Abstract
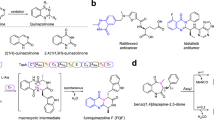
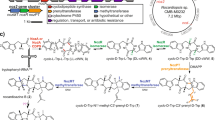
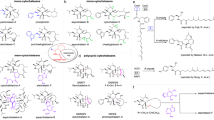
Prenylated indole alkaloids such as the calmodulin-inhibitory malbrancheamides and anthelmintic paraherquamides possess great structural diversity and pharmaceutical utility. Here, we report complete elucidation of the malbrancheamide biosynthetic pathway accomplished through complementary approaches. These include a biomimetic total synthesis to access the natural alkaloid and biosynthetic intermediates in racemic form and in vitro enzymatic reconstitution to provide access to the natural antipode (+)-malbrancheamide. Reductive cleavage of an l-Pro–l-Trp dipeptide from the MalG non-ribosomal peptide synthetase (NRPS) followed by reverse prenylation and a cascade of post-NRPS reactions culminates in an intramolecular [4+2] hetero-Diels–Alder (IMDA) cyclization to furnish the bicyclo[2.2.2]diazaoctane scaffold. Enzymatic assembly of optically pure (+)-premalbrancheamide involves an unexpected zwitterionic intermediate where MalC catalyses enantioselective cycloaddition as a bifunctional NADPH-dependent reductase/Diels–Alderase. The crystal structures of substrate and product complexes together with site-directed mutagenesis and molecular dynamics simulations demonstrate how MalC and PhqE (its homologue from the paraherquamide pathway) catalyse diastereo- and enantioselective cyclization in the construction of this important class of secondary metabolites.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Finefield, J. M., Frisvad, J. C., Sherman, D. H. & Williams, R. M. Fungal origins of the bicyclo[2.2.2]diazaoctane ring system of prenylated indole alkaloids. J. Nat. Prod. 75, 812–833 (2012).
Klas, K., Tsukamoto, S., Sherman, D. H. & Williams, R. M. Natural Diels–Alderases: elusive and irresistable. J. Org. Chem. 80, 11672–11685 (2015).
Klas, K. R. et al. Structural and stereochemical diversity in prenylated indole alkaloids containing the bicyclo[2.2.2]diazaoctane ring system from marine and terrestrial fungi. Nat. Prod. Rep. 35, 532–558 (2018).
Robertson, A. P. et al. Paraherquamide and 2-deoxy-paraherquamide distinguish cholinergic receptor subtypes in ascaris muscle. J. Pharmacol. Exp. Ther. 303, 853–860 (2002).
Little, P. R. et al. Efficacy of a combined oral formulation of derquantel–abamectin against the adult and larval stages of nematodes in sheep, including anthelmintic-resistant strains. Vet. Parasitol. 181, 180–193 (2011).
Buxton, S. K. et al. Investigation of acetylcholine receptor diversity in a nematode parasite leads to characterization of tribendimidine- and derquantel-sensitive nAChRs. PLoS Pathog. 10, e1003870 (2014).
Mugishima, T. et al. Absolute stereochemistry of citrinadins A and B from marine-derived fungus. J. Org. Chem. 70, 9430–9435 (2005).
Mercado-Marin, E. V. et al. Total synthesis and isolation of citrinalin and cyclopiamine congeners. Nature 509, 318–324 (2014).
Porter, A. E. A. & Sammes, P. G. A Diels–Alder reaction of possible biosynthetic importance. J. Chem. Soc. D 1970, 1103a (1970).
Stocking, E. M. & Williams, R. M. Chemistry and biology of biosynthetic Diels–Alder reactions. Angew. Chem. Int. Ed. 42, 3078–3115 (2003).
Li, S. et al. Comparative analysis of the biosynthetic systems for fungal bicyclo[2.2.2]diazaoctane indole alkaloids: the (+)/(−)-notoamide, paraherquamide and malbrancheamide pathways. MedChemComm 3, 987–996 (2012).
Stocking, E. M., Sanz-Cervera, J. F. & Williams, R. M. Studies on the biosynthesis of paraherquamide: synthesis and incorporation of a hexacyclic indole derivative as an advanced metabolite. Angew. Chem. Int. Ed. 40, 1296–1298 (2001).
Ding, Y. S. et al. Detection of VM55599 and preparaherquamide from Aspergillus japonicus and Penicillium fellutanum: biosynthetic implications. J. Nat. Prod. 71, 1574–1578 (2008).
Ding, Y. S., Greshock, T. J., Miller, K. A., Sherman, D. H. & Williams, R. M. Premalbrancheamide: synthesis, isotopic labeling, biosynthetic incorporation and detection in cultures of Malbranchea aurantiaca. Org. Lett. 10, 4863–4866 (2008).
Ding, Y. et al. Genome-based characterization of two prenylation steps in the assembly of the stephacidin and notoamide anticancer agents in a marine-derived Aspergillus sp. J. Am. Chem. Soc 132, 12733–12740 (2010).
Wu, C. J., Li, C. W., Gao, H., Huang, X. J. & Cui, C. B. Penicimutamides D–E: two new prenylated indole alkaloids from a mutant of the marine-derived Penicillium purpurogenum G59. RSC Adv. 7, 24718–24722 (2017).
Martinez-Luis, S. et al. Malbrancheamide, a new calmodulin inhibitor from the fungus Malbranchea aurantiaca. Tetrahedron 62, 1817–1822 (2006).
Kim, H. J., Ruszczycky, M. W., Choi, S. H., Liu, Y. N. & Liu, H. W. Enzyme-catalysed [4+2] cycloaddition is a key step in the biosynthesis of spinosyn A. Nature 473, 109–112 (2011).
Hudson, G. A., Zhang, Z. G., Tietz, J. I., Mitchell, D. A. & van der Donk, W. A. In vitro biosynthesis of the core scaffold of the thiopeptide thiomuracin. J. Am. Chem. Soc. 137, 16012–16015 (2015).
Wever, W. J. et al. Chemoenzymatic synthesis of thiazolyl peptide natural products featuring an enzyme-catalyzed formal [4+2] cycloaddition. J. Am. Chem. Soc. 137, 3494–3497 (2015).
Tian, Z. H. et al. An enzymatic [4+2] cyclization cascade creates the pentacyclic core of pyrroindomycins. Nat. Chem. Biol. 11, 259–265 (2015).
Ohashi, M. et al. SAM-dependent enzyme-catalysed pericyclic reactions in natural product biosynthesis. Nature 549, 502–506 (2017).
Li, L. et al. Genome mining and assembly-line biosynthesis of the UCS1025A pyrrolizidinone family of fungal alkaloids. J. Am. Chem. Soc. 140, 2067–2071 (2018).
Kato, N. et al. Control of the stereochemical course of [4+2] cycloaddition during trans-decalin formation by fsa2-family enzymes. Angew. Chem. Int. Ed. 57, 9754–9758 (2018).
Fage, C. D. et al. The structure of SpnF, a standalone enzyme that catalyzes [4+2] cycloaddition. Nat. Chem. Biol. 11, 256–258 (2015).
Cai, Y. et al. Structural basis for stereoselective dehydration and hydrogen-bonding catalysis by the SAM-dependent pericyclase LepI. Nat. Chem. 11, 812–820 (2019).
Zheng, Q. et al. Enzyme-dependent [4+2] cycloaddition depends on lid-like interaction of the N-terminal sequence with the catalytic core in PyrI4. Cell Chem. Biol. 23, 352–360 (2016).
Byrne, M. J. et al. The catalytic mechanism of a natural Diels–Alderase revealed in molecular detail. J. Am. Chem. Soc. 138, 6095–6098 (2016).
Zheng, Q. F. et al. Structural insights into a flavin-dependent [4+2] cyclase that catalyzes trans-decalin formation in pyrroindomycin biosynthesis. Cell Chem. Biol. 25, 718–728 (2018).
Domingo, L. R., Zaragoza, R. J. & Williams, R. M. Studies on the biosynthesis of paraherquamide A and VM99955. A theoretical study of intramolecular Diels–Alder cycloaddition. J. Org. Chem. 68, 2895–2902 (2003).
Quadri, L. E. N. et al. Characterization of Sfp, a Bacillus subtilis phosphopantetheinyl transferase for peptidyl carrier protein domains in peptide synthetases. Biochemistry 37, 1585–1595 (1998).
Nodvig, C. S., Nielsen, J. B., Kogle, M. E. & Mortensen, U. H. A CRISPR-Cas9 system for genetic engineering of filamentous fungi. PLoS One 10, e0133085 (2015).
Fraley, A. E. et al. Function and structure of MalA/MalA’, iterative halogenases for late-stage C–H functionalization of indole alkaloids. J. Am. Chem. Soc. 139, 12060–12068 (2017).
Filling, C. et al. Critical residues for structure and catalysis in short-chain dehydrogenases/reductases. J. Biol. Chem. 277, 25677–25684 (2002).
Oppermann, U. et al. Short-chain dehydrogenases/reductases (SDR): the 2002 update. Chem. Biol. Interact. 143–144, 247–253 (2003).
Man, H. et al. Structures of alcohol dehydrogenases from Ralstonia and Sphingobium spp. reveal the molecular basis for their recognition of ‘bulky–bulky’ ketones. Top. Catal. 57, 356–365 (2014).
Birch, A. J. & Wright, J. J. Studies in relation to biosynthesis. XLII. The structural elucidation and some aspects of the biosynthesis of the brevianamides-A and -E. Tetrahedron 26, 2329–2344 (1970).
Acknowledgements
This work was supported by the National Institutes of Health R01 CA070375 to (R.M.W. and D.H.S.), R35 GM118101 and the Hans W. Vahlteich Professorship (to D.H.S.), and R01 DK042303 and the Margaret J. Hunter Professorship (to J.L.S.). J.N.S. and K.N.H. acknowledge support from the National Institute of General Medical Sciences of the National Institutes of Health under awards F32GM122218 (to J.N.S.) and R01GM124480 (to K.N.H.). Computational resources were provided by the UCLA Institute for Digital Research and Education (IDRE) and the Extreme Science and Engineering Discovery Environment (XSEDE), which is supported by the NSF (OCI-1053575). Anton 2 computer time was provided by the Pittsburgh Supercomputing Center (PSC) through grant no. R01GM116961 from the National Institutes of Health. The Anton 2 machine at PSC was generously made available by D.E. Shaw Research. GM/CA@APS is supported by the National Institutes of Health, National Institute of General Medical Sciences (AGM-12006) and National Cancer Institute (ACB-12002). We thank S. Ragsdale for assistance with anaerobic enzyme assays and P. Nagorny for assistance with polarimetry measurements.
Author information
Authors and Affiliations
Contributions
Q.D., S.A.N., J.L.S., R.M.W. and D.H.S. contributed to the experimental design. Q.D., S.A.N., A.E.F. and W.C.B. performed molecular cloning, protein expression and purification. Q.D., S.A.N. and A.E.F. performed all enzymatic assays and LC/MS analysis. S.A.N. and Q.D. carried out all crystallographic experiments, structural analysis and structure-based site-directed mutagenesis. K.R.K., J.D.S., A.D.S., T.J.M., L.Z., S.A.N. and V.V.S. synthesized and validated all the compounds described in this study. Y.Y. and F.Y. carried out the genetic knockout experiment, and F.Y. and Q.D. performed genetic annotation. J.N.S. and S.A.N. performed molecular dynamics simulations. R.S.P. performed DFT calculations. Q.D., S.A.N., K.N.H., J.L.S., R.M.W. and D.H.S. evaluated the data and prepared the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Methods, Figs. 1–26, Tables 1–4 (including X-ray data collection and refinement statistics) and all NMR spectra of newly synthesized compounds and references.
Rights and permissions
About this article
Cite this article
Dan, Q., Newmister, S.A., Klas, K.R. et al. Fungal indole alkaloid biogenesis through evolution of a bifunctional reductase/Diels–Alderase. Nat. Chem. 11, 972–980 (2019). https://doi.org/10.1038/s41557-019-0326-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0326-6
This article is cited by
-
An NmrA-like enzyme-catalysed redox-mediated Diels–Alder cycloaddition with anti-selectivity
Nature Chemistry (2023)
-
Modular assembly of indole alkaloids enabled by multicomponent reaction
Nature Communications (2023)
-
Tandem intermolecular [4 + 2] cycloadditions are catalysed by glycosylated enzymes for natural product biosynthesis
Nature Chemistry (2023)
-
A cyclase that catalyses competing 2 + 2 and 4 + 2 cycloadditions
Nature Chemistry (2023)
-
Selective cycloadditions
Nature Chemistry (2023)