Abstract
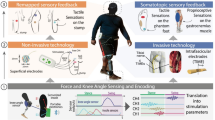
Restoring somatosensory feedback in individuals with lower-limb amputations would reduce the risk of falls and alleviate phantom limb pain. Here we show, in three individuals with transtibial amputation (one traumatic and two owing to diabetic peripheral neuropathy), that sensations from the missing foot, with control over their location and intensity, can be evoked via lateral lumbosacral spinal cord stimulation with commercially available electrodes and by modulating the intensity of stimulation in real time on the basis of signals from a wireless pressure-sensitive shoe insole. The restored somatosensation via closed-loop stimulation improved balance control (with a 19-point improvement in the composite score of the Sensory Organization Test in one individual) and gait stability (with a 5-point improvement in the Functional Gait Assessment in one individual). And over the implantation period of the stimulation leads, the three individuals experienced a clinically meaningful decrease in phantom limb pain (with an average reduction of nearly 70% on a visual analogue scale). Our findings support the further clinical assessment of lower-limb neuroprostheses providing somatosensory feedback.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Source data for the figures in this study are available in the Data Archive for the Brain Initiative, with the identifier https://doi.org/10.18120/8qby-hk82 (ref. 59). The raw and analysed datasets generated during the study are available for research purposes from the corresponding author on reasonable request.
Code availability
The custom code used to generate figures for this manuscript is available at https://github.com/pitt-rnel/NatureBME2023_SCSLowerLimb.
Change history
28 December 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41551-023-01175-2
References
Sexton, A. T. & Fleming, L. L. Lower extremity amputations. In Medical Management of the Surgical Patient: A Textbook of Perioperative Medicine (eds Walker, H. K., Lubin, M. F., Spell, N. O., Smith, R. B. & Dodson, T. F.) 741–743 (Cambridge University Press, 2006).
Viseux, F. J. F. The sensory role of the sole of the foot: review and update on clinical perspectives. Neurophysiol. Clin. 50, 55–68 (2020).
Petersen, B. A., Nanivadekar, A. C., Chandrasekaran, S. & Fisher, L. E. Phantom limb pain: peripheral neuromodulatory and neuroprosthetic approaches to treatment. Muscle Nerve 59, 154–167 (2019).
Kuffler, D. P. Origins of phantom limb pain. Mol. Neurobiol. 55, 60–69 (2018).
Valle, G. et al. Mechanisms of neuro-robotic prosthesis operation in leg amputees. Sci. Adv. 7, eabd8354 (2021).
Petrini, F. M. et al. Enhancing functional abilities and cognitive integration of the lower limb prosthesis. Sci. Transl. Med. 11, eaav8939 (2019).
Charkhkar, H. et al. High-density peripheral nerve cuffs restore natural sensation to individuals with lower-limb amputations. J. Neural Eng. 15, 056002 (2018).
Petrini, F. M. et al. Sensory feedback restoration in leg amputees improves walking speed, metabolic cost and phantom pain. Nat. Med. 25, 1356–1363 (2019).
Raspopovic, S. et al. Restoring natural sensory feedback in real-time bidirectional hand prostheses. Sci. Transl. Med. 6, 222ra19–222ra19 (2014).
Dhillon, G. S. & Horch, K. W. Direct neural sensory feedback and control of a prosthetic arm. IEEE Trans. Neural Syst. Rehabil. Eng. 13, 468–472 (2005).
Charkhkar, H., Christie, B. P. & Triolo, R. J. Sensory neuroprosthesis improves postural stability during Sensory Organization Test in lower-limb amputees. Sci. Rep. 10, 1–13 (2020).
Ortiz-Catalan, M. et al. An osseointegrated human–machine gateway for long-term sensory feedback and motor control of artificial limbs. Sci. Transl. Med. 6, 257re6–257re6 (2014).
Horch, K., Meek, S., Taylor, T. G. & Hutchinson, D. T. Object discrimination with an artificial hand using electrical stimulation of peripheral tactile and proprioceptive pathways with intrafascicular electrodes. IEEE Trans. Neural Syst. Rehabil. Eng. 19, 483–489 (2011).
Tan, D. W. et al. A neural interface provides long-term stable natural touch perception. Sci. Transl. Med. 6, 257ra138–257ra138 (2014).
Davis, T. S. et al. Restoring motor control and sensory feedback in people with upper extremity amputations using arrays of 96 microelectrodes implanted in the median and ulnar nerves. J. Neural Eng. 13, 036001 (2016).
Marasco, P. D., Schultz, A. E. & Kuiken, T. A. Sensory capacity of reinnervated skin after redirection of amputated upper limb nerves to the chest. Brain 132, 1441–1448 (2009).
Rossini, P. M. et al. Double nerve intraneural interface implant on a human amputee for robotic hand control. Clin. Neurophysiol. 121, 777–783 (2010).
Dillingham, T. R., Pezzin, L. E. & MacKenzie, E. J. Limb amputation and limb deficiency: epidemiology and recent trends in the United States. South. Med. J. 95, 875–883 (2002).
Kumar, K. & Rizvi, S. Historical and present state of neuromodulation in chronic pain. Curr. Pain Headache Rep. 18, 387 (2014).
Fanciullo, G. J., Rose, R. J., Lunt, P. G., Whalen, P. K. & Ross, E. The state of implantable pain therapies in the United States: a nationwide survey of academic teaching programs. Anesth. Analg. 88, 1311–1316 (1999).
Chandrasekaran, S. et al. Sensory restoration by epidural stimulation of the lateral spinal cord in upper-limb amputees. Elife 9, 1–26 (2020).
Lee, M. W. L., McPhee, R. W. & Stringer, M. D. An evidence‐based approach to human dermatomes. Clin. Anat. 21, 363–373 (2008).
Kim, L. H., McLeod, R. S. & Kiss, Z. H. T. A new psychometric questionnaire for reporting of somatosensory percepts. J. Neural Eng. 15, 13002 (2018).
Graczyk, E. L. et al. The neural basis of perceived intensity in natural and artificial touch (accepted). Sci. Transl Med. 142, 1–11 (2016).
Page, D. M. et al. Discriminability of multiple cutaneous and proprioceptive hand percepts evoked by intraneural stimulation with Utah slanted electrode arrays in human amputees. J. Neuroeng. Rehabil. 18, 12 (2021).
Mastinu, E. et al. Grip control and motor coordination with implanted and surface electrodes while grasping with an osseointegrated prosthetic hand. J. Neuroeng. Rehabil. 16, 49 (2019).
Flesher, S. N. et al. Intracortical microstimulation of human somatosensory cortex. Sci. Transl. Med. 8, 361ra141–361ra141 (2016).
Wrisley, D. M. & Kumar, N. A. Functional gait assessment: concurrent, discriminative, and predictive validity in community-dwelling older adults. Phys. Ther. 90, 761–773 (2010).
Koehler-McNicholas, S. R., Danzl, L., Cataldo, A. Y. & Oddsson, L. I. E. Neuromodulation to improve gait and balance function using a sensory neuroprosthesis in people who report insensate feet—a randomized control cross-over study. PLoS ONE 14, e0216212 (2019).
Melzack, R. The McGill Pain Questionnaire: major properties and scoring methods. Pain 1, 277–299 (1975).
Cho, T. A. Spinal cord functional anatomy. Contin. Lifelong Learn. Neurol. 21, 13–35 (2015).
Muret, D. & Makin, T. R. The homeostatic homunculus: rethinking deprivation-triggered reorganisation. Curr. Opin. Neurobiol. 67, 115–122 (2021).
Selvarajah, D. et al. Structural and functional abnormalities of the primary somatosensory cortex in diabetic peripheral neuropathy: a multimodal MRI study. Diabetes 68, 796–806 (2019).
Ekman, G. ös Weber’s law and related functions. J. Psychol. 47, 343–352 (1959).
Kim, S. et al. Behavioral assessment of sensitivity to intracortical microstimulation of primate somatosensory cortex. Proc. Natl Acad. Sci. USA 112, 15202–15207 (2015).
Petersen, B., Sparto, P. J. & Fisher, L. E. Clinical measures of balance and gait cannot differentiate somatosensory impairments in people with lower-limb amputation. Gait Posture 99, 104–110 (2023).
Jones, M. G. et al. Neuromodulation using ultra low frequency current waveform reversibly blocks axonal conduction and chronic pain. Sci. Transl. Med. 13, eabg9890 (2021).
Lewandowski, A. S., Palermo, T. M., Kirchner, H. L. & Drotar, D. Comparing diary and retrospective reports of pain and activity restriction in children and adolescents with chronic pain conditions. Clin. J. Pain 25, 299–306 (2009).
Saal, H. P. & Bensmaia, S. J. Biomimetic approaches to bionic touch through a peripheral nerve interface. Neuropsychologia 79, 344–353 (2015).
Okorokova, E. V., He, Q. & Bensmaia, S. J. Biomimetic encoding model for restoring touch in bionic hands through a nerve interface. J. Neural Eng. 15, 66033 (2018).
Valle, G. et al. Biomimetic intraneural sensory feedback enhances sensation naturalness, tactile sensitivity, and manual dexterity in a bidirectional prosthesis. Neuron 100, 37–45.e7 (2018).
George, J. A. et al. Biomimetic sensory feedback through peripheral nerve stimulation improves dexterous use of a bionic hand. Sci. Robot. 4, 1–12 (2019).
Sankar, S. et al. Texture discrimination with a soft biomimetic finger using a flexible neuromorphic tactile sensor array that provides sensory feedback. Soft Robot. 8, 577–587 (2021).
Stone, A. A. & Broderick, J. E. Real-time data collection for pain: appraisal and current status. Pain Med. 8, S85–S93 (2007).
Gwaltney, C. J., Shields, A. L. & Shiffman, S. Equivalence of electronic and paper-and-pencil administration of patient-reported outcome measures: a meta-analytic review. Value Health 11, 322–333 (2008).
Broderick, J. E. et al. The accuracy of pain and fatigue items across different reporting periods. Pain 139, 146–157 (2008).
Garcia-Palacios, A. et al. Ecological momentary assessment for chronic pain in fibromyalgia using a smartphone: a randomized crossover study. Eur. J. Pain 18, 862–872 (2014).
Teirlinck, C. H., Sonneveld, D. S., Bierma-Zeinstra, S. M. A. & Luijsterburg, P. A. J. Daily pain measurements and retrospective pain measurements in hip osteoarthritis patients with intermittent pain. Arthritis Care Res. 71, 768–776 (2019).
Toossi, A. et al. Comparative neuroanatomy of the lumbosacral spinal cord of the rat, cat, pig, monkey, and human. Sci. Rep. 11, 1955 (2021).
Nanivadekar, A., Chandrasekaran, S., Gaunt, R. & Fisher, L. RNEL PerceptMapper (2020). https://doi.org/10.5281/ZENODO.3939658
Prieto, T. E., Myklebust, J. B., Hoffmann, R. G., Lovett, E. G. & Myklebust, B. M. Measures of postural steadiness: differences between healthy young and elderly adults. IEEE Trans. Biomed. Eng. 43, 956–966 (1996).
Cripps, A., Livingston, S. C. & DeSantis, B. M. The test–retest reliability and minimal detectable change of the Sensory Organization Test and Head-Shake Sensory Organization Test. J. Sports Med. Allied Health Sci. 2, 2 (2016).
Wrisley, D. M., Marchetti, G. F., Kuharsky, D. K. & Whitney, S. L. Reliability, internal consistency, and validity of data obtained with the functional gait assessment. Phys. Ther. 84, 906–918 (2004).
Beninato, M., Fernandes, A. & Plummer, L. S. Minimal clinically important difference of the functional gait assessment in older adults. Phys. Ther. 94, 1594–1603 (2014).
Pesarin, F. & Salmaso, L. The permutation testing approach: a review. Statistica 70, 481–509 (2010).
Powell, M. P. et al. Epidural stimulation of the cervical spinal cord for post-stroke upper-limb paresis. Nat. Med. 29, 689–699 (2023).
Sdrulla, A. D., Guan, Y. & Raja, S. N. Spinal cord stimulation: clinical efficacy and potential mechanisms. Pain Pract. 18, 1048–1067 (2018).
Forouzanfar, T., Weber, W. E. J., Kemler, M. & van Kleef, M. What is a meaningful pain reduction in patients with complex regional pain syndrome type 1? Clin. J. Pain 19, 281–285 (2003).
Nanivadekar, A. C., et al. Dataset for ‘Restoration of sensory feedback from the foot and reduction of phantom limb pain via closed-loop spinal cord stimulation.’ Data Archive for the Brain Initiative (2023); https://doi.org/10.18120/8qby-hk82
Acknowledgements
We dedicate this work to our dear friend and colleague Sliman J. Bensmaia, without whose support, insight and passion for science and mentorship this project would not have been possible. We wish to thank P. Sparto for his assistance with the blinded review of functional outcome measures. All authors acknowledge support for the research described in this study from the National Institutes of Health cooperative agreement UH3NS100541. B.A.P. acknowledges support for the research described in this study from National Institutes of Health training grant F30HD098794. B.B. acknowledges support for the research described in this study from Swiss National Science Foundation Doc.Mobility fellowship P1FRP3_188027.
Author information
Authors and Affiliations
Contributions
L.E.F., D.J.W., S.J.B. and M.L.B. conceived the study and designed the research. A.C.N., R.B., B.A.P., D.S., T.J.M., B.B., J.F., A.N.D., I.L. and L.E.F. performed experiments. E.R.H. and V.J.M. performed implantation procedures. E.R.H., V.J.M., M.L.B. and I.L. managed medical care and oversight for the study. A.C.N., R.B., B.A.P. and E.V.O. performed data analysis. L.E.F., A.C.N., R.B., B.A.P., E.V.O., S.J.B. and M.C. wrote the manuscript with input from all authors. L.E.F. supervised the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Rickard Brånemark, Troels S. Jensen, Winfried Mayr, Solomon Tesfaye and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Sensory integrity and impairments for each participant.
Shaded regions indicate areas with either impaired (light) or absent (dark) light touch sensation as determined by clinical neurological testing.
Extended Data Fig. 2 Heatmaps showing the rate of occurrence of sensations in the missing limb across weeks.
Darker shades indicate higher rate of occurrence of sensations in that location. No testing was done on week 11 for Participant 3.
Extended Data Fig. 3 Comparison of the threshold amplitude that evoked sensation in the missing limb (with co-activation in the residual limb) and the threshold amplitude that evoked sensation only in the residual limb.
The threshold amplitude for each testing day was determined by increasing the stimulation amplitude in 0.5 or 1 mA steps and asking the participants to report the location where they perceived the evoked sensation. Error bars show the mean ± standard deviation across multiple days (N = 4 for Participant 1, N = 13 for Participant 2 and N = 8 for Participant 3).
Extended Data Fig. 4 Dermatomal activation by electrodes located at different vertebrae levels for Participant 2 and Participant 3.
The left image shows the expected dermatomal innervation in the leg22. In the right, the horizontal bars indicate different dermatomes and the white ovals indicate the approximate electrode position that evoked sensations in that dermatome with respect to the vertebrae level. Participant 1 had substantial lead migration across weeks, making it challenging to precisely define the location of the electrodes with respect to vertebrae levels, so we have not included those results.
Extended Data Fig. 5 Percept quality of evoked sensations in the missing limb.
The participants were given a list of 13 natural descriptors and 5 paresthetic descriptors to describe the quality of the sensation. The top panel shows the frequency of each descriptor for the two evoked sensations for each participant shown in Fig. 2a. For all reported sensations, we stimulated via each electrode with a 1-sec long pulse train. The bottom panel shows the total number of descriptors used to describe the sensations each week.
Extended Data Fig. 6 Additional results from psychophysical discrimination assessment.
a, Variation of Weber fraction for different electrodes in Participant 1 and 2 as a function of the reference amplitude in the discrimination task. b, Variation of JND for the same electrodes in Participants 1 and 2 as a function of the reference amplitude. Participant 3 was discarded from these analyses due to insufficient data points.
Extended Data Fig. 7 Full results of Sensory Organization Test (SOT).
a, Participant 2 performed the SOT without stimulation (light blue) with sham stimulation (that is, stimulation in the residual limb only, gray) and with stimulation (stimulation in the prosthetic foot, dark blue). Sham stimulation substantially decreased performance for three of six conditions (with greater than minimum detectable change [MDC, 3.98]), suggesting that stimulation on the residual limb alone was not sufficient to improve performance. b, Participant 3 performed the SOT without stimulation (light magenta) and with stimulation (dark magenta). Both Participant 2 and Participant 3 exhibited improved performance on conditions with platform sway and eyes closed (+5.12 Participant 2, +9.60 Participant 3) and with visual surround sway (+4.04 Participant 2, +13.39 Participant 3). Both participants, however, exhibited decreased performance with stimulation during static standing with eyes closed (−6.25 Participant 2, −4.32 Participant 3). Additionally, Participant 3 had worse performance on static standing with eyes open with stimulation (−4.13). Change in median values reported. * represents a MDC, ** represents a clinically meaningful difference (>8.0).
Extended Data Fig. 8 McGill Pain Questionnaire results.
a, Weekly McGill Pain Questionnaire results. b, McGill Pain Questionnaire score before the implant and 1-month post-explant. The pre-implant score for Participant 2 was not recorded and we did not perform testing on week 11 for Participant 3 (indicated by the dashed line).
Supplementary information
Supplementary Information
Supplementary table and video caption.
Video
Video of participant 3 walking with real-time somatosensory feedback delivered via SCS.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nanivadekar, A.C., Bose, R., Petersen, B.A. et al. Restoration of sensory feedback from the foot and reduction of phantom limb pain via closed-loop spinal cord stimulation. Nat. Biomed. Eng (2023). https://doi.org/10.1038/s41551-023-01153-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-023-01153-8
This article is cited by
-
Neuromorphic hardware for somatosensory neuroprostheses
Nature Communications (2024)