Abstract
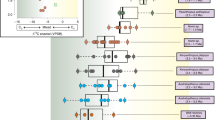
Evolutionary radiations generate most of Earth’s biodiversity, but are there common ecomorphological traits among the progenitors of radiations? In Synapsida (the mammalian total group), ‘small-bodied faunivore’ has been hypothesized as the ancestral state of most major radiating clades, but this has not been quantitatively assessed across multiple radiations. To examine macroevolutionary patterns in a phylogenetic context, we generated a time-calibrated metaphylogeny (‘metatree’) comprising 1,888 synapsid species from the Carboniferous through the Eocene (305–34 Ma) based on 269 published character matrices. We used comparative methods to investigate body size and dietary evolution during successive synapsid radiations. Faunivory is the ancestral dietary regime of each major synapsid radiation, but relatively small body size is only established as the common ancestral state of radiations near the origin of Mammaliaformes in the Late Triassic. The faunivorous ancestors of synapsid radiations typically have numerous novel characters compared with their contemporaries, and these derived traits may have helped them to survive faunal turnover events and subsequently radiate.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data used in this study are available through the main tables and the Supplementary Information.
Code availability
A simplified version of the R code used for this study is available as a supplementary text file. The code used to create the metatree is available at https://github.com/graemetlloyd/metatree.
References
Rabosky, D. L. & Lovette, I. J. Explosive evolutionary radiations: decreasing speciation or increasing extinction through time? Evolution 62, 1866–1875 (2008).
Alfaro, M. E. et al. Nine exceptional radiations plus high turnover explain species diversity in jawed vertebrates. Proc. Natl Acad. Sci. USA 106, 13410–13414 (2009).
O’Leary, M. A. et al. The placental mammal ancestor and the post-K–Pg radiation of placentals. Science 339, 662–667 (2013).
Grossnickle, D. M. & Newham, E. Therian mammals experience an ecomorphological radiation during the Late Cretaceous and selective extinction at the K–Pg boundary. Proc. R. Soc. B 283, 20160256 (2016).
Bouchenak-Khelladi, Y., Onstein, R. E., Xing, Y., Schwery, O. & Linder, H. P. On the complexity of triggering evolutionary radiations. New Phytol. 207, 313–326 (2015).
Cope, E. D. The Primary Factors of Organic Evolution (Open Court Publishing, 1896).
Stanley, S. M. An explanation for Cope’s rule. Evolution 27, 1–26 (1973).
Pough, F. H. Lizard energetics and diet. Ecology 54, 837–844 (1973).
Gaulin, S. J. A Jarman/Bell model of primate feeding niches. Hum. Ecol. 7, 1–20 (1979).
Tomiya, S. Body size and extinction risk in terrestrial mammals above the species level. Am. Nat. 182, E196–E214 (2013).
Zanno, L. E. & Makovicky, P. J. No evidence for directional evolution of body mass in herbivorous theropod dinosaurs. Proc. R. Soc. Lond. B 280, 20122526 (2013).
Reisz, R. R. & Fröbisch, J. The oldest caseid synapsid from the Late Pennsylvanian of Kansas, and the evolution of herbivory in terrestrial vertebrates. PLoS ONE 9, e94518 (2014).
Price, S. A. & Hopkins, S. S. The macroevolutionary relationship between diet and body mass across mammals. Biol. J. Linn. Soc. 115, 173–184 (2015).
Pineda-Munoz, S., Evans, A. R. & Alroy, J. The relationship between diet and body mass in terrestrial mammals. Paleobiology 42, 659–669 (2016).
Brocklehurst, N. & Brink, K. S. Selection towards larger body size in both herbivorous and carnivorous synapsids during the Carboniferous. FACETS 2, 68–84 (2017).
Grossnickle, D. M. Feeding ecology has a stronger evolutionary influence on functional morphology than on body mass in mammals. Evolution 74, 610–628 (2020).
Simpson, G. G. Tempo and Mode in Evolution (Columbia Univ. Press, 1944).
Liow, L. H. A test of Simpson’s “Rule of the Survival of the Relatively Unspecialized” using fossil crinoids. Am. Nat. 164, 431–443 (2004).
Liow, L. H. Lineages with long durations are old and morphologically average: an analysis using multiple datasets. Evolution 61, 885–901 (2007).
Smits, P. D. Expected time-invariant effects of biological traits on mammal species duration. Proc. Natl Acad. Sci. USA 112, 13015–13020 (2015).
Raia, P. & Fortelius, M. Cope’s law of the unspecialized, Cope’s rule, and weak directionality in evolution. Evol. Ecol. Res. 15, 747–756 (2013).
Raia, P. et al. Progress to extinction: increased specialisation causes the demise of animal clades. Sci. Rep. 6, 30965 (2016).
Angielczyk, K.D. & Kammerer, C. F. in Handbook of Zoology: Mammalia: Mammalian Evolution, Diversity and Systematics (eds Zachos, F. E. & Asher, R. J.) 117–198 (De Gruyter, 2018).
Osborn, H. F. The law of adaptive radiation. Am. Nat. 36, 353–363 (1902).
Broom, R. 1932. The Mammal-like Reptiles of South Africa and the Origin of Mammals (H. F. & G. Witherby, 1932).
Hopson, J. A. The origin and adaptive radiation of mammal-like reptiles and nontherian mammals. Ann. N. Y. Acad. Sci. 167, 199–216 (1969).
Kemp, T. S. The Origin and Evolution of Mammals (Oxford Univ. Press, 2005).
Grossnickle, D. M., Smith, S. M. & Wilson, G. P. Untangling the multiple ecological radiations of early mammals. Trends Ecol. Evol. 34, 936–949 (2019).
Alroy, J. Cope’s rule and the dynamics of body mass evolution in North American fossil mammals. Science 280, 731–734 (1998).
Raia, P., Carotenuto, F., Passaro, F., Fulgione, D. & Fortelius, M. Ecological specialization in fossil mammals explains Cope’s rule. Am. Nat. 179, 328–337 (2012).
Polly, P. D. Cope’s rule. Science 282, 50–52 (1998).
Lloyd, G. T. et al. Probabilistic divergence time estimation without branch lengths: dating the origins of dinosaurs, avian flight, and crown birds. Biol. Lett. 12, 20160609 (2016).
Wilson, G. P. et al. Adaptive radiation of multituberculate mammals before the extinction of dinosaurs. Nature 483, 457–460 (2012).
Grossnickle, D. M. & Polly, P. D. Mammal disparity decreases during the Cretaceous angiosperm radiation. Proc. R. Soc. Lond. B 280, 20132110 (2013).
Hu, Y., Meng, J., Wang, Y. & Li, C. Large Mesozoic mammals fed on young dinosaurs. Nature 433, 149–152 (2005).
Luo, Z.-X. et al. New evidence for mammaliaform ear evolution and feeding adaptation in a Jurassic ecosystem. Nature 548, 326–329 (2017).
Chen, M., Strömberg, C. A. & Wilson, G. P. Assembly of modern mammal community structure driven by Late Cretaceous dental evolution, rise of flowering plants, and dinosaur demise. Proc. Natl Acad. Sci. USA 116, 9931–9940 (2019).
Morales-García, N. M., Gill, P. G., Janis, C. M. & Rayfield, E. J. Jaw shape and mechanical advantage are indicative of diet in Mesozoic mammals. Commun. Biol. 4, 242 (2021).
Krause, D. W. et al. First cranial remains of a gondwanatherian mammal reveal remarkable mosaicism. Nature 515, 512–517 (2014).
Harper, T., Parras, A. & Rougier, G. W. Reigitherium (Meridiolestida, Mesungulatoidea) an enigmatic Late Cretaceous mammal from Patagonia, Argentina: morphology, affinities, and dental evolution.J. Mamm. Evol. 26, 447–478 (2019).
Peters, R. H. The Ecological Implications of Body Size (Cambridge Univ. Press, 1986).
Angielczyk, K. D. & Walsh, M. L. Patterns in the evolution of nares size and secondary palate length in anomodont therapsids (Synapsida): implications for hypoxia as a cause of end-Permian tetrapod extinctions. J. Paleontol. 82, 528–542 (2008).
Huttenlocker, A. K. Body size reductions in nonmammalian eutheriodont therapsids (Synapsida) during the end-Permian mass extinction. PLoS ONE 9, e87553 (2014).
Grunert, H. R., Brocklehurst, N. & Fröbisch, J. Diversity and disparity of Therocephalia: macroevolutionary patterns through two mass extinctions. Sci. Rep. 9, 5063 (2019).
Wilson, G. P. Mammals across the K/Pg boundary in northeastern Montana, USA: dental morphology and body-size patterns reveal extinction selectivity and immigrant-fueled ecospace filling. Paleobiology 39, 429–469 (2013).
Tolchard, F. et al. A new large gomphodont from the Triassic of South Africa and its implications for Gondwanan biostratigraphy. J. Vertebr. Paleontol. 41, e1929265 (2021).
Sookias, R. B., Butler, R. J. & Benson, R. B. J. Rise of dinosaurs reveals major body-size transitions are driven by passive processes of trait evolution. Proc. R. Soc. Lond. B 279, 2180–2187 (2012).
Sidor, C. A. Simplification as a trend in synapsid cranial evolution. Evolution 55, 1419–1442 (2001).
Sidor, C. A. Evolutionary trends and the origin of the mammalian lower jaw. Paleobiology 29, 605–640 (2003).
Kielan-Jaworowska, Z., Cifelli, R. L. & Luo, Z.-X. Mammals from the Age of Dinosaurs: Origins, Evolution, and Structure (Columbia Univ. Press, 2004).
Rowe, T. B., Macrini, T. E. & Luo, Z.-X. Fossil evidence on origin of the mammalian brain. Science 332, 955–957 (2011).
Benoit, J., Abdala, F., Manger, P. R. & Rubidge, B. S. The sixth sense in mammalian forerunners: variability of the parietal foramen and the evolution of the pineal eye in South African Permo-Triassic eutheriodont therapsids. Acta Palaeontol. Pol. 61, 777–789 (2016).
Rey, K. et al. Oxygen isotopes suggest elevated thermometabolism within multiple Permo-Triassic therapsid clades. eLife 6, e28589 (2017).
Guignard, M. L., Martinelli, A. G. & Soares, M. B. Reassessment of the postcranial anatomy of Prozostrodon brasiliensis and implications for postural evolution of non-mammaliaform cynodonts. J. Vertebr. Paleontol. 38, e1511570 (2018).
Guignard, M. L., Martinelli, A. G. & Soares, M. B. The postcranial anatomy of Brasilodon quadrangularis and the acquisition of mammaliaform traits among non-mammaliaform cynodonts. PLoS ONE 14, e0216672 (2019).
Benoit, J. et al. The evolution of the maxillary canal in Probainognathia (Cynodontia, Synapsida): reassessment of the homology of the infraorbital foramen in mammalian ancestors. J. Mamm. Evol. 27, 329–348 (2020).
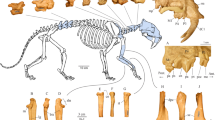
Jones, K. E., Gonzalez, S., Angielczyk, K. D. & Pierce, S. E. Regionalization of the axial skeleton predates functional adaptation in the forerunners of mammals. Nat. Ecol. Evol. 4, 470–478 (2020).
Luo, Z.-X. & Manley, G. A. in The Senses: A Comprehensive Reference Vol. 2 (eds Fritzsch, B. & Grothe, B.) 207–252 (Elsevier Academic Press, 2020).
Araújo, R. et al. Inner ear biomechanics reveals a Late Triassic origin for mammalian endothermy. Nature 607, 726–731 (2022).
Lautenschlager, S. et al. The role of miniaturization in the evolution of the mammalian jaw and middle ear. Nature 561, 533–537 (2018).
Hopson, J. A. Endothermy, small size, and the origin of mammalian reproduction. Am. Nat. 107, 446–452 (1973).
McNab, B. K. The evolution of endothermy in the phylogeny of mammals. Am. Nat. 112, 1–21 (1978).
West, G. B., Woodruff, W. H. & Brown, J. H. Allometric scaling of metabolic rate from molecules and mitochondria to cells and mammals. Proc. Natl Acad. Sci. USA 99, 2473–2478 (2002).
McShea, D. W. Mechanisms of large-scale evolutionary trends. Evolution 48, 1747–1763 (1994).
Alroy, J. Understanding the dynamics of trends within evolving lineages. Paleobiology 26, 319–329 (2000).
Lillegraven, J. A., Kielan-Jaworowska, Z. & Clemens, W. A. Mesozoic Mammals: The First Two-thirds of Mammalian History (Univ. of California Press, 1979).
Alroy, J. The fossil record of North American mammals: evidence for a Paleocene evolutionary radiation. Syst. Biol. 48, 107–118 (1999).
Smith, F. A. et al. The evolution of maximum body size of terrestrial mammals. Science 330, 1216–1219 (2010).
Slater, G. J. Phylogenetic evidence for a shift in the mode of mammalian body size evolution at the Cretaceous-Palaeogene boundary. Methods Ecol. Evol. 4, 734–744 (2013).
Clauset, A. & Redner, S. Evolutionary model of species body mass diversification. Phys. Rev. Lett. 102, 038103 (2009).
Brocklehurst, N., Panciroli, E., Benevento, G. L. & Benson, R. B. Mammaliaform extinctions as a driver of the morphological radiation of Cenozoic mammals. Curr. Biol. 31, 2955–2963 (2021).
Lloyd, G. T. et al. Dinosaurs and the Cretaceous terrestrial revolution. Proc. R. Soc. Lond. B 275, 2483–2490 (2008).
Lyson, T. R. et al. Exceptional continental record of biotic recovery after the Cretaceous–Paleogene mass extinction. Science 366, 977–983 (2019).
Weaver, L. N. & Wilson, G. P. Shape disparity in the blade-like premolars of multituberculate mammals: functional constraints and the evolution of herbivory. J. Mammal. 102, 967–985 (2021).
Gill, P. G. et al. Dietary specializations and diversity in feeding ecology of the earliest stem mammals. Nature 512, 303–305 (2014).
Conith, A. J., Imburgia, M. J., Crosby, A. J. & Dumont, E. R. The functional significance of morphological changes in the dentitions of early mammals. J. R. Soc. Interface 13, 20160713 (2016).
Schultz, J. A. & Martin, T. Function of pretribosphenic and tribosphenic mammalian molars inferred from 3D animation. Naturwissenschaften 101, 771–781 (2014).
Jäger, K. R., Cifelli, R. L. & Martin, T. Molar occlusion and jaw roll in early crown mammals. Sci. Rep. 10, 22378 (2020).
Williams, S. H. et al. A preliminary analysis of correlated evolution in mammalian chewing motor patterns. Integr. Comp. Biol. 51, 247–259 (2011).
Grossnickle, D. M. The evolutionary origin of jaw yaw in mammals. Sci. Rep. 7, 45094 (2017).
Bhullar, B. A. S. et al. Rolling of the jaw is essential for mammalian chewing and tribosphenic molar function. Nature 566, 528–532 (2019).
Grossnickle, D. M., Weaver, L. N., Jäger, K. R. & Schultz, J. A. The evolution of anteriorly directed molar occlusion in mammals. Zool. J. Linn. Soc. 194, 349–365 (2022).
Higashiyama, H. et al. Mammalian face as an evolutionary novelty. Proc. Natl Acad. Sci. USA 118, e2111876118 (2021).
Sidor, C. A. & Hopson, J. A. Ghost lineages and “mammalness”: assessing the temporal pattern of character acquisition in the Synapsida. Paleobiology 24, 254–273 (1998).
Kemp, T. S. The origin and early radiation of the therapsid mammal‐like reptiles: a palaeobiological hypothesis. J. Evol. Biol. 19, 1231–1247 (2006).
Hopson, J. A. & Kitching, J. W. A probainognathian cynodont from South Africa and the phylogeny of nonmammalian cynodonts. Bull. Mus. Comp. Zool. 156, 5–35 (2001).
Brocklehurst, N., Ruta, M., Müller, J. & Fröbisch, J. Elevated extinction rates as a trigger for diversification rate shifts: early amniotes as a case study. Sci. Rep. 5, 17104 (2015).
Jablonski, D. Approaches to macroevolution: 1. General concepts and origin of variation. Evol. Biol. 44, 427–450 (2017).
Luo, Z.-X. et al. A Jurassic eutherian mammal and divergence of marsupials and placentals. Nature 476, 442–445 (2011).
Chen, Z.-Q. & Benton, M. J. The timing and pattern of biotic recovery following the end-Permian mass extinction. Nat. Geosci. 5, 375–383 (2012).
Benton, M. J. & Newell, A. J. Impacts of global warming on Permo-Triassic terrestrial ecosystems. Gondwana Res. 25, 1308–1337 (2014).
Benton, M. J. When Life Nearly Died: The Greatest Mass Extinction of All Time (Thames & Hudson, 2015).
Botha, J. & Smith, R. M. H. Lystrosaurus species composition across the Permo-Triassic boundary of South Africa. Lethaia 40, 125–137 (2007).
Botha-Brink, J. & Abdala, F. A new cynodont record from the Tropidostoma Assemblage Zone of the Beaufort Group: implications for the early evolution of cynodonts in South Africa. Palaeontol. Afr. 43, 1–6 (2008).
Abdala, F. & Ribeiro, A. M. Distribution and diversity patterns of Triassic cynodonts (Therapsida, Cynodontia) in Gondwana. Palaeogeogr. Palaeoclimatol. Palaeoecol. 286, 202–217 (2010).
Irmis, R. B. & Whiteside, J. H. Delayed recovery of non-marine tetrapods after the end-Permian mass extinction tracks global carbon cycle. Proc. R. Soc. Lond. B 279, 1310–1318 (2012).
Botha-Brink, J., Abdala, F. & Chinsamy-Turan, A. in Forerunners of Mammals: Radiation, Histology, Biology (ed Chinsamy-Turan, A.) 223–246 (Indiana Univ. Press, 2012).
Lloyd, G. T. & Slater, G. J. A total-group phylogenetic metatree for Cetacea and the importance of fossil data in diversification analyses. Syst. Biol. 70, 922–939 (2021).
Lloyd, G. T. Estimating morphological diversity and tempo with discrete character-taxon matrices: implementation, challenges, progress, and future directions. Biol. J. Linn. Soc. 118, 131–151 (2016).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2021).
Goloboff, P. A., Farris, J. S. & Nixon, K. C. TNT, a free program for phylogenetic analysis. Cladistics 24, 774–786 (2008).
Burgin, C. J., Colella, J. P., Kahn, P. L. & Upham, N. S. How many species of mammals are there? J. Mammal. 99, 1–14 (2018).
Peters, S. E. & McClennen, M. The Paleobiology Database application programming interface. Paleobiology 42, 1–7 (2016).
Kammerer, C. F., Angielczyk, K. D. & Frobisch, J. A comprehensive taxonomic revision of Dicynodon (Therapsida, Anomodontia) and its implications for dicynodont phylogeny, biogeography, and biostratigraphy. J. Vertebr. Paleontol. 11, 1–158 (2011).
Bapst, D. W. paleotree: an R package for paleontological and phylogenetic analyses of evolution. Methods Ecol. Evol. 3, 803–807 (2012).
Jones, K. E., Angielczyk, K. D. & Pierce, S. E. Stepwise shifts underlie evolutionary trends in morphological complexity of the mammalian vertebral column. Nat. Commun. 10, 5071 (2019).
Pinheiro, J. et al. nlme: Linear and nonlinear mixed effects models, R package v.3.1–140 (2017); https://CRAN.R-project.org/package=nlme
Hörandl, E. Beyond cladistics: extending evolutionary classifications into deeper time levels. Taxon 59, 345–350 (2010).
Lungmus, J. K. & Angielczyk, K. D. Phylogeny, function and ecology in the deep evolutionary history of the mammalian forelimb. Proc. R. Soc. Lond. B 288, 20210494 (2021).
Brocklehurst, N. & Benson, R. B. J. Multiple paths to morphological diversification during the origin of amniotes. Nat. Ecol. Evol. 5, 1243–1249 (2021).
Carroll, R. L. The earliest reptiles. Zool. J. Linn. Soc. 45, 61–83 (1964).
Clavel, J., Escarguel, G. & Merceron, G. mvMORPH: an R package for fitting multivariate evolutionary models to morphometric data. Methods Ecol. Evol. 6, 1311–1319 (2015).
Hansen, T. F. Stabilizing selection and the comparative analysis of adaptation. Evolution 51, 1341–1351 (1997).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference: A Practical Information-theoretic Approach (Springer, 2002).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Acknowledgements
For discussions and feedback, we thank G. Slater, G. W. Mantilla, L. Weaver, S. Santana, K.-D. Benton, A. Bormet, C. Law and E. Panciroli. This work was funded by the National Science Foundation: DEB-1754502 (to K.D.A) and DBI-1812126 (to D.M.G.).
Author information
Authors and Affiliations
Contributions
S.M.H., D.M.G. and K.D.A. designed the study. S.M.H., G.T.L. and D.M.G. produced the metatree phylogeny. All authors (S.M.H., D.M.G., G.T.L., C.F.K. and K.D.A.) helped in collecting data and writing the paper. S.M.H. and D.M.G. performed analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks John Alroy and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
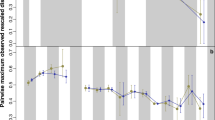
Extended Data Fig. 1 Jaw length (log10 mm) and diet regressed against patristic distance for pelycosaur subclades.
The dashed lines are used for clades in which there is only a single diet. Grey bands are 95% confidence intervals.
Extended Data Fig. 2 Jaw length (log10 mm) and diet regressed against patristic distance for therapsid subclades.
We do not include a quasibinomial model for Therocephalia because the model algorithm did not converge on a reasonable result; we instead present a linear model (dashed line). We did not fit regression models for diets of Biarmosuchia or Gorgonopsia because their members all have the same diets – the dashed lines are included to help emphasize the lack of diet change. Grey bands are 95% confidence intervals.
Extended Data Fig. 3 Jaw length (log10 mm) and diet regressed against patristic distance for cynodont subclades.
We do not include a quasibinomial model for Probainognathia because the model algorithm did not converge on a reasonable result; we instead present a linear model (dashed line). Grey bands are 95% confidence intervals.
Extended Data Fig. 4 Jaw length (log10 mm) and diet regressed against patristic distance for mammaliaform subclades.
The dashed lines are used for clades in which there is only a single diet. Grey bands are 95% confidence intervals.
Extended Data Fig. 5 Jaw length (log10 mm) and diet regressed against patristic distance for therian subclades.
Grey bands are 95% confidence intervals.
Extended Data Fig. 6 Diet (A) and jaw lengths (B) for all synapsids in our sample are plotted against patristic distance.
These are the similar plots to those in Figs. 1B and C, but the subclades (rather than five major radiations) are highlighted in this figure. The smaller, straight lines represent linear regressions for synapsid subclades, and the grey curves are LOESS fitted regression curves (fitted to all data) and associated 95% confidence interval bands. For diet, we plot linear models rather than quasibinomial models (such as in Fig. 2) because quasibinomial models could not be fit to many of the subclades (for example, see Extended Data Fig. 1–5). Multituberculates are not included in this plot as a separate group, but they are the major clade within Allotheria (Extended Data Fig. 4).
Supplementary information
Supplementary Information
Supplementary Methods, Results, Fig. 1 and Tables 1–7.
Supplementary Tables 6 and 7
Supplementary Table 6 Morphological character matrices used to create the phylogenetic metatree. Supplementary Table 7 Morphometric, diet and geologic age information on the sampled species (n = 404).
Supplementary Data 1
Nexus file of the dated majority-rule metatree for the sampled species (n = 404).
Supplementary Data 2
Nexus file of the full dated majority-rule metatree.
Supplementary Data 3
Nexus file of the full undated majority-rule metatree.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hellert, S.M., Grossnickle, D.M., Lloyd, G.T. et al. Derived faunivores are the forerunners of major synapsid radiations. Nat Ecol Evol 7, 1903–1913 (2023). https://doi.org/10.1038/s41559-023-02200-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-023-02200-y
This article is cited by
-
Predatory synapsid ecomorphology signals growing dynamism of late Palaeozoic terrestrial ecosystems
Communications Biology (2024)