Abstract
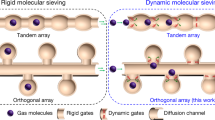
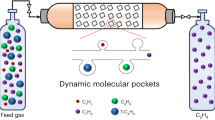
Molecular sieving can lead to ultrahigh selectivity and low regeneration energy because it completely excludes all larger molecules via a size restriction mechanism. However, it allows adsorption of all molecules smaller than the pore aperture and so separations of complicated mixtures can be hindered. Here, we report an intermediate-sized molecular sieving (iSMS) effect in a metal–organic framework (MAF-41) designed with restricted flexibility, which also exhibits superhydrophobicity and ultrahigh thermal/chemical stabilities. Single-component isotherms and computational simulations show adsorption of styrene but complete exclusion of the larger analogue ethylbenzene (because it exceeds the maximal aperture size) and smaller toluene/benzene molecules that have insufficient adsorption energy to open the cavity. Mixture adsorption experiments show a high styrene selectivity of 1,250 for an ethylbenzene/styrene mixture and 3,300 for an ethylbenzene/styrene/toluene/benzene mixture (orders of magnitude higher than previous reports). This produces styrene with a purity of 99.9%+ in a single adsorption–desorption cycle. Controlling/restricting flexibility is the key for iSMS and can be a promising strategy for discovering other exceptional properties.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other finding of this study are available from the corresponding authors upon reasonable request. CCDC 1581460−1581461 entries contain supplementary single-crystal X-ray diffraction data for 1·pX and structural details of 1′. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
References
Kirk-Othmer. Kirk-Othmer Encyclopedia of Chemical Technology 5th edn (Wiley, 2007).
Maes, M. et al. Separation of styrene and ethylbenzene on metal–organic frameworks: analogous structures with different adsorption mechanisms. J. Am. Chem. Soc. 132, 15277–15285 (2010).
Fritz, U. Ullmann’s Encyclopedia of Industrial Chemistry 7th edn (Wiley, 2011).
Mohanty, S. & McCormick, A. V. Prospects for principles of size and shape selective separations using zeolites. Chem. Eng. J. 74, 1–14 (1999).
Nugent, P. et al. Porous materials with optimal adsorption thermodynamics and kinetics for CO2 separation. Nature 495, 80–84 (2013).
Wang, Y. & Zhao, D. Beyond equilibrium: metal–organic frameworks for molecular sieving and kinetic gas separation. Cryst. Growth Des. 17, 2291–2308 (2017).
Kuznicki, S. M. et al. A titanosilicate molecular sieve with adjustable pores for size-selective adsorption of molecules. Nature 412, 720–724 (2001).
Bereciartua, P. J. et al. Control of zeolite framework flexibility and pore topology for separation of ethane and ethylene. Science 358, 1068–1071 (2017).
Li, B. et al. An ideal molecular sieve for acetylene removal from ethylene with record selectivity and productivity. Adv. Mater. 29, 201704210 (2017).
Aguado, S., Bergeret, G., Daniel, C. & Farrusseng, D. Absolute molecular sieve separation of ethylene/ethane mixtures with silver zeolite A. J. Am. Chem. Soc. 134, 14635–14637 (2012).
Cadiau, A., Adil, K., Bhatt, P. M., Belmabkhout, Y. & Eddaoudi, M. A metal–organic framework-based splitter for separating propylene from propane. Science 353, 137–140 (2016).
Lin, R.-B. et al. Molecular sieving of ethylene from ethane using a rigid metal–organic framework. Nat. Mater. 17, 1128–1133 (2018).
Assen, A. H. et al. Ultra-tuning of the rare-earth fcu-MOF aperture size for selective molecular exclusion of branched paraffins. Angew. Chem. Int. Ed. 54, 14353–14358 (2015).
Zhang, Z. et al. Sorting of C4 olefins with interpenetrated hybrid ultramicroporous materials by combining molecular recognition and size-sieving. Angew. Chem. Int. Ed. 56, 16282–16287 (2017).
Jin, Z. et al. A novel microporous MOF with the capability of selective adsorption of xylenes. Chem. Commun. 46, 8612–8614 (2010).
Lai, Z. et al. Microstructural optimization of a zeolite membrane for organic vapor separation. Science 300, 456–460 (2003).
Kolvenbach, R. et al. A comparative study of diffusion of benzene/p-xylene mixtures in MFI particles, pellets and grown membranes. Catal. Today 168, 147–157 (2011).
Islamoglu, T. et al. Postsynthetic tuning of metal–organic frameworks for targeted applications. Acc. Chem. Res. 50, 805–813 (2017).
Zhai, Q. G., Bu, X., Zhao, X., Li, D. S. & Feng, P. Pore space partition in metal–organic frameworks. Acc. Chem. Res. 50, 407–417 (2017).
Evans, J. D., Sumby, C. J. & Doonan, C. J. Post-synthetic metalation of metal–organic frameworks. Chem. Soc. Rev. 43, 5933–5951 (2014).
Verploegh, R. J., Nair, S. & Sholl, D. S. Temperature and loading-dependent diffusion of light hydrocarbons in ZIF-8 as predicted through fully flexible molecular simulations. J. Am. Chem. Soc. 137, 15760–15771 (2015).
Zhang, C. et al. Unexpected molecular sieving properties of zeolitic imidazolate framework-8. J. Phys. Chem. Lett. 3, 2130–2134 (2012).
Mukherjee, S. et al. Framework-flexibility driven selective sorption of p-xylene over other isomers by a dynamic metal–organic framework. Sci. Rep. 4, 5761 (2014).
Schneemann, A. et al. Flexible metal–organic frameworks. Chem. Soc. Rev. 43, 6062–6096 (2014).
Zhang, J.-P., Zhou, H.-L., Zhou, D.-D., Liao, P.-Q. & Chen, X.-M. Controlling flexibility of metal–organic frameworks. Natl Sci. Rev. 5, 907–919 (2018).
Tanaka, D. et al. Kinetic gate-opening process in a flexible porous coordination polymer. Angew. Chem. Int. Ed. 47, 3914–3918 (2008).
Gu, C. et al. Design and control of gas diffusion process in a nanoporous soft crystal. Science 363, 387–391 (2019).
Hamon, L. et al. Co-adsorption and separation of CO2-CH4 mixtures in the highly flexible MIL-53(Cr) MOF. J. Am. Chem. Soc. 131, 17490–17499 (2009).
DeVries, L. D., Barron, P. M., Hurley, E. P., Hu, C. & Choe, W. ‘Nanoscale lattice fence’ in a metal–organic framework: interplay between hinged topology and highly anisotropic thermal response. J. Am. Chem. Soc. 133, 14848–14851 (2011).
Zhou, D.-D. et al. A flexible porous Cu(ii) bis-imidazolate framework with ultrahigh concentration of active sites for efficient and recyclable CO2 capture. Chem. Commun. 49, 11728–11730 (2013).
Zhang, J.-P. & Kitagawa, S. Supramolecular isomerism, framework flexibility, unsaturated metal center, and porous property of Ag(i)/Cu(i) 3,3', 5,5'-tetrametyl-4,4'-bipyrazolate. J. Am. Chem. Soc. 130, 907–917 (2008).
Loiseau, T. et al. A rationale for the large breathing of the porous aluminum terephthalate (MIL-53) upon hydration. Chem. Eur. J. 10, 1373–1382 (2004).
Jiang, H. L. et al. An exceptionally stable, porphyrinic Zr metal–organic framework exhibiting pH-dependent fluorescence. J. Am. Chem. Soc. 135, 13934–13938 (2013).
Li, R. J., Li, M., Zhou, X. P., Li, D. & O'Keeffe, M. A highly stable MOF with a rod SBU and a tetracarboxylate linker: unusual topology and CO2 adsorption behaviour under ambient conditions. Chem. Commun. 50, 4047–4049 (2014).
Li, J. R., Kuppler, R. J. & Zhou, H. C. Selective gas adsorption and separation in metal–organic frameworks. Chem. Soc. Rev. 38, 1477–1504 (2009).
He, C.-T. et al. Hyperfine adjustment of flexible pore-surface pockets enables smart recognition of gas size and quadrupole moment. Chem. Sci. 8, 7560–7565 (2017).
Alaerts, L. et al. Selective adsorption and separation of xylene isomers and ethylbenzene with the microporous vanadium(iv) terephthalate MIL-47. Angew. Chem. Int. Ed. 46, 4293–4297 (2007).
Liao, P.-Q., Huang, N.-Y., Zhang, W.-X., Zhang, J.-P. & Chen, X.-M. Controlling guest conformation for efficient purification of butadiene. Science 356, 1193–1196 (2017).
Jie, K. et al. Styrene purification by guest-induced restructuring of pillar[6]arene. J. Am. Chem. Soc. 139, 2908–2911 (2017).
Mukherjee, S. et al. Exploiting framework flexibility of a metal–organic framework for selective adsorption of styrene over ethylbenzene. Inorg. Chem. 54, 4403–4408 (2015).
Krishna, R. Methodologies for evaluation of metal–organic frameworks in separation applications. RSC Adv. 5, 52269–52295 (2015).
Liu, S.-Y. et al. Porous Cu(i) triazolate framework and derived hybrid membrane with exceptionally high sensing efficiency for gaseous oxygen. Adv. Funct. Mater. 24, 5866–5872 (2014).
Grzywa, M. et al. CFA-2 and CFA-3 (coordination framework augsburg university-2 and -3); novel MOFs assembled from trinuclear Cu(i)/Ag(i) secondary building units and 3,3',5,5'-tetraphenyl-bipyrazolate ligands. Dalton Trans. 42, 6909–6921 (2013).
Wang, J. H., Li, M. & Li, D. An exceptionally stable and water-resistant metal–organic framework with hydrophobic nanospaces for extracting aromatic pollutants from water. Chem. Eur. J. 20, 12004–12008 (2014).
Zhang, J.-P., Zhang, Y.-B., Lin, J.-B. & Chen, X.-M. Metal azolate frameworks: from crystal engineering to functional materials. Chem. Rev. 112, 1001–1033 (2012).
Moghadam, P. Z. et al. Adsorption and molecular siting of CO2, water, and other gases in the superhydrophobic, flexible pores of FMOF-1 from experiment and simulation. Chem. Sci. 8, 3989–4000 (2017).
Nguyen, J. G. & Cohen, S. M. Moisture-resistant and superhydrophobic metal–organic frameworks obtained via postsynthetic modification. J. Am. Chem. Soc. 132, 4560–4561 (2010).
Rao, K. P. et al. Design of superhydrophobic porous coordination polymers through the introduction of external surface corrugation by the use of an aromatic hydrocarbon building unit. Angew. Chem. Int. Ed. 53, 8225–8230 (2014).
Mukherjee, S. et al. An ultrahydrophobic fluorous metal–organic framework derived recyclable composite as a promising platform to tackle marine oil spills. Chem. Eur. J. 22, 10937–10943 (2016).
Yu, M., Noble, R. D. & Falconer, J. L. Zeolite membranes: microstructure characterization and permeation nechanisms. Acc. Chem. Res. 44, 1196–1206 (2011).
Torres-Knoop, A., Krishna, R. & Dubbeldam, D. Separating xylene isomers by commensurate stacking of p-xylene within channels of MAF-X8. Angew. Chem. Int. Ed. 53, 7774–7778 (2014).
Lin, J.-M., He, C.-T., Liao, P.-Q., Lin, R.-B. & Zhang, J.-P. Structural, energetic, and dynamic insights into the abnormal xylene separation behavior of hierarchical porous crystal. Sci. Rep. 5, 11537 (2015).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Sun, H. COMPASS: an ab initio force-field optimized for condensed-phase applicationss overview with details on alkane and benzene compounds. J. Phys. Chem. B 102, 7338–7364 (1998).
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996).
Mayo, S. L., Olafson, B. D. & Goddard, W. A. III DREIDING: a generic force field for molecular simulations. J. Phys. Chem. 94, 8897–8909 (1990).
Addicoat, M. A., Vankova, N., Akter, I. F. & Heine, T. Extension of the universal force field to metal–organic frameworks. J. Chem. Theory Comput. 10, 880–891 (2014).
Vanduyfhuys, L. et al. QuickFF: a program for a quick and easy derivation of force fields for metal–organic frameworks from ab initio input. J. Comput. Chem. 36, 1015–1027 (2015).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Francl, M. M., Carey, C., Chirlian, L. E. & Gange, D. M. Charges fit to electrostatic potentials. II. Can atomic charges be unambiguously fit to electrostatic potentials? J. Comput. Chem. 17, 367–383 (1996).
Acknowledgements
This work was supported by the NSFC (21731007, 21821003, 91622109 and 21701191), Guangdong Pearl River Talents Program (2017BT01C161) and Guangdong Province Key Area R&D Program (2019B010940001).
Author information
Authors and Affiliations
Contributions
J.-P.Z. designed the research. D.-D.Z. performed syntheses and measurements. C.W., S.-S.W. and Z.-W.M. performed adsorption and selectivity measurements. D.-D.Z., Z.-M.Y. and R.-K.H. performed crystal structure determination. D.-D.Z., P.C., Y.D., H.Y. and C.-T.H. performed computational simulations and analyses. N.-Y.H. performed GC-MS analyses. D.-D.Z. and J.-P.Z. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, Supplementary Figs. 1–37, Supplementary Tables 1–7, Supplementary references 1,2
Supplementary Data
Crystallographic information file for the MAF-41 structure (1·pX), and the MAF-41 structure after removal of the para-xylene guest (1′)
Rights and permissions
About this article
Cite this article
Zhou, DD., Chen, P., Wang, C. et al. Intermediate-sized molecular sieving of styrene from larger and smaller analogues. Nat. Mater. 18, 994–998 (2019). https://doi.org/10.1038/s41563-019-0427-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0427-z
This article is cited by
-
Dynamic molecular pockets on one-dimensional channels for splitting ethylene from C2–C4 alkynes
Nature Chemical Engineering (2024)
-
Soft corrugated channel with synergistic exclusive discrimination gating for CO2 recognition in gas mixture
Nature Communications (2023)
-
Adsorption of gas-styrene on activated carbon from agro-waste of Silybum marianum L. as a sustainable precursor
Biomass Conversion and Biorefinery (2023)
-
Th-MOF showing six-fold imide-sealed pockets for middle-size-separation of propane from natural gas
Nano Research (2023)
-
Fluorescent porous organic polymers for detection and adsorption of nitroaromatic compounds
Scientific Reports (2022)