Abstract
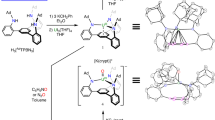
A fundamental bonding model in coordination and organometallic chemistry is the synergic, donor–acceptor interaction between a metal and a neutral π-acceptor ligand, in which the ligand σ donates to the metal, which π back-bonds to the ligand. This interaction typically involves a metal with an electron-rich, mid-, low- or even negative oxidation state and a ligand with a π* orbital. Here, we report that treatment of a uranium–carbene complex with an organoazide produces a uranium(v)–bis(imido)–dinitrogen complex, stabilized by a lithium counterion. This complex, which was isolated in a crystalline form, involves an electron-poor, high-oxidation-state uranium(v) 5f1 ion that is π back-bonded to the poor π-acceptor ligand dinitrogen. We propose that this is made possible by a combination of cooperative heterobimetallic uranium–lithium effects and the presence of suitable ancillary ligands that render the uranium ion unusually electron rich. This electron-poor back-bonding could have implications for the field of dinitrogen activation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The X-ray crystallographic data for 2 have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition number CCDC 1869009. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre (www.ccdc.cam.ac.uk/data_request/cif). All the other data supporting the findings of this study are available within the article and its Supplementary Information, or from the corresponding author upon reasonable request.
References
Fryzuk, M. D. & Johnson, S. A. The continuing story of dinitrogen activation. Coord. Chem. Rev. 200–202, 379–409 (2000).
Kushto, G. P., Souter, P. F. & Andrews, L. An infrared spectroscopic and quasirelativistic theoretical study of the coordination and activation of dinitrogen by thorium and uranium atoms. J. Chem. Phys. 108, 7121–7130 (1998).
Andrews, L., Wang, X., Gong, Y., Vlaisavljevich, B. & Gagliardi, L. Infrared spectra and electronic structure calculations for the NUN(NN)1–5 and NU(NN)1–6 complexes in solid argon. Inorg. Chem. 52, 9989–9993 (2013).
Burford, R. J. & Fryzuk, M. D. Examining the relationship between coordination mode and reactivity of dinitrogen. Nat. Rev. Chem. 1, 0026 (2017).
Dell’Amico, D. B., Calderazzo, F., Marchetti, F. & Merlino, S. Synthesis and molecular structure of [Au4Cl8], and the isolation of [Pt(CO)Cl5]– in thionyl chloride. J. Chem. Soc. Dalton Trans. 1982, 2257–2260 (1982).
Bernhardt, E. & Preetz, W. Snythesis and spectroscopic characterization of fluorocarbonylosmates, normal coordinate analysis and crystal structure of fac-[OsF3Br2(CO)]2–. Z. Anorg. Allg. Chem. 624, 694–700 (1998).
Höhling, M. & Preetz, W. Synthesis and crystal structure of tetraphenylarsonium pentachlorocarbonylosmate(iv), (Ph4As)[OsCl5(CO)]. Z. Naturforsch. B52, 978–980 (1997).
Wovchko, E. A. & Yates, J. T. Activation of O2 on a photochemically generated Rhi site on an Al2O3 surface: low temperature O2 dissociation and CO oxidation. J. Am. Chem. Soc. 120, 10523–10527 (1998).
Crayston, J. A., Almond, M. J., Downs, A. J., Poliakoff, M. & Turner, J. J. Formation of trans-M(O)2(CO)4 (M = Mo and W): intermediates in the photooxidation of matrix-isolated M(CO)6. Inorg. Chem. 23, 3051–3056 (1984).
Bernhardt, E., Willner, H., Jonas, V., Thiel, W. & Aubke, F. The tetrakis(carbonyl)dioxoosmium(vi) cation: trans-[OsO2(CO)4]2+. Angew. Chem. Int. Ed. 39, 168–171 (2000).
Malischewski, M., Seppelt, K., Sutter, J., Munz, D. & Meyer, K. A ferrocene-based dicationic iron(iv) carbonyl complex. Angew. Chem. Int. Ed. 57, 14597–14601 (2018).
Marsella, J. A., Curtis, C. J., Bercaw, J. E. & Caulton, K. G. Low-temperature infrared study of d 0 carbonyl complexes. J. Am. Chem. Soc. 102, 7244–7246 (1980).
Guram, A. S., Swenson, D. C. & Jordan, R. F. Synthesis and characterization of Cp2Zr(CH{Me}{6-ethylpyrid-2-yl})(CO)+, a d 0 metal alkyl carbonyl complex. Coordination chemistry of the four-membered azazirconacycle Cp2Zr(η2-C,N-CH{Me}{6-ethylpyrid-2-yl})+. J. Am. Chem. Soc. 114, 8991–8996 (1992).
Hurlburt, P. K. et al. Nonclassical metal carbonyls: [Ag(CO)]+ and [Ag(CO)2]+. J. Am. Chem. Soc. 116, 10003–10014 (1994).
Lupinetti, A. J., Frenking, G. & Strauss, S. H. Nonclassical metal carbonyls: appropriate definitions with a theoretical justification. Angew. Chem. Int. Ed. 37, 2113–2116 (1998).
Roussel, P. & Scott, P. Complex of dinitrogen with trivalent uranium. J. Am. Chem. Soc. 120, 1070–1071 (1998).
Cloke, F. G. N. & Hitchcock, P. B. Reversible binding and reduction of dinitrogen by a uranium(iii) pentalene complex. J. Am. Chem. Soc. 124, 9352 (2002).
Mansell, S. M., Kaltsoyannis, N. & Arnold, P. L. Small molecule activation by uranium tris(aryloxides): experimental and computational studies of binding of N2, coupling of CO, and deoxygenation insertion of CO2 under ambient conditions. J. Am. Chem. Soc. 133, 9036–9051 (2011).
Mansell, S. M., Farnaby, J. H., Germeroth, A. I. & Arnold, P. L. Thermally stable uranium dinitrogen complex with siloxide supporting ligands. Organometallics 32, 4214–4222 (2013).
Korobkov, I., Gambarotta, S. & Yap, G. P. A. A highly reactive uranium complex supported by the calix[4]tetrapyrrole tetraanion affording dinitrogen cleavage, solvent deoxygenation, and polysilanol depolymerisation. Angew. Chem. Int. Ed. 41, 3433–3436 (2002).
Falcone, M., Chatelain, L., Scopelliti, R., Zivkovic, I. & Mazzanti, M. Nitrogen reduction and functionalization by a multimetallic uranium nitride complex. Nature 547, 332–335 (2017).
Brennan, J. G., Andersen, R. A. & Robbins, J. L. Preparation of the first molecular carbon monoxide complex of uranium, (Me3SiC5H4)3UCO. J. Am. Chem. Soc. 108, 335–336 (1986).
Parry, J., Carmona, E., Coles, S. & Hursthouse, M. Synthesis and single crystal X-ray diffraction study on the first isolable carbonyl complex of an actinide, (C5Me4H)3U(CO). J. Am. Chem. Soc. 117, 2649–2650 (1995).
Del Mar Conejo, M. et al. Carbon monoxide and isocyanide complexes of trivalent uranium metallocenes. Chem. Eur. J. 5, 3000–3009 (1999).
Evans, W. J., Kozimor, S. A., Nyce, G. W. & Ziller, J. W. Comparative reactivity of sterically crowded nf 3 (C5Me5)3Nd and (C5Me5)3U complexes with CO: formation of a nonclassical carbonium ion versus an f element metal carbonyl complex. J. Am. Chem. Soc. 125, 13831–13835 (2003).
Castro-Rodriguez, I. & Meyer, K. Carbon dioxide reduction and carbon monoxide activation employing a reactive uranium(iii) complex. J. Am. Chem. Soc. 127, 11242–11243 (2005).
Langeslay, R. R. et al. Synthesis, structure, and reactivity of the sterically crowded Th3+ complex (C5Me5)3Th including formation of the thorium carbonyl, [(C5Me5)3Th(CO)][BPh4]. J. Am. Chem. Soc. 139, 3387–3398 (2017).
Evans, W. J., Kozimor, S. A. & Ziller, J. W. A monometallic f element complex of dinitrogen: (C5Me5)3U(η1-N2). J. Am. Chem. Soc. 125, 14264–14265 (2003).
Siladke, N. A. et al. Synthesis, structure, and magnetism of an f element nitrosyl complex, (C5Me4H)3UNO. J. Am. Chem. Soc. 134, 1243–1249 (2012).
Maron, L., Eisenstein, O. & Andersen, R. A. The bond between CO and Cp′3U in Cp′3U(CO) involves back-bonding from the Cp′3U ligand-based orbitals of π-symmetry, where Cp′ represents a substituted cyclopentadienyl ligand. Organometallics 28, 3629–3635 (2009).
Lu, E., Boronski, J. T., Gregson, M., Wooles, A. J. & Liddle, S. T. Silyl–phosphino–carbene complexes of uranium(iv). Angew. Chem. Int. Ed. 57, 5506–5511 (2018).
Pyykkö, P. Additive covalent radii for single-, double-, and triple-bonded molecules and tetrahedrally bonded crystals: a summary. J. Phys. Chem. A 119, 2326–2337 (2015).
Odom, A. L., Arnold, P. L. & Cummins, C. C. Heterodinuclear uranium/molybdenum dinitrogen complexes. J. Am. Chem. Soc. 120, 5836–5837 (1998).
Hayton, T. W. et al. Synthesis of imido analogs of the uranyl ion. Science 310, 1941–1943 (2005).
Lu, E. et al. Synthesis, characterization, and reactivity of a uranium(vi) carbene imido oxo complex. Angew. Chem. Int. Ed. 53, 6696–6700 (2014).
Cooper, O. J. et al. Uranium–carbon multiple bonding: facile access to the pentavalent uranium carbene [U{C(PPh2NSiMe3)2}(Cl)2(I)] and comparison of Uv=C and Uiv=C double bonds. Angew. Chem. Int. Ed. 50, 2383–2386 (2011).
Mills, D. P. et al. Synthesis of a uranium(vi)–carbene: reductive formation of uranyl(v)–methanides, oxidative preparation of a [R2C=U=O]2+ analogue of the [O=U=O]2+ uranyl ion (R = Ph2PNSiMe3), and comparison of the nature of Uiv=C, Uv=C and Uvi=C double bonds. J. Am. Chem. Soc. 134, 10047–10054 (2012).
Cooper, O. J. et al. The nature of the U=C bond: pushing the stability of high oxidation state uranium carbenes to the limit. Chem. Eur. J. 19, 7071–7083 (2013).
Cohen, J. D., Mylvaganam, M., Fryzuk, M. D. & Loehr, T. M. Resonance Raman studies of dinuclear zirconium complexes with a bridging dinitrogen ligand. Possible N2-coordination models for nitrogenase. J. Am. Chem. Soc. 116, 9529–9534 (1994).
Laplaza, C. E. et al. Dinitrogen cleavage by three-coordinate molybdenum(iii) complexes: mechanistic and structural data. J. Am. Chem. Soc. 118, 8623–8638 (1996).
Liddle, S. T. The renaissance of non-aqueous uranium chemistry. Angew. Chem. Int. Ed. 54, 8604–8641 (2015).
King, D. M. et al. Molecular and electronic structure of terminal and alkali metal-capped uranium(v)-nitride complexes. Nat. Commun. 7, 13773 (2016).
Castro-Rodríguez, I. & Meyer, K. Small molecule activation at uranium coordination complexes: control of reactivity via molecular architecture. Chem. Commun. 2006, 1353–1368 (2006).
Kindra, D. R. & Evans, W. J. Magnetic susceptibility of uranium complexes. Chem. Rev. 114, 8865–8882 (2014).
Minasian, S. G., Krinsky, J. L. & Arnold, J. Evaluating f-element bonding from structure and thermodynamics. Chem. Eur. J 17, 12234–12245 (2011).
Bader, R. F. W., Slee, T. S., Cremer, D. & Kraka, E. Description of conjugation and hyperconjugation in terms of electron distributions. J. Am. Chem. Soc. 105, 5061–5068 (1983).
MacLeod, K. C. & Holland, P. L. Recent developments in the homogeneous reduction of dinitrogen by molybdenum and iron. Nat. Chem. 5, 559–565 (2013).
Foster, S. L. et al. Catalysts for nitrogen reduction to ammonia. Nat. Cat. 1, 490–500 (2018).
Acknowledgements
We acknowledge funding and support from the UK Engineering and Physical Sciences Research Council (grants EP/M027015/ and EP/P001386/1), European Research Council (grant CoG612724), Royal Society (grant UF110005), the National EPSRC UK EPR Facility, The University of Manchester and the UK National Nuclear Laboratory. B.E.A. and N.K. also thank the University of Manchester for computational resources and associated support services from the Computational Shared Facility.
Author information
Authors and Affiliations
Contributions
E.L. and J.T.B. prepared and characterized the compound and its precursors. B.E.A. and N.K. performed the energy-scan calculations and analysed the results. E.L., A.J.W., I.J.V.-Y. and G.F.S.W. collected, solved, refined and analysed the crystallographic data. E.L., L.R.D., J.D.C. and P.J.C. recorded and interpreted the Raman data. F.T. recorded and interpreted the EPR data. S.T.L. originated the central idea, supervised the work, analysed the data, performed the DFT, NBO and QTAIM calculations and analysed the results, and wrote the manuscript with contributions from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary experimental details, computational details, characterization data, computational data tables and references.
Crystallographic data
CIF for compound 2; CCDC reference: 1869009.
Rights and permissions
About this article
Cite this article
Lu, E., Atkinson, B.E., Wooles, A.J. et al. Back-bonding between an electron-poor, high-oxidation-state metal and poor π-acceptor ligand in a uranium(v)–dinitrogen complex. Nat. Chem. 11, 806–811 (2019). https://doi.org/10.1038/s41557-019-0306-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0306-x