M, Kogler H, Zieseniss A, Wilting J, Schafer K, Toischer K, et al. Impaired Ca(2+)-handling in HIF-1alpha(+/-) mice as a consequence of pressure overload. Pflug Arch. 2010;459:569–77. https://doi.org/10.1007/s00424-009-0748-x.
Hartley CJ, Reddy AK, Michael LH, Entman ML, Taffet GE. Coronary flow reserve as an index of cardiac function in mice with cardiovascular abnormalities. In: Proceedings of the Annual International Conference on IEEE Engineering in Medicine and Biology Society 2009; 2009:1094–7. https://doi.org/10.1109/IEMBS.2009.5332488.
Taqueti VR, Hachamovitch R, Murthy VL, Naya M, Foster CR, Hainer J, et al. Global coronary flow reserve is associated with adverse cardiovascular events independently of luminal angiographic severity and modifies the effect of early revascularization. Circulation. 2015;131:19–27. https://doi.org/10.1161/CIRCULATIONAHA.114.011939.
Wang X, Wu J, Zhu D, You J, Zou Y, Qian J, et al. Characterization of coronary flow reserve and left ventricular remodeling in a mouse model of chronic aortic regurgitation with carvedilol intervention. J Ultrasound Med. 2015;34:483–93. https://doi.org/10.7863/ultra.34.3.483.
Layland J, Carrick D, Lee M, Oldroyd K, Berry C. Adenosine: physiology, pharmacology, and clinical applications. JACC Cardiovasc Intervent. 2014;7:581–91. https://doi.org/10.1016/j.jcin.2014.02.009.
Kim JH, Jiang YP, Cohen IS, Lin RZ, Mathias RT. Pressure-overload-induced angiotensin-mediated early remodeling in mouse heart. PLoS ONE. 2017;12:e0176713 https://doi.org/10.1371/journal.pone.0176713.
Shukri MZ, Tan JW, Manosroi W, Pojoga LH, Rivera A, Williams JS, et al. Biological sex modulates the adrenal and blood pressure responses to angiotensin II. Hypertension. 2018;71:1083–90. https://doi.org/10.1161/HYPERTENSIONAHA.117.11087.
Yoshiyama M, Nakamura Y, Omura T, Izumi Y, Matsumoto R, Oda S, et al. Angiotensin converting enzyme inhibitor prevents left ventricular remodelling after myocardial infarction in angiotensin II type 1 receptor knockout mice. Heart. 2005;91:1080–5. https://doi.org/10.1136/hrt.2004.035618.
Acknowledgements
This work was supported by Chi-Mei Medical Center, Scientist Developed Grant of National Health Research Institute, Taiwan (NHRI-EX106- 10618SC), Ministry of Science and Technology (MOST 109-2326-B-384 -001 -MY3) (W-T Chang), and National Institute of Health grants R01HL131532 (SC), R01HL134168 (SC), HL093148 (RL), HL128135 (RL) and HL099073 (RL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chang, WT., Fisch, S., Dangwal, S. et al. Angiotensin II blockers improve cardiac coronary flow under hemodynamic pressure overload. Hypertens Res 44, 803–812 (2021). https://doi.org/10.1038/s41440-021-00617-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-021-00617-1