Abstract
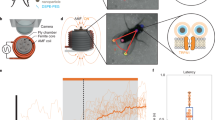
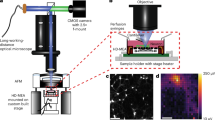
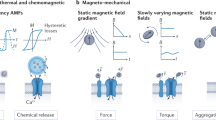
Among physical stimulation modalities, magnetism has clear advantages, such as deep penetration and untethered interventions in biological subjects. However, some of the working principles and effectiveness of existing magnetic neurostimulation approaches have been challenged, leaving questions to be answered. Here we introduce m-Torquer, a magnetic toolkit that mimics magnetoreception in nature. It comprises a nanoscale magnetic torque actuator and a circular magnet array, which deliver piconewton-scale forces to cells over a working range of ~70 cm. With m-Torquer, stimulation of neurons expressing bona fide mechanosensitive ion channel Piezo1 enables consistent and reproducible neuromodulation in freely moving mice. With its long working distance and cellular targeting capability, m-Torquer provides versatility in its use, which can range from single cells to in vivo systems, with the potential application in large animals such as primates.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The statistical data are provided with the paper as source data. Additional data that support the findings of this study are available from the corresponding authors on reasonable request.
References
Fenno, L., Yizhar, O. & Deisseroth, K. The development and application of optogenetics. Annu. Rev. Neurosci. 34, 389–412 (2011).
Hong, G. et al. A method for single-neuron chronic recording from the retina in awake mice. Science 360, 1447–1451 (2018).
Munshi, R. et al. Magnetothermal genetic deep brain stimulation of motor behaviors in awake, freely moving mice. eLife 6, e27069 (2017).
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Beurrier, C., Bioulac, B., Audin, J. & Hammond, C. High-frequency stimulation produces a transient blockade of voltage-gated currents in subthalamic neurons. J. Neurophysiol. 85, 1351–1356 (2001).
Huang, H., Delikanli, S., Zeng, H., Ferkey, D. M. & Pralle, A. Remote control of ion channels and neurons through magnetic-field heating of nanoparticles. Nat. Nanotechnol. 5, 602–606 (2010).
Chen, R., Romero, G., Christiansen, M. G., Mohr, A. & Anikeeva, P. Wireless magnetothermal deep brain stimulation. Science 347, 1477–1480 (2015).
Delmas, P., Hao, J. & Rodat-Despoix, L. Molecular mechanisms of mechanotransduction in mammalian sensory neurons. Nat. Rev. Neurosci. 12, 139–153 (2011).
Huse, M. Mechanical forces in the immune system. Nat. Rev. Immunol. 17, 679–690 (2017).
Li, T. L. et al. Engineering a genetically encoded magnetic protein crystal. Nano Lett. 19, 6955–6963 (2019).
Gregurec, D. et al. Magnetic vortex nanodiscs enable remote magnetomechanical neural stimulation. ACS Nano 14, 8036–8045 (2020).
Christiansen, M. G., Senko, A. W. & Anikeeva, P. Magnetic strategies for nervous system control. Annu. Rev. Neurosci. 42, 271–293 (2019).
Wheeler, M. A. et al. Genetically targeted magnetic control of the nervous system. Nat. Neurosci. 19, 756–761 (2016).
Meister, M. Physical limits to magnetogenetics. eLife 5, e17210 (2016).
Xu, F. X. et al. Magneto is ineffective in controlling electrical properties of cerebellar Purkinje cells. Nat. Neurosci. https://doi.org/10.1038/s41593-019-0475-3 (2019).
Cullity, B. D. & Graham, C. D. Introduction to Magnetic Materials 2nd edn (IEEE/Wiley, 2009).
Strick, T. R., Allemand, J. F., Bensimon, D., Bensimon, A. & Croquette, V. The elasticity of a single supercoiled DNA molecule. Science 271, 1835–1837 (1996).
Lipfert, J., Kerssemakers, J. W., Jager, T. & Dekker, N. H. Magnetic torque tweezers: measuring torsional stiffness in DNA and RecA-DNA filaments. Nat. Methods 7, 977–980 (2010).
Wang, N., Butler, J. P. & Ingber, D. E. Mechanotransduction across the cell surface and through the cytoskeleton. Science 260, 1124–1127 (1993).
Lipfert, J., van Oene, M. M., Lee, M., Pedaci, F. & Dekker, N. H. Torque spectroscopy for the study of rotary motion in biological systems. Chem. Rev. 115, 1449–1474 (2015).
Lamb, H. Hydrodynamics 6th edn (Dover Publications, 1945).
Romano, G., Sacconi, L., Capitanio, M. & Pavone, F. S. Force and torque measurements using magnetic micro beads for single molecule biophysics. Opt. Commun. 215, 323–331 (2003).
Morimatsu, M., Mekhdjian, A. H., Adhikari, A. S. & Dunn, A. R. Molecular tension sensors report forces generated by single integrin molecules in living cells. Nano Lett. 13, 3985–3989 (2013).
Seo, D. et al. A mechanogenetic toolkit for interrogating cell signaling in space and time. Cell 165, 1507–1518 (2016).
Yao, M. et al. Mechanical activation of vinculin binding to talin locks talin in an unfolded conformation. Sci. Rep. 4, 4610 (2014).
Svoboda, K. & Block, S. M. Force and velocity measured for single kinesin molecules. Cell 77, 773–784 (1994).
Corey, D. P. & Howard, J. Models for ion channel gating with compliant states. Biophys. J. 66, 1254–1257 (1994).
Kim, D. H. et al. Biofunctionalized magnetic-vortex microdiscs for targeted cancer-cell destruction. Nat. Mater. 9, 165–171 (2010).
Shen, Y. et al. Elongated nanoparticle aggregates in cancer cells for mechanical destruction with low frequency rotating magnetic field. Theranostics 7, 1735–1748 (2017).
Kole, K. et al. Assessing the utility of Magneto to control neuronal excitability in the somatosensory cortex. Nat. Neurosci. https://doi.org/10.1038/s41593-019-0474-4 (2019).
Wang, G. et al. Revaluation of magnetic properties of Magneto. Nat. Neurosci. https://doi.org/10.1038/s41593-019-0473-5 (2019).
Coste, B. et al. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 330, 55–60 (2010).
Ranade, S. S., Syeda, R. & Patapoutian, A. Mechanically activated ion channels. Neuron 87, 1162–1179 (2015).
Coste, B. et al. Piezo1 ion channel pore properties are dictated by C-terminal region. Nat. Commun. 6, 7223 (2015).
Adamantidis, A. R., Zhang, F., Aravanis, A. M., Deisseroth, K. & de Lecea, L. Neural substrates of awakening probed with optogenetic control of hypocretin neurons. Nature 450, 420–424 (2007).
Sheng, M. & Greenberg, M. E. The regulation and function of c-fos and other immediate early genes in the nervous system. Neuron 4, 477–485 (1990).
Covington, H. E. R. et al. Antidepressant effect of optogenetic stimulation of the medial prefrontal cortex. J. Neurosci. 30, 16082–16090 (2010).
Howard, J. & Hudspeth, A. J. Compliance of the hair bundle associated with gating of mechanoelectrical transduction channels in the bullfrog’s saccular hair cell. Neuron 1, 189–199 (1988).
Sen, S., Subramanian, S. & Discher, D. E. Indentation and adhesive probing of a cell membrane with AFM: theoretical model and experiments. Biophys. J. 89, 3203–3213 (2005).
Airan, R. D., Thompson, K. R., Fenno, L. E., Bernstein, H. & Deisseroth, K. Temporally precise in vivo control of intracellular signalling. Nature 458, 1025–1029 (2009).
Gradinaru, V. et al. Targeting and readout strategies for fast optical neural control in vitro and in vivo. J. Neurosci. 27, 14231–14238 (2007).
Ranade, S. S. et al. Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature 516, 121–125 (2014).
Mammoto, T. & Ingber, D. E. Mechanical control of tissue and organ development. Development 137, 1407–1420 (2010).
Jeanes, A., Gottardi, C. J. & Yap, A. S. Cadherins and cancer: how does cadherin dysfunction promote tumor progression? Oncogene 27, 6920–6929 (2008).
Jang, J. T. et al. Critical enhancements of MRI contrast and hyperthermic effects by dopant-controlled magnetic nanoparticles. Angew. Chem. Int. Ed. 48, 1234–1238 (2009).
Jewett, J. C. & Bertozzi, C. R. Cu-free click cycloaddition reactions in chemical biology. Chem. Soc. Rev. 39, 1272–1279 (2010).
Kim, J. W. et al. Single-cell mechanogenetics using monovalent magnetoplasmonic nanoparticles. Nat. Protoc. 12, 1871–1889 (2017).
Mittereder, N., March, K. L. & Trapnell, B. C. Evaluation of the concentration and bioactivity of adenovirus vectors for gene therapy. J. Virol. 70, 7498–7509 (1996).
Shin, W. et al. Identification of Ras-degrading small molecules that inhibit the transformation of colorectal cancer cells independent of beta-catenin signaling. Exp. Mol. Med. 50, 71 (2018).
Dong, H. W. Allen Reference Atlas: A Digital Color Brain Atlas of the C57Black/6J Male Mouse (Wiley, 2008).
Acknowledgements
Myc897–Piezo1 (Myc tag at the 897 N-terminal residue of Piezo1) in pcDNA3.1 was kindly provided by A. Patapoutian (The Scripps Research Institute, La Jolla, CA, USA). We thank E. Chung, J. Kim, J.-w. Kim, and C. Mikuni for initial help and discussions on this research. This work was supported by the Institute for Basic Science (IBS-R026-D1).
Author information
Authors and Affiliations
Contributions
J.-u.L. performed the overall experiments. Y.L. provided magnetic nanoparticles. J.K. and H.K. performed magnetic field simulation. W.S. and W.R.K. performed in vitro and in vivo experiments. J.-H.L. and J.C. wrote the manuscript. J.C. conceived and supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Descriptions of Supplementary Videos 1–4, Supplementary Figs. 1–22, Table 1, and Notes 1 and 2.
Supplementary Video 1
Representative fluorescence video of suspended m-Torquer (in 98% glycerol) and bound m-Torquer on cell membrane in rotating magnetic field.
Supplementary Video 2
In situ calcium influx induced by m-Torquer in Piezo1-expressing neuron.
Supplementary Video 3
In situ calcium influx of repetitive stimulation of Piezo1.
Supplementary Video 4
Mouse behaviour control experiment with freely moving mouse.
Source data
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Rights and permissions
About this article
Cite this article
Lee, Ju., Shin, W., Lim, Y. et al. Non-contact long-range magnetic stimulation of mechanosensitive ion channels in freely moving animals. Nat. Mater. 20, 1029–1036 (2021). https://doi.org/10.1038/s41563-020-00896-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-020-00896-y
This article is cited by
-
Self-rectifying magnetoelectric metamaterials for remote neural stimulation and motor function restoration
Nature Materials (2024)
-
Spinning nanomotor with a DNA clutch
Nature Nanotechnology (2024)
-
A comprehensive approach to characterize navigation instruments for magnetic guidance in biological systems
Scientific Reports (2024)
-
A magnetically powered nanomachine with a DNA clutch
Nature Nanotechnology (2024)
-
Janus microparticles-based targeted and spatially-controlled piezoelectric neural stimulation via low-intensity focused ultrasound
Nature Communications (2024)