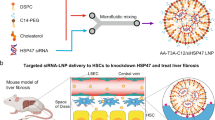
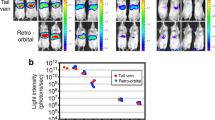
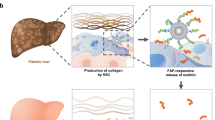
Abstract
Relaxin is an antifibrotic peptide hormone previously assumed to directly reverse the activation of hepatic stellate cells for liver fibrosis resolution. Using nanoparticle-mediated delivery, here we show that, although relaxin gene therapy reduces liver fibrosis in vivo, in vitro treatment fails to induce quiescence of the activated hepatic stellate cells. We show that hepatic macrophages express the primary relaxin receptor, and that, on relaxin binding, they switch from the profibrogenic to the pro-resolution phenotype. The latter releases exosomes that promote the relaxin-mediated quiescence of activated hepatic stellate cells through miR-30a-5p. Building on these results, we developed lipid nanoparticles that preferentially target activated hepatic stellate cells in the fibrotic liver and encapsulate the relaxin gene and miR-30a-5p mimic. The combinatorial gene therapy achieves synergistic antifibrosis effects in models of mouse liver fibrosis. Collectively, our findings highlight the key role that macrophages play in the relaxin-primed alleviation of liver fibrosis and demonstrate a proof-of-concept approach to devise antifibrotic strategies through the complementary application of nanotechnology and basic science.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The MicroRNA Data Integration Portal was used to identify gene targets for exosomal miRNAs and can be accessed with http://ophid.utoronto.ca/mirDIP28. All data supporting the findings of this study are available within the article and its Supplementary Information. Source data are provided with this paper.
References
Troeger, J. S. et al. Deactivation of hepatic stellate cells during liver fibrosis resolution in mice. Gastroenterology 143, 1073–1083. e1022 (2012).
Hu, M. et al. Relaxin gene delivery mitigates liver metastasis and synergizes with check point therapy. Nat. Commun. 10, 2993 (2019).
Fallowfield, J. A. et al. Relaxin modulates human and rat hepatic myofibroblast function and ameliorates portal hypertension in vivo. Hepatology 59, 1492–1504 (2014).
Tacke, F. & Zimmermann, H. W. Macrophage heterogeneity in liver injury and fibrosis. J. Hepatol. 60, 1090–1096 (2014).
Ramachandran, P. et al. Differential Ly-6C expression identifies the recruited macrophage phenotype, which orchestrates the regression of murine liver fibrosis. Proc. Natl Acad. Sci. USA 109, E3186–E3195 (2012).
Pellicoro, A., Ramachandran, P., Iredale, J. P. & Fallowfield, J. A. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat. Rev. Immunol. 14, 181–194 (2014).
Francis, H. et al. Regulation of the extrinsic apoptotic pathway by microRNA-21 in alcoholic liver injury. J. Biol. Chem. 289, 27526–27539 (2014).
Chen, S. A. et al. The pharmacokinetics of recombinant human relaxin in nonpregnant women after intravenous, intravaginal, and intracervical administration. Pharma. Res 10, 834–838 (1993).
Uhlén, M. et al. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Friedman, S. L. Hepatic stellate cells: protean, multifunctional, and enigmatic cells of the liver. Physiol. Rev. 88, 125–172 (2008).
Hu, M. & Huang, L. Nanomaterial manipulation of immune microenvironment in the diseased liver. Adv. Funct. Mater. 29, 1805760 (2019).
Kushiyama, T. et al. Alteration in the phenotype of macrophages in the repair of renal interstitial fibrosis in mice. Nephrology 16, 522–535 (2011).
Ma, P. F. et al. Cytotherapy with M1-polarized macrophages ameliorates liver fibrosis by modulating immune microenvironment in mice. J. Hepatol. 67, 770–779 (2017).
Kern, K. et al. CD200 selectively upregulates prostaglandin E2 and D2 synthesis in LPS-treated bone marrow-derived macrophages. Prostag. Oth. Lipid Mediat. 133, 53–59 (2017).
Wang, M. et al. Chronic alcohol ingestion modulates hepatic macrophage populations and functions in mice. J. Leukoc. Biol. 96, 657–665 (2014).
Liaskou, E. et al. Monocyte subsets in human liver disease show distinct phenotypic and functional characteristics. Hepatology 57, 385–398 (2013).
Zimmermann, H. W. et al. Functional contribution of elevated circulating and hepatic non-classical CD14 + CD16 + monocytes to inflammation and human liver fibrosis. PloS ONE 5, e11049 (2010).
Krenkel, O. & Tacke, F. Liver macrophages in tissue homeostasis and disease. Nat. Rev. Immunol. 17, 306–321 (2017).
Hanna, R. N. et al. The transcription factor NR4A1 (Nur77) controls bone marrow differentiation and the survival of Ly6C− monocytes. Nat. Immunol. 12, 778–785 (2011).
Mildner, A. et al. Genomic characterization of murine monocytes reveals C/EBPβ transcription factor dependence of Ly6C− cells. Immunity 46, 849–862. e847 (2017).
Palumbo-Zerr, K. et al. Orphan nuclear receptor NR4A1 regulates transforming growth factor-β signaling and fibrosis. Nat. Med. 21, 150–158 (2015).
Hsu, S. Y. et al. Activation of orphan receptors by the hormone relaxin. Science 295, 671–674 (2002).
Maruoka, H. et al. Dibutyryl-cAMP up-regulates Nur77 expression via histone modification during neurite outgrowth in PC12 cells. J. Biochem. 148, 93–101 (2010).
Lough, J., Rosenthall, L., Arzoumanian, A. & Goresky, C. A. Kupffer cell depletion associated with capillarization of liver sinusoids in carbon tetrachloride-induced rat liver cirrhosis. J. Hepatol. 5, 190–198 (1987).
Simons, M. & Raposo, G. Exosomes—vesicular carriers for intercellular communication. Curr. Opin. Cell Biol. 21, 575–581 (2009).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9, 654–659 (2007).
Kim, K. M., Abdelmohsen, K., Mustapic, M., Kapogiannis, D. & Gorospe, M. RNA in extracellular vesicles. WIREs RNA 8, e1413 (2017).
Tokar, T. et al. mirDIP 4.1—integrative database of human microRNA target predictions. Nucleic Acids Res. 46, D360–D370 (2017).
Zhang, P. et al. The deubiquitinating enzyme TNFAIP3 mediates inactivation of hepatic ASK1 and ameliorates nonalcoholic steatohepatitis. Nat. Med. 24, 84–94 (2018).
Liles, J. T. et al. ASK1 contributes to fibrosis and dysfunction in models of kidney disease. J. Clin. Investig. 128, 4485–4500 (2018).
Singh, S., Simpson, R. L. & Bennett, R. G. Relaxin activates peroxisome proliferator-activated receptor γ (PPARγ) through a pathway involving PPARγ coactivator 1α (PGC1α). J. Biol. Chem. 290, 950–959 (2015).
Singh, S. & Bennett, R. G. Relaxin signaling activates peroxisome proliferator-activated receptor gamma. Mol. Cell. Endocrinol. 315, 239–245 (2010).
Hazra, S., Miyahara, T., Rippe, R. A. & Tsukamoto, H. PPAR gamma and hepatic stellate cells. Comp. Hepatol. 3, S7 (2004).
Wei, J. et al. PPARγ downregulation by TGF-β in fibroblast and impaired expression and function in systemic sclerosis: a novel mechanism for progressive fibrogenesis. PloS ONE 5, e13778 (2010).
Chibon, F. et al. ASK1 (MAP3K5) as a potential therapeutic target in malignant fibrous histiocytomas with 12q14–q15 and 6q23 amplifications. Genes Chromosomes Cancer 40, 32–37 (2004).
Tang, X. et al. An RNA interference-based screen identifies MAP4K4/NIK as a negative regulator of PPARγ, adipogenesis, and insulin-responsive hexose transport. Proc. Natl Acad. Sci. USA 103, 2087–2092 (2006).
Xiong, J. et al. hnRNPU/TrkB defines a chromatin accessibility checkpoint for liver injury and nonalcoholic steatohepatitis pathogenesis. Hepatology 71, 1228–1246 (2020).
Matsumoto, M. et al. An improved mouse model that rapidly develops fibrosis in non‐alcoholic steatohepatitis. Int. J. Exp. Pathol. 94, 93–103 (2013).
Bani, D., Bigazzi, M., Masini, E., Bani, G. & Sacchi, T. B. Relaxin depresses platelet aggregation: in vitro studies on isolated human and rabbit platelets. Lab. Invest. 73, 709–716 (1995).
Erikson, M. S. & Unemori, E. N. in Relaxin 2000 (eds Tregear G.W., Ivell R., Bathgate R.A. & Wade J.D.) 373–381 (Springer, 2001).
Amitrano, L., Guardascione, M. A., Brancaccio, V. & Balzano, A. Coagulation disorders in liver disease. Semin. Liver Dis. 22, 83–96 (2002).
Rios, R., Sangro, B., Herrero, I., Quiroga, J. & Prieto, J. The role of thrombopoietin in the thrombocytopenia of patients with liver cirrhosis. Am. J. Gastroenterol. 100, 1311 (2005).
Berres, M. L. et al. Antagonism of the chemokine Ccl5 ameliorates experimental liver fibrosis in mice. J. Clin. Investig. 120, 4129–4140 (2010).
Mitchell, C. et al. Dual role of CCR2 in the constitution and the resolution of liver fibrosis in mice. Am. J. Pathol. 174, 1766–1775 (2009).
Lefebvre, E. et al. Antifibrotic effects of the dual CCR2/CCR5 antagonist cenicriviroc in animal models of liver and kidney fibrosis. PloS ONE 11, e0158156 (2016).
Banerjee, R., Tyagi, P., Li, S. & Huang, L. Anisamide-targeted stealth liposomes: a potent carrier for targeting doxorubicin to human prostate cancer cells. Int. J. Cancer 112, 693–700 (2004).
Constandinou, C., Henderson, N. & Iredale, J. P. in Fibrosis Research: Methods and Protocols (eds Varga, J., David A. Brenner, D. A. & Phan, S. H.) 237–250 (Springer, 2005).
Wang, Y. et al. Nanoparticle-mediated HMGA1 silencing promotes lymphocyte infiltration and boosts checkpoint blockade immunotherapy for cancer. Adv. Funct. Mater. 28, 1802847 (2018).
Teerlink, J. R. et al. Serelaxin, recombinant human relaxin-2, for treatment of acute heart failure (RELAX-AHF): a randomised, placebo-controlled trial. Lancet 381, 29–39 (2013).
Ying, W., Cheruku, P. S., Bazer, F. W., Safe, S. H. & Zhou, B. Investigation of macrophage polarization using bone marrow derived macrophages. J. Vis. Exp. 76, e50323 (2013).
Acknowledgements
We thank S. Li for helpful discussions regarding the flow cytometry analysis on hepatic macrophages. The work in LH.’s lab was supported by NIH grants DK100664 and CA198999, and by a grant from Eshelman Institute for Innovation. Human sample collection and procession was done by J.T.’s lab and supported by Huamei Research Foundation (Grant 2017HMKY07), the Medicine and Health Sciences Research Foundation of Zhejiang Province (Grant 2019KY177).
Author information
Authors and Affiliations
Contributions
M.H. and L.H. conceived and designed the research. M.H., Y.W., Z.L., Z.Y., K.G., M.L. and M.W. performed the in vivo mouse experiments. J.T. provided paraffin-embedded liver tissues from patients. M.H. and Y.W. prepared the frozen sections, immunofluorescence and histological staining. M.H. and Y.W. analysed and quantified the microscopic images. M.H. and Y.W. purified the liver macrophage and mononuclear cells, did the flow cytometry assay and analysed the results. M.H. and Y.W. performed the western blot and qPCR assays on the cell and tissue samples. M.H. and Y.W. did the hydroxyproline assay on liver tissues. M.H. isolated and cultured the primary bone marrow-derived macrophages. M.H. designed the in vitro experiments to analyse the interaction between the macrophages and aHSCs. M.H., Y.W. and Z.L. isolated exosomes from Raw264.7 cells, hBMDMs, and differentiated THP-1 cells. M.H. performed miRNA array analyses. M.H. and Y.W. prepared lipid nanoparticles and characterized the exosomes and lipid nanoparticles. M.H., Y.W., K.G. and M.W. analysed the data. M.H. and L.H. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks Frank Tacke and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Figs. 1–29 and Tables 1–3.
Supplementary video 1
Bleeding time recording for PBS treatment group.
Supplementary video 2
Bleeding time recording for free recombinant RLN treatment group.
Supplementary video 3
Bleeding time recording for pRLN LPD treatment group.
Source data
Source Data Fig. 1
Unprocessed Western Blots.
Source Data Fig. 2
Unprocessed Western Blots.
Source Data Fig. 3
Unprocessed Western Blots.
Source Data Fig. 4
Unprocessed Western Blots.
Source Data Fig. 5
Unprocessed Western Blots.
Source Data Fig. 6
Unprocessed Western Blots.
Source Data Fig. 7
Unprocessed Western Blots.
Rights and permissions
About this article
Cite this article
Hu, M., Wang, Y., Liu, Z. et al. Hepatic macrophages act as a central hub for relaxin-mediated alleviation of liver fibrosis. Nat. Nanotechnol. 16, 466–477 (2021). https://doi.org/10.1038/s41565-020-00836-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-020-00836-6
This article is cited by
-
Exosomal mediators in sepsis and inflammatory organ injury: unraveling the role of exosomes in intercellular crosstalk and organ dysfunction
Military Medical Research (2024)
-
Identifying a distinct fibrosis subset of NAFLD via molecular profiling and the involvement of profibrotic macrophages
Journal of Translational Medicine (2023)
-
Immunology of human fibrosis
Nature Immunology (2023)
-
Ligand-tethered lipid nanoparticles for targeted RNA delivery to treat liver fibrosis
Nature Communications (2023)
-
Advanced glycation end-products as mediators of the aberrant crosslinking of extracellular matrix in scarred liver tissue
Nature Biomedical Engineering (2023)