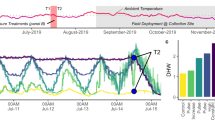
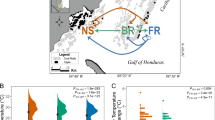
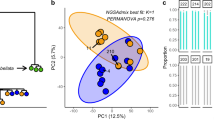
Abstract
Local adaptation is ubiquitous, but the molecular mechanisms that give rise to this ecological phenomenon remain largely unknown. A year-long reciprocal transplant of mustard hill coral (Porites astreoides) between a highly environmentally variable inshore habitat and a more stable offshore habitat demonstrated that populations exhibit phenotypic signatures that are consistent with local adaptation. We characterized the genomic basis of this adaptation in both coral hosts and their intracellular symbionts (Symbiodinium sp.) using genome-wide gene expression profiling. Populations differed primarily in their capacity for plasticity: following transplantation to a novel environment, inshore-origin coral expression profiles became significantly more similar to the local population's profiles than those in offshore-origin corals. Furthermore, elevated plasticity of the environmental stress response expression was correlated with lower susceptibility to a natural summer bleaching event, suggesting that plasticity is adaptive in the inshore environment. Our results reveal a novel genomic mechanism of resilience to a variable environment, demonstrating that corals are capable of a more diverse molecular response to stress than previously thought.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kawecki, T. J. & Ebert, D. Conceptual issues in local adaptation. Ecol. Lett. 7, 1225–1241 (2004).
Hereford, J. A quantitative survey of local adaptation and fitness trade-offs. Am. Nat. 173, 579–588 (2009).
Savolainen, O., Lascoux, M. & Merila, J. Ecological genomics of local adaptation. Nat. Rev. Genet. 14, 807–820 (2013).
Hodgins-Davis, A. & Townsend, J. P. Evolving gene expression: from G to E to GxE. Trends Ecol. Evol. 24, 649–658 (2009).
Lopez-Maury, L., Marguerat, S. & Bahler, J. Tuning gene expression to changing environments: from rapid responses to evolutionary adaptation Nat. Rev. Genet. 9, 583–593 (2008).
Whitehead, A. & Crawford, D. L. Variation within and among species in gene expression: raw material for evolution. Molec. Ecol. 15, 1197–1211 (2006).
Schadt, E. E. et al. Genetics of gene expression surveyed in maize, mouse and man. Nature 422, 297–302 (2003).
Brem, R. B. & Kruglyak, L. The landscape of genetic complexity across 5,700 gene expression traits in yeast. Proc. Natl Acad. Sci. USA 102, 1572–1577 (2004).
Whitehead, A. & Crawford, D. L. Neutral and adaptive variation in gene expression. Proc. Natl Acad. Sci. USA 103, 5425–5430 (2006).
Meier, K. et al. Local adaptation at the transcriptome level in brown trout: evidence from early life history temperature genomic reaction norms. PLoS ONE 9, e85171 (2014).
Kenkel, C. D., Almanza, A. T. & Matz, M. V. Fine-scale environmental specialization of reef-building corals might be limiting reef recovery in the Florida Keys. Ecology 96, 3197–3212 (2015).
Kenkel, C. et al. Evidence for a host role in thermotolerance divergence between populations of the mustard hill coral (Porites astreoides) from different reef environments. Mol. Ecol. 22, 4335–4348 (2013).
Kenkel, C. D., Setta, S. P. & Matz, M. V. Heritable differences in fitness-related traits among populations of the mustard hill coral, Porites astreoides. Heredity 115, 509–516 (2015).
Meyer, E., Aglyamova, G. V. & Matz, M. V. Profiling gene expression responses of coral larvae (Acropora millepora) to elevated temperature and settlement inducers using a novel RNA-Seq procedure. Mol. Ecol. 20, 3599–3616 (2011).
Lohman, B. K., Weber, J. N. & Bolnick, D. I. Evaluation of TagSeq, a reliable low-cost alternative for RNAseq . Molec. Ecol. Res. http://dx.doi.org/10.1111/1755-0998.12529 (2016).
Langfelder, P. & Horvath, S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9, 559 (2008).
Jombart, T., Devillard, S. & Balloux, F. Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet. 11, 94 (2010).
Langfelder, P. & Horvath, S. Tutorials for the WGCNA Package (UCLA, 2014); http://labs.genetics.ucla.edu/horvath/CoexpressionNetwork/Rpackages/WGCNA/Tutorials
Boyer, J., Briceño, H., Absten, J., Gilliam, D. & Dodge, D. 2011 Annual Report of the Water Quality Monitoring Project for the Southeast Florida Coral Reef Initiative (SERC Research Reports 72, Southeast Environmental Research Center, 2011); http://digitalcommons.fiu.edu/sercrp/72
Yonge, C. M. & Nicholls, A. G. in Great Barrier Reef Expedition 1928–1929 Vol. 1, 135–176 (British Museum, 1931).
Barshis, D. et al. Genomic basis for coral resilience to climate change. Proc. Natl Acad. Sci. USA 110, 1387–1392 (2013).
Berkelmans, R. & van Oppen, M. J. H. The role of zooxanthellae in the thermal tolerance of corals: a ‘nugget of hope’ for coral reefs in an era of climate change. Proc. R. Soc. B 273, 2305–2312 (2006).
Oliver, T. A. & Palumbi, S. R. Many corals host thermally resistant symbionts in high-temperature habitat. Coral Reefs 30, 241–250 (2011).
Weis, V. M. Cellular mechanisms of Cnidarian bleaching: stress causes the collapse of symbiosis. J. Exp. Biol. 211, 3059–3066 (2008).
Hill, R. & Ralph, P. J. Photosystem II heterogeneity of in hospite zooxanthellae in scleractinian corals exposed to bleaching conditions. Photochem. Photobiol. 82, 1577–1585 (2006).
Schlichting, C. D. & Pigliucci, M. Phenotypic Evolution: a Reaction Norm Perspective (Sinauer, 1998).
Aubin-Horth, N. & Renn, S. C. Genomic reaction norms: using integrative biology to understand molecular mechanisms of phenotypic plasticity. Mol. Ecol. 18, 3763–3780 (2009).
S⊘rensen, J. G., Kristensen, T. N. & Loeschcke, V. The evolutionary and ecological role of heat shock proteins. Ecol. Lett. 6, 1025–1037 (2003).
Van Tienderen, P. H. Evolution of generalists and specialists in spatially heterogenous environments. Evolution 45, 1317–1331 (1991).
DeWitt, T. J., Sih, A. & Wilson, D. S. Costs and limits of phenotypic plasticity. Trends Ecol. Evol. 13, 77–81 (1998).
Hendry, A. P. Key questions on the role of phenotypic plasticity in eco-evolutionary dynamics. J. Hered. 107, 25–41 (2016).
Draghi, J. A. & Whitlock, M. C. Phenotypic plasticity facilitates mutational variance, genetic variance, and evolvability along the major axis of environmental variation. Evolution 66, 2891–2902 (2012).
Yeh, P. J. & Price, T. D. Adaptive phenotypic plasticity and the successful colonization of a novel environment. Am. Nat. 164, 531–542 (2004).
Kenkel, C. et al. Development of gene expression markers of acute heat-light stress in reef-building corals of the genus Porites. PLoS ONE 6, e26914 (2011).
Kenkel, C., Meyer, E. & Matz, M. Gene expression under chronic heat stress in populations of the mustard hill coral (Porites astreoides) from different thermal environments. Mol. Ecol. 22, 4322–4334 (2013).
Bayer, T. et al. Symbiodinium transcriptomes: genome insights into the dinoflagellate symbionts of reef-building corals. PLoS ONE 7, e35269 (2012).
Thornhill, D. J., Fitt, W. K. & Schmidt, G. W. Highly stable symbioses among western Atlantic brooding corals. Coral Reefs 25, 515–519 (2006).
Langmead, B. & Salzbert, S. Fast gapped-read alignment with Bowtie 2. Nat. Meth. 9, 357–359 (2012).
The R Core Team. R A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-Seq data with DESeq2. Genome Biol. 15, 550 (2014).
Jombart, T. adegenet: an R package for the multivariate analysis of genetic markers. Bioinformatics 24, 1403–1405 (2008).
Hadfield, J. D. MCMC methods for multi-response generalized linear mixed models: the MCMCglmm R package. J. Stat. Softw. 33, 1–22 (2010).
Langfelder, P. & Horvath, S. Fast R functions for robust correlations and hierarchical clustering. J. Stat. Softw. 46, 1–17 (2012).
The Gene Ontology Consortium. Gene ontology: tool for the unification of biology. Nat. Genet. 25, 25–29 (2000).
Acknowledgements
G. Aglyamova prepared the library of samples for Illumina sequencing. M. Strader created the GIS map of the Florida Keys. Bioinformatic analyses were carried out using the computational resources of the Texas Advanced Computing Center (TACC). Funding for this study was provided by NSF DDIG award DEB-1311220 to C.D.K. and M.V.M.
Author information
Authors and Affiliations
Contributions
C.D.K. and M.V.M. conceived and designed experiments. C.D.K. performed experiments, analysed data and wrote the first draft of the manuscript. Both authors contributed to revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–8 and Supplementary Table 1 (PDF 1053 kb)
Rights and permissions
About this article
Cite this article
Kenkel, C., Matz, M. Gene expression plasticity as a mechanism of coral adaptation to a variable environment. Nat Ecol Evol 1, 0014 (2017). https://doi.org/10.1038/s41559-016-0014
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41559-016-0014
This article is cited by
-
Gene expression plasticity facilitates acclimatization of a long-lived Caribbean coral across divergent reef environments
Scientific Reports (2024)
-
Genetically encoded transcriptional plasticity underlies stress adaptation in Mycobacterium tuberculosis
Nature Communications (2024)
-
Local habitat heterogeneity rivals regional differences in coral thermal tolerance
Coral Reefs (2024)
-
Bio-optical signatures of in situ photosymbionts predict bleaching severity prior to thermal stress in the Caribbean coral species Acropora palmata
Coral Reefs (2024)
-
Multi-omic characterization of mechanisms contributing to rapid phenotypic plasticity in the coral Acropora cervicornis under divergent environments
Coral Reefs (2024)