Abstract
Despite its position as the first-line drug for treatment of type 2 diabetes mellitus, the mechanisms underlying the plasma glucose level-lowering effects of metformin (1,1-dimethylbiguanide) still remain incompletely understood. Metformin is thought to exert its primary antidiabetic action through the suppression of hepatic glucose production. In addition, the discovery that metformin inhibits the mitochondrial respiratory chain complex 1 has placed energy metabolism and activation of AMP-activated protein kinase (AMPK) at the centre of its proposed mechanism of action. However, the role of AMPK has been challenged and might only account for indirect changes in hepatic insulin sensitivity. Various mechanisms involving alterations in cellular energy charge, AMP-mediated inhibition of adenylate cyclase or fructose-1,6-bisphosphatase 1 and modulation of the cellular redox state through direct inhibition of mitochondrial glycerol-3-phosphate dehydrogenase have been proposed for the acute inhibition of gluconeogenesis by metformin. Emerging evidence suggests that metformin could improve obesity-induced meta-inflammation via direct and indirect effects on tissue-resident immune cells in metabolic organs (that is, adipose tissue, the gastrointestinal tract and the liver). Furthermore, the gastrointestinal tract also has a major role in metformin action through modulation of glucose-lowering hormone glucagon-like peptide 1 and the intestinal bile acid pool and alterations in gut microbiota composition.
Key points
-
Metformin is the first-line drug for treatment of type 2 diabetes mellitus, with an excellent safety profile, high efficacy in glycaemic control and clear but incompletely understood cardioprotective benefits.
-
The pleiotropic properties of metformin suggest that the drug acts on multiple tissues through various underlying mechanisms rather than on a single organ via a unifying mode of action.
-
Mitochondrial respiratory chain complex 1 is targeted by metformin and its inhibition is involved in AMP-activated protein kinase-independent regulation of hepatic gluconeogenesis by triggering alterations in cellular energy charge and redox state.
-
Metformin might contribute to improvements in obesity-associated meta-inflammation and tissue-specific insulin sensitivity through direct and indirect effects on various resident immune cells in metabolic organs.
-
The gastrointestinal tract has an important role in the action of metformin, which modulates bile acid recirculation and enhances the secretion of the glucose-lowering gut incretin hormone glucagon-like peptide 1.
-
The gut microbiota is a novel target in the mechanisms of metformin action and is involved in both the therapeutic and adverse effects of the drug.
Similar content being viewed by others
Introduction
Metformin is the first-line drug for the treatment of patients with type 2 diabetes mellitus (T2DM) as recommended by clinical guidelines published by the American Diabetes Association and the European Association for the Study of Diabetes1,2, which are supported by prospective studies3,4 and meta-analyses published in 2016 (ref.5,6). These guidelines are based on the improved glycaemic profile and reduction in cardiovascular mortality induced by metformin treatment, without the risk of hypoglycaemia and/or body weight gains that are associated with the use of other antidiabetic drugs3,4,6,7. Furthermore, metformin is also the favoured antidiabetic drug because of its good safety profile and minimal cost8.
The drug metformin is a biguanide derivative (1,1-dimethylbiguanide hydrochloride) that originates from the plant goat’s rue or French lilac (Galega officinalis)9. Originally used in medieval Europe to relieve the symptoms of diabetes mellitus, this plant contains galegine, which is an isoprenyl guanidine. Although monoguanidine and diguanidine derivatives are toxic, the biguanides (two N-linked molecules of guanidine) synthesized from the reaction between dimethylamine hydrochloride and dicyanodiamide10 have been used for the treatment of diabetes mellitus since the late 1950s9. Of note, metformin has been approved for use in Europe and Canada since 1957, although it was only introduced in the USA in 1995. The more potent biguanides, phenformin and buformin, were quite popular in the USA and Europe in the 1960s but were removed from the market in most countries in the late 1970s owing to an increased risk of lactic acidosis11. By contrast, the incidence of lactic acidosis associated with metformin treatment is very low (approximately 3–10 cases per 100,000 person-years and ~1.5 deaths per 100,000 patient-years), except in high-risk groups, particularly patients with chronically impaired renal function or acute kidney disease8. The most common adverse effect of metformin is gastrointestinal intolerance (Box 1).
Metformin is described as an anti-hyperglycaemic agent that does not cause clinical hypoglycaemia in patients with T2DM or alter glucose homeostasis in non-diabetic individuals5,6,7. Although metformin has been available for over 60 years for the treatment of T2DM, the exact mechanism (or mechanisms) of its effects on blood levels of glucose still remain elusive12. However, several actions have been attributed to its ability to lower blood levels of glucose. For example, metformin acts primarily by the suppression of enhanced basal endogenous glucose production in individuals with T2DM through a 25–40% decrease in the hepatic gluconeogenesis rate as determined by 13C NMR spectroscopy or tracer infusion13,14. Moreover, some euglycaemic–hyperinsulinaemic clamp studies have shown a beneficial effect of metformin on insulin sensitivity in skeletal muscle15,16, although not all studies17. Metformin treatment is accompanied by insulin-stimulated systemic glucose disposal, which occurs predominantly in skeletal muscle16,18. In addition, the drug increases peripheral glucose utilization by the intestine, primarily via non-oxidative metabolism19. Emphasis is now given to the importance of metformin actions within the gut20,21, possibly via alteration of the intestinal microbiota22. The notion that biguanides decrease mitochondrial oxygen consumption has offered a plausible molecular mechanism of action for direct suppression of hepatic gluconeogenesis, which occurs via decreases in cellular energy charge and subsequent activation of AMP-activated protein kinase, a cellular energy sensor23. However, the contribution of the aforementioned mechanism to metformin's therapeutic effects is a subject of intense debate12,24. In addition, the literature is confounded with a plethora of putative mechanisms of action for metformin, which have been elucidated from the use of supra-therapeutic concentrations of metformin in in vitro and preclinical studies, as well as the differences reported between acute and chronic metformin administration in human and animal models.
In this Review, we discuss our current understanding of the molecular mechanisms that underlie the pleiotropic actions of metformin on metabolism and inflammation. In addition, we focus on recent research developments on the actions of metformin in the liver and intestine to regulate blood levels of glucose in patients with T2DM.
Pharmacokinetics and pharmacogenetics
Metformin is administered orally and has a low bioavailability (40–60%)25. The drug is not metabolized and is excreted unchanged in the urine. After a single oral dose of 0.5 g, metformin is distributed systemically within 6 h following absorption, which predominantly occurs in the upper small intestine, with negligible absorption in the large intestine25. The peak plasma metformin concentration occurs 3 h after administration and leads to a concentration of ∼3 μg/ml (∼18 μM)26. Notably, analysis of tissue-specific biodistribution of the drug in mice using 14C-labelled metformin or dynamic positron emission tomography (PET) with 11C-metformin revealed its substantial accumulation in the gastrointestinal tract, liver and kidney27,28. Interestingly, these results have been partially replicated in humans, showing 11C-metformin biodistribution mainly in the liver and rapid excretion through the kidneys29.
Metformin is a strongly hydrophilic compound with an acid dissociation constant (pKa) of 11.5; at physiological pH the drug exists as a monoprotonated cation23. Given these chemical properties, transport of the drug across biological membranes involves uptake via specific transporters. This process is in line with the inverse relationship observed in humans between the dose ingested and the relative absorption rate25. The absorption, distribution and excretion of metformin depend mainly on the organic cation transporters (OCTs), multidrug and toxin extruders (MATEs) and plasma membrane monoamine transporter (PMAT). For example, metformin is taken up from the intestinal lumen by PMAT (encoded by SLC29A4) and OCT3 (encoded by SLC22A3) that are localized at the apical membrane of enterocytes. Subsequently, the drug is transported into the bloodstream by OCT1 (encoded by SLC22A1), which is localized at the basolateral membrane of enterocytes30. The hepatic uptake of metformin depends on OCT1 and possibly OCT3, whereas MATE1 (encoded by SLC47A1) contributes to its hepatic elimination30. In the kidney, uptake of metformin into renal epithelial cells is primarily facilitated by OCT2 (encoded by SLC22A2), whereas MATE1 and MATE2 (encoded by SLC47A2) contribute to the renal excretion of the drug into the urine30. Importantly, the role of these transporters in the pharmacokinetics of metformin has been confirmed by 11C-metformin functional PET imaging in mice deficient in these transporters or treated with specific inhibitors28.
Some human genetic studies have associated genetic polymorphisms in the aforementioned transporter genes with modifications of metformin pharmacokinetics30. However, studies of the effect of these variants altering metformin transport on clinical responses in patients with T2DM have yielded contradictory results, and uncertainty remains31. Although all these transporters clearly play a role in metformin pharmacokinetics, they do not appear to be critical to the pharmacodynamics of the drug in patients with diabetes mellitus. A 2017 large-scale meta-analysis found that polymorphisms in genes encoding transporters involved in metformin pharmacokinetics (SLC22A1, SLC22A2, SLC22A4, SLC47A1 and SLC47A2) have no statistically significant effect on the glycaemic response to the drug32, indicating possible transporter redundancy or the contribution of mechanisms of action that are independent of the systemic exposure to the drug. However, studies of pharmacodynamic genetics have revealed that variation in the glucose transporter 2 (GLUT2) gene (SLC2A2) is associated with a greater glycaemic response to metformin33. Moreover, this effect could be linked to the restoration of glucose sensitivity by the drug34. Other genetic candidates for metformin response are the genes encoding the subunits of AMPK (PRKAA1, PRKAA2 and PRKAB2) and the AMPK upstream kinase LKB1 (STK11)35. However, additional clinical and mechanistic studies are needed to clearly delineate influences of these genes on the clinical response to metformin.
Targeting mitochondria
The notion that metformin could exert some of its action through direct interaction with the mitochondria dates back to as early as the 1950s and 1960s, when various biguanides were shown to inhibit mitochondrial oxidative phosphorylation (OXPHOS)36,37. However, the hypothesis that mitochondria are involved in the cellular mechanism of action of metformin was conceptually strengthened in the 2000s, when two independent groups reported for the first time that metformin inhibits the mitochondrial respiratory chain complex 1 (the first and largest enzyme complex of the electron transfer chain)38,39. This effect is highly specific but also weak and reversible38,39, contrasting with the characteristics of canonical mitochondrial respiratory chain complex 1 inhibitors, such as rotenone and piericidin A40, which are both potent uncharged and highly hydrophobic molecules with a very low half-maximal inhibitory concentration (IC50) (~2 µM). The inhibition of mitochondrial respiratory chain complex 1 by metformin leads to a mild decrease in ATP generation by mitochondrial OXPHOS and a concomitant moderate increase in the AMP:ATP ratio in hepatocytes38,39. During the past 20 years, the selective inhibition of mitochondrial respiratory chain complex 1 by metformin has been further confirmed in multiple biological models and species41,42, including human primary hepatocytes43 and various cancer cell lines44,45,46,47,48,49,50,51,52,53. Furthermore, when these aspects were studied in the aforementioned models this effect was usually associated with a substantial decrease in mitochondrial NADH oxidation, a decrease in the proton gradient across the inner mitochondrial membrane and reductions in cellular oxygen consumption rate42 (Fig. 1).
After accumulation into the mitochondria by an as-yet-unclear process, which might be associated with the positive charge of the molecule, metformin inhibits the mitochondrial respiratory respiratory chain complex 1 (C1) in a reversible and non-competitive manner. This inhibition presumably occurs by direct interaction of the drug with the Cys39-containing matrix loop of the respiratory chain subunit ND3, which stabilizes the enzyme in an open-loop deactive conformation state. The inhibition of C1 leads to a decrease in NADH oxidation, a decrease in proton pumping across the inner mitochondrial membrane and a decrease in oxygen consumption rate, resulting in lower proton gradient (Δψ) and a decrease in proton-driven ATP synthesis from ADP and inorganic phosphate (Pi). C, cytochrome c; C1–C5, mitochondrial respiratory chain complex 1–5; IMS, inner-membrane space; Q, coenzyme Q (ubiquinone).
Inhibition of mitochondrial complex 1
The exact mechanism by which metformin specifically targets the mitochondria remains unclear, and whether the drug inhibits mitochondrial respiratory chain complex 1 directly or not is still a matter of debate41,42,54. Although many studies have shown that metformin accumulates at high concentrations within mitochondria39,45, others have shown that the distribution of metformin in hepatocytes is predominantly within the cytosol55. The main reasons for this controversy are variations in the concentrations of metformin and the experimental model (for example, intact cells versus isolated organelles) used in in vitro studies. Indeed, although clinically relevant drug concentrations of metformin (<100 µM) are sufficient to achieve a dose-dependent and time-dependent in situ inhibition of mitochondrial respiratory chain complex 1 in various cell types38,46,56,57,58, these concentrations have no effect on the activity of complex 1 in isolated mitochondria. By contrast, only supra-therapeutic concentrations (that is, greater than the therapeutic dose) of the drug (20–100 mM) have been reported to directly inhibit mitochondrial respiratory chain complex 1 in both isolated mitochondria and sub-mitochondrial particles42.
The low intra-mitochondrial accumulation of metformin in intact cells could primarily be explained by slow transporter-mediated permeation of the drug across the plasma membrane. However, some studies also suggested the existence of a specific protein-mediated transport system for importing metformin into mitochondria59. In line with this theory, experiments performed in oocytes of Xenopus laevis (African clawed frog) have shown that therapeutic concentrations of metformin are able to directly inhibit mitochondrial respiratory chain complex 1 activity in isolated mitochondria, but only when delivered in liposome-encapsulated form. This finding led to the hypothesis of an endocytic vesicular process that routes the drug from the plasma membrane to the mitochondria57. However, the other hypothesis that is currently favoured by the majority of the metformin research community is based on the peculiar physicochemical properties of this biguanide. The positive charge of metformin at physiological pH was proposed to account for its slow accumulation within the matrix of energized mitochondria of intact cells, driven by mitochondrial transmembrane electrochemical potential (ΔΨ)39,45. As such, it has been proposed that metformin can accumulate in mitochondria, theoretically reaching millimolar concentrations in the matrix of the organelle despite a cytoplasmic level in the micromolar range39,41. This ΔΨ-driven mitochondrial import theory might also provide an explanation for the weak inhibitory effect of metformin on mitochondrial respiratory chain complex 1: as metformin is imported into mitochondria, its progressive accumulation causes a decrease in mitochondrial membrane potential, which limits its own accumulation.
Of note, metformin is known for its metal-binding properties, particularly its high affinity for copper. The interaction of metformin with mitochondrial copper ions has been suggested to be involved in the inhibition of mitochondrial respiratory chain complex 1 by the drug through a mechanism that remains to be fully characterized60.
Metformin–mitochondrial respiratory chain complex 1 interactions
The mammalian mitochondrial respiratory chain complex 1 is a large L-shaped membrane-bound redox enzyme (also known as NADH:ubiquinone oxidoreductase) that consists of 14 core subunits and more than 30 accessory subunits that are encoded by both nuclear and mitochondrial genes61,62. The complex couples the transfer of electrons from NADH to the ubiquinone pool with a transfer of four protons from the mitochondrial matrix towards the intermembrane space61. Of note, resolution of the crystal structure of mitochondrial respiratory chain complex 1 showed that it is organized into four functional modules (N, Q, proximal pump (PP) and distal pump (PD)) and exists in two distinct forms: a fully active one and a catalytically inactive so-called ‘deactive’ D-form61,62. Although research has shown that metformin does not alter the structural integrity of the whole complex45 and probably binds to one of the phylogenetically conserved core subunits61, the exact nature of molecular interactions between the drug and mitochondrial respiratory chain complex 1 remains unknown. A substantial breakthrough came in 2014 in an elegant study in which the effect of metformin was methodically dissected at different levels of the catalytic cycle of the enzyme45. Altogether, the authors proposed that metformin is a reversible non-competitive inhibitor that binds to the Cys39-containing matrix loop of the mitochondrial respiratory chain complex 1 subunit ND3, which is located within the amphipathic region between the redox and proton-transfer domains of the PP module. This interaction stabilizes the enzyme in an open-loop deactive conformation state45 (Fig. 1).
Metformin and hepatic gluconeogenesis
The liver has a central role in the blood glucose-lowering effect of metformin in patients with T2DM. Metformin treatment decreases endogenous hepatic glucose production by a mechanism thought to mainly involve inhibition of gluconeogenesis13,16. In addition, inhibition of hepatic glycogenolysis by metformin has been reported, but less consistently13,14. After its intestinal absorption, metformin concentration is high in the portal vein, resulting in the accumulation of higher metformin levels in the liver than in other surrounding organs27. Furthermore, PET imaging with 11C-metformin has confirmed that the drug is primarily taken up in the liver in both humans and mice27,28,29.
The main hepatic metformin transporter is OCT1. Mice deficient in OCT1 show a decrease in metformin accumulation in the liver, and the drug loses its ability to lower plasma levels of glucose28,63. Similarly, polymorphisms in OCT1 (SLC22A1) are thought to decrease the acute effects of metformin on glucose tolerance in humans64. However, the influence of variants of OCT1 with decreased function on metformin response has not been consistently reported31,32. The discrepancy in study results could be explained by key differences in the assessment of metformin response, which is measured either during a dynamic response to an oral glucose challenge64 or after long-term oral metformin use32. Although it seems clear that metformin could act via both acute and long-term direct and indirect effects to decrease hepatic glucose production, several conflicting mechanisms have been proposed to explain the inhibition of gluconeogenesis by the drug.
Controversy on metformin concentrations
Over the past few years, controversy has arisen concerning the use of high concentrations of metformin in preclinical (200–400 mg/kg) and in vitro studies (250 µM to 2 mM) using intact cells (specifically primary hepatocytes) when compared with the concentrations achieved in plasma from patients with diabetes mellitus treated with the drug65. However, owing to wide interindividual variability in metformin pharmacokinetics, the plasma distribution of the drug at steady-state is highly changeable in patients66,67, which considerably enlarges the range of clinically relevant metformin concentrations. In rodent studies, the optimal dosage was established on the ability of metformin to induce a relevant glucose-lowering effect, with an effective single oral dose of metformin in mice estimated to be around 200–250 mg/kg (ref.68,69,70,71). Of note, the high dose of metformin required in rodents compared with humans (humans require approximately 25–30 mg/kg per day, with patients with T2DM receiving an oral dose of 500 or 850 mg either twice or three times per day, up to a total of 2,550 mg per day) to achieve biological effects might in part result from a faster renal metformin clearance. Indeed, metformin has a mean plasma elimination half-life of 1–2 h in mice and 4–9 h in humans72,73.
For in vitro studies, metformin concentrations above those found in patients with diabetes mellitus (40–70 uM, measured in the portal vein) are considered clinically irrelevant24,65,74,75. However, studies in mice treated with oral metformin have shown an accumulation of a higher concentration of metformin in liver than in hepatic portal vein plasma27,55. Although concentrations of 100–250 μM can be viewed as supra-pharmacological, these concentrations are reached in the liver of mice treated orally with 50 mg/kg metformin (a dose that is generally accepted as falling in the clinical range)27. Interestingly, chronic treatment of mice fed a high-fat diet (HFD) with 50 mg/kg metformin (leading to plasma concentrations of 29 μM) improved metabolic parameters in HFD-fed mice; however, a higher dose of 200 mg/kg was needed to lower glucose in acute metformin tolerance tests69. The acute administration of 250 mg/kg metformin to fasted mice lead to metformin concentrations of ~120 μM and ~0.8 μmol/g in plasma and liver, respectively70. This concentration of metformin accumulating in the liver is similar to intracellular metformin concentrations measured in primary rat hepatocytes treated with 100 µM metformin for 4 h (ref.76). Therefore, the treatment of primary hepatocytes with high metformin concentrations that are above those found in the circulation might mimic therapeutic concentrations of the drug found in the liver.
Of note, the uptake of metformin into hepatocytes is slow, but the drug has the ability to accumulate at high concentrations within the cell and the mitochondrial matrix due to its positive charge, which allows it to interact with the polarized mitochondrial membrane39,45,77. Accordingly, the ratio of intracellular to extracellular metformin has been shown to be ∼5:1 in primary rat hepatocytes incubated with 100–500 µM metformin76. Consequently, the effects of metformin are both concentration-dependent and time-dependent. Moreover, this point has been well illustrated in studies showing inhibition of mitochondrial respiratory chain complex 1 in rat hepatoma H4IIE cells after long incubation (24 and 60 h) with low metformin concentrations (50–100 µM)39. Similarly, a decrease in glucose production occurs after incubation of mouse primary hepatocytes with 250 µM metformin for 8 h (ref.68), whereas a separate study showed inhibition of glucose output with lower metformin concentrations (80 µM) after a longer incubation of 24 h (ref.24). Therefore, effects observed with relatively high concentrations of metformin might also occur at lower concentrations after longer incubation. In future studies, it would be interesting to correlate the levels of intracellular metformin accumulation with the effect of the drug on hepatic glucose production.
Metformin-induced AMPK activation
The cellular energy sensor AMPK is a critical regulator of energy homeostasis that is activated by energy stresses that increase cellular ADP:ATP and/or AMP:ATP ratios23. In 2001, metformin was reported to stimulate AMPK activation in rat primary hepatocytes77. Furthermore, this effect was later attributed to inhibition of mitochondrial respiratory chain complex 1 by the drug, which causes an altered cellular energy charge characterized by a decline in intracellular ATP levels concomitantly with an increase in intracellular ADP and AMP intracellular levels23,43,68 (Fig. 2). An elegant 2010 study confirmed this finding using cell lines that express an AMPK mutant insensitive to AMP and showing that metformin-induced AMPK activation is dependent on AMP binding to the regulatory AMPK γ-subunit, even if changes in the levels of intracellular adenine nucleotide levels are technically undetectable78.
Metformin is transported into hepatocytes mainly through OCT1 and accumulates in mitochondria. a–d | In mitochondria, metformin partially inhibits mitochondrial respiratory chain complex 1 (complex 1), resulting in decreased ATP levels and accumulation of AMP. Thus, metformin has a mild effect on the overall cellular energy charge. a | Metformin-induced changes in the AMP:ATP ratio activate AMPK through its phosphorylation by LKB1. Subsequently, AMPK inhibits gluconeogenic gene transcription (Pck1 and G6pc) via the phosphorylation and cytoplasmic sequestration of the transcriptional cofactor CRTC2. This AMPK-dependent mechanism has been challenged by the use of liver AMPK-deficient mice. b | Gluconeogenesis is a highly energy-consuming metabolic pathway. Therefore, reduction in cellular ATP levels is sufficient to reduce glucose production flux. In addition, elevation of AMP levels contributes to inhibition of glucose production through allosteric inhibition of fructose-1,6-bisphosphatase 1 (FBP1), a key gluconeogenic enzyme. c | Metformin-induced AMP accumulation inhibits adenylate cyclase and decreases cAMP synthesis, resulting in decreased protein kinase A (PKA) activity and downstream signalling. Gluconeogenesis is suppressed through both the decrease in gluconeogenic enzyme activity and the inhibition of glucagon-induced gluconeogenic gene expression associated with the lack of phosphorylation of regulators (for example, CREB1 and I3PR). d | Chronic metformin-induced AMPK activation might indirectly decrease gluconeogenesis by enhancing hepatic insulin sensitivity. AMPK inhibits lipogenesis through phosphorylation and inhibition of acetyl-CoA carboxylase (ACC), thereby decreasing malonyl-CoA synthesis, which activates carnitine palmitoyltransferase 1 (CPT1) and stimulates fatty acid oxidation by increasing the import of acyl-CoA into mitochondria. Over time, metformin-induced AMPK activation decreases hepatic steatosis and improves insulin sensitivity, which in turn inhibits gluconeogenesis. GLUT2, glucose transporter type 2; PFK/FBPase, 6-phosphofructo-2-kinase/fructose 2,6-bisphosphatase 1.
Although it is assumed that AMPK activation is generally mediated by the canonical energy stress mechanism, evidence is accumulating that glucose starvation can activate AMPK by a non-canonical mechanism that is independent of changes in the AMP:ATP ratio, which involves the formation of a complex comprising the v-ATPase, Ragulator, axin, LKB1 and AMPK at the surface of lysosomes79,80. Whether metformin could also activate AMPK via this lysosomal pathway has been examined in mice and primary hepatocytes deficient in axin. Interestingly, metformin-mediated AMPK activation was abolished in liver-specific axin-deficient mice after chronic intraperitoneal administration of 50 mg/kg of metformin and in axin-deficient hepatocytes treated with 70 µM metformin for 3 h (ref.81). Nonetheless, the consequence of axin deficiency on metformin-induced inhibition of glucose production has not been addressed. These data raise the intriguing possibility that metformin acts at the lysosome surface as well as within the mitochondria; therefore, future studies examining the functional link between metformin and the lysosomal pathway are warranted.
An important 2001 study suggested that activation of AMPK is necessary for metformin-induced inhibition of glucose production in rat primary hepatocytes based on the use of the AMPK inhibitor Compound C (also known as dorsomorphin)77. Later, the involvement of AMPK in metformin action was confirmed by a study showing that deletion of LKB1 (the upstream kinase of AMPK) prevents activation of AMPK and suppresses the glucose-lowering effects of metformin in vivo in HFD-fed mice82. Moreover, this study suggested that activation of the LKB1–AMPK signalling pathway by metformin repressed the gluconeogenic transcriptional programme through the phosphorylation and nuclear exclusion of the transcriptional co-activator cAMP-response element-binding protein (CREB)-regulated transcription co-activator 2 (CRTC2), which is a key regulator of gluconeogenic gene expression82 (Fig. 2a). In addition, other mechanisms induced by metformin that lead to the inactivation of CRTC2 and involve AMPK have also been proposed83,84,85.
Importantly a breakthrough 2010 study challenged the aforementioned model of AMPK-dependent transcriptional inhibition68. This study demonstrated that metformin-induced inhibition of glucose production is preserved in primary hepatocytes, even when the expression of gluconeogenic enzymes is forced through overexpression of peroxisome proliferator-activated receptor-γ co-activator-1α (PGC1α), which is a master co-activator of gluconeogenic genes. As such, the findings indicate that metformin inhibits the activity of gluconeogenic enzymes rather than their gene expression68. Importantly, metformin still inhibits glucose production and gluconeogenic gene expression in primary hepatocytes from mice deficient in AMPK catalytic subunits or LKB1 expression in the liver, which demonstrates that neither AMPK nor LKB1 is crucial for inhibition of hepatic glucose production by metformin68. Furthermore, hepatic AMPK-deficient mice display normal glycaemia, pyruvate tolerance and blood glucose-lowering effect of metformin68,86,87.
To further support the absence of a direct effect of AMPK activation on the regulation of hepatic gluconeogenesis, treatment with the small-molecule direct AMPK activator (A-769662) showed no influence on endogenous hepatic glucose production both in vitro in primary hepatocytes68 and in vivo in mice75. Similarly, the more potent AMPK activators PF-249 and PF-739 have no effect on endogenous glucose production after acute administration in mice87. Thus, although metformin activates AMPK by reducing cellular energy charge, the metformin-induced acute suppression of glucose production is seemingly independent of the LKB1–AMPK signalling pathway and transcriptional alterations.
Several studies have suggested that the effects of metformin that are associated with a reduction in cellular energy charge result from supra-pharmacological concentrations of the drug; at therapeutic concentrations, metformin has been proposed to act without affecting cellular energy levels24,74,75,88. Specifically, low concentrations of metformin have been shown to suppress glucose production in primary hepatocytes via AMPK activation, independently of any detectable changes in the AMP:ATP ratio24.
AMP-dependent mechanisms
It has been proposed that metformin acutely inhibits hepatic glucose production through AMPK-independent mechanisms that involve modulation of cellular energy status and allosteric inhibition of crucial gluconeogenic enzymes. Gluconeogenesis is a highly energy-consuming metabolic pathway, which requires six ATP equivalents (four ATP and two GTP) per molecule of glucose synthesized. As such, the decrease in intracellular ATP levels resulting from the inhibition of mitochondrial respiratory chain complex 1 could be the simplest explanation for the reduction in hepatic gluconeogenic flux by metformin68. This model is supported by the strong correlation between decreases in intracellular ATP levels and inhibition of glucose production in mouse primary hepatocytes that are incubated with metformin, which emphasizes the close relationship between hepatic energy status and glucose production68.
The alteration in cytosolic and mitochondrial NADH:NAD+ ratios by metformin38,39 could also be involved in the inhibition of glucose production (Fig. 2b). Furthermore, the mild metformin-induced increase in intracellular AMP levels through compromised hepatic energy status might also have a major role in the flux control of gluconeogenesis via an allosteric inhibition of fructose 1,6-bisphosphatase (FBP1), which is a key enzyme in gluconeogenesis68 (Fig. 2b). Interestingly, in one study the glucose-lowering effect of metformin was shown to be significantly decreased in a mouse model bearing a point mutation in FBP1 that rendered it insensitive to inhibition by AMP70. In addition, it has been shown that the increase in intracellular AMP levels induced by metformin decreases glucagon-stimulated gluconeogenesis in the liver71. Indeed, AMP inhibits the glucagon-induced activation of adenylate cyclase (the enzyme that catalyses the cyclization of ATP into cyclic AMP) by binding to an inhibitor site on the enzyme known as site P, which subsequently lowers intracellular cyclic AMP levels, decreases protein kinase A (PKA) activity and decreases the phosphorylation of essential factors that stimulate gluconeogenesis, which include 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 1, inositol trisphosphate receptor and CREB1. These events in turn lead to a decrease in glucagon-stimulated glucose production71 (Fig. 2c).
Alteration of redox state
An alternative model of metformin action has been suggested, in which the drug decreases hepatic gluconeogenesis independently of AMPK-mediated and energy charge-mediated effects through a direct inhibition of mitochondrial glycerol-3-phosphate dehydrogenase (mG3PDH)75,88. The concerted action of mG3PDH, which is located on the outer side of the inner membrane, and the cytosolic isoform of G3PDH (cG3PDH) creates the glycerol–phosphate shuttle. This shuttle is one of two shuttles that transfer the cytosolic reducing equivalents (that is, NADH) directly into the mitochondrial electron transport system for reoxidation. Importantly, two outcomes of the inhibition of the glycerol–phosphate shuttle by metformin have been proposed: first, a disruption of glucose production from glycerol, and, second, an increase in cytosolic redox potential (NADH:NAD+), which impedes the utilization of lactate for gluconeogenesis75,88 (Fig. 3a). This mechanism predicts that metformin suppresses gluconeogenesis from glycerol or reduced gluconeogenic substrates (such as lactate) but not from oxidized substrates (such as alanine or pyruvate).
a | Metformin suppresses gluconeogenesis through a direct inhibition of mitochondrial glycerol-3-phosphate dehydrogenase (mG3PDH), an enzyme involved in the glycerol–phosphate shuttle. Metformin-induced inhibition of mG3PDH disrupts glucose production from glycerol and increases cytosolic redox potential (NADH:NAD+), which impedes the utilization of lactate for glucose production. b | The accumulation of metformin in mitochondria caused by its positive charge leads to mitochondrial depolarization and inhibition of the electrogenic transporter for aspartate (Asp) of the malate–aspartate (Mal–Asp) shuttle, resulting in an increase in the cytosolic NADH:NAD+ ratio. Metformin-induced inhibition of the malate–aspartate shuttle stimulates the glycerol–phosphate shuttle, leading to a decrease in levels of glycerol-3-phosphate (glycerol-3P), a potent allosteric inhibitor of phosphofructokinase 1 (PFK1). As a result, gluconeogenesis is inhibited through the partitioning of gluconeogenic substrate towards glycolysis. DHAP, dihydroxyacetone phosphate; FBP1, fructose-1,6-bisphosphatase 1; cG3PDH, cytosolic G3PDH; GLUT2, glucose transporter type 2; LDH, lactate dehydrogenase; OCT1, organic transporter 1.
The aforementioned hypothesis has been experimentally supported by studies in vivo and in primary hepatocytes75,88. Metformin treatment decreased hepatic glucose production, while increasing cytosolic redox and decreasing mitochondrial redox states in the liver75,88. These findings were replicated in mG3PDH-deficient mice75. Similarly, primary hepatocytes that are deficient in mG3PDH showed inhibited gluconeogenesis, which recapitulates the effects of metformin in control hepatocytes. In mG3PDH-deficient hepatocytes, glucose production was abolished from glycerol or reduced substrates (for example, lactate) but was unaffected from oxidized substrates (for example, alanine or pyruvate)75. However, this model of metformin action raises several concerns. The glycerol–phosphate shuttle has an important role in tissues that oxidize glucose rapidly (such as skeletal muscle and the brain) to regenerate NAD from NADH that is produced by glycolysis89,90. However, in the liver, the malate–aspartate shuttle is the primary NADH shuttle91,92. Furthermore, as highlighted elsewhere93, inhibition of the glycerol–phosphate shuttle might not be sufficient to affect gluconeogenesis. In a mouse model, disruption of the glycerol–phosphate shuttle had no effect on glycaemia whereas disruption of the malate–aspartate shuttle lowered fasting glycaemia and increased the cytosolic NADH:NAD+ ratio63.
Finally, the model of inhibition of mG3PDH by metformin was challenged in a 2019 study74. In this study, researchers found that metformin had no effect on mG3PDH activity in cell-free assays and that the drug inhibited hepatic glucose production from both reduced and oxidized gluconeogenic substrates. In addition, the study showed that pharmacological inhibition of mG3PDH had no effect on glucose production74. These findings led to the proposal that metformin increases the cytosolic NADH:NAD+ ratio, independently of the inhibition of mG3PDH or of mitochondrial respiratory chain complex 1, via mitochondrial membrane depolarization, which is caused by metformin accumulation in mitochondria. In turn, this accumulation then inhibits the electrogenic transporter for aspartate of the malate–aspartate shuttle74. However, the same study suggested that metformin inhibits gluconeogenesis by an energy charge-independent and redox-independent mechanism. Metformin-induced inhibition of the malate–aspartate shuttle might stimulate the glycerol–phosphate shuttle, leading to a decrease in glycerol-3-phosphate (glycerol-3P, a potent allosteric inhibitor of phosphofructokinase 1 (PFK1)) levels. As a result, gluconeogenesis is inhibited through the partitioning of substrates towards glycolysis74 (Fig. 3b).
The alteration in hepatic redox potential caused by metformin probably has an important role in the inhibition of gluconeogenesis. However, concern exists about whether the effect of metformin on the redox state (that is, the NADH:NAD+ ratio) is dissociable from an effect on energy charge (that is, the AMP:ATP ratio). NADH is the major source of electrons that flow through the mitochondrial electron transport chain to generate ATP. Consequently, whether metformin hampers NADH mitochondrial transfer by mG3PDH inhibition or alternatively by acting to depolarize the mitochondrial membrane, de facto cellular energy charge is expected to be impaired. Thus, a model in which metformin modifies redox status without compromising energy charge seems contradictory. Of note, in the studies supporting the metformin redox state hypothesis, changes in the redox state induced by metformin occurred concomitantly with AMPK activation74,75,88, which is a very sensitive marker of cellular energy stress. In addition, in the proposed metformin mechanism of action that involves a mild inhibition of mitochondrial respiratory chain complex 1, both the AMP:ATP and the NADH:NAD+ ratios are altered38,39. Altogether, despite intense debate, the relative contributions of energy charge and redox state changes in metformin action still need to be established.
Effects of chronic AMPK activation
Although AMPK activation does not account for the acute glucose-lowering effect of metformin, chronic metformin-induced AMPK activation might indirectly inhibit hepatic glucose production by increasing hepatic insulin sensitivity. For example, pharmacological activation of AMPK has been reported to alleviate hepatic steatosis in fatty liver mouse models86,94,95 via the inhibitory phosphorylation of acetyl CoA carboxylase 1 (ACC1) and ACC2, which results in the inhibition of lipid synthesis and stimulation of fatty acid oxidation (Fig. 2d). Moreover, the importance of the AMPK–ACC pathway in the action of metformin on glucose and lipid metabolism was further demonstrated in mouse models in which ACC1 and ACC2 were rendered insensitive to AMPK phosphorylation69. These mouse models are resistant to the lipid-lowering and glucose-lowering effects of metformin, which indicates that the decrease in blood levels of glucose in response to metformin depends on the ability of the drug to lower the intracellular lipid content. Thus, the ability of metformin to improve insulin sensitivity through the AMPK–ACC pathway might, at least in the long-term, contribute to the suppression of hepatic glucose production in the context of insulin resistance. Importantly, this mechanism might also explain the resistance of HFD-fed LKB1-deficient mice to the glucose-lowering effect of metformin, which has been reported previously82. However, it is not clear whether this mechanism is relevant in humans owing to the controversy that exists regarding the efficacy of metformin treatment in reducing hepatic lipid content96.
Metformin and meta-inflammation
The chronic low-grade inflammation associated with obesity, which is also called meta-inflammation or metaflammation, is one of the major contributors to insulin resistance and impaired glucose and lipid homeostasis in the metabolic syndrome and T2DM97. The immune system is now well recognized to have a key role in the regulation of whole-body metabolic homeostasis through a variety of innate and adaptive immune cells that are present in various organs, most notably in adipose tissue and the liver98,99. In obesity, the over-supply of dietary nutrients (especially fatty acids) triggers enhanced recruitment of monocytes to metabolic organs (that is, adipose tissue and liver), which become polarized towards pro-inflammatory ‘M1-like’ macrophages. Importantly, M1-like macrophages are believed to mediate, at least in part, tissue-specific insulin resistance100.
In healthy adipose tissue, a network of type 2 immune cells secretes anti-inflammatory cytokines, which trigger macrophages to become polarized towards an alternatively activated ‘M2-like’ state. These M2-like macrophages support the resolution of inflammation and tissue-specific insulin responsiveness, the latter through a currently largely unknown mechanism (or mechanisms). In obesity and T2DM, this finely tuned system is altered101, leading to a change in adipose tissue macrophage polarization in favour of classically activated M1-like macrophages, which orchestrate insulin resistance through enhanced secretion of pro-inflammatory cytokines100. Similarly, both newly recruited and resident liver macrophages (Kupffer cells) and neutrophils have been shown to play a pathogenic role in insulin resistance in both rodents and humans by increasing hepatic inflammation through the secretion of pro-inflammatory cytokines and elastase, respectively102,103,104.
Other immune cells are present in adipose tissue and the liver that are also involved in meta-inflammation, for example CD4+ T helper 1 (TH1) cells and TH17 cells, B cells and natural killer T cells98,99. At the molecular level, both in vitro and in vivo studies in rodent models of obesity have shown that the pro-inflammatory cytokines produced by macrophages (IL-6, TNF and IL-1β), TH1 cells (IFNγ) and TH17 cells (IL-17) contribute to the inhibition of the canonical insulin signalling, notably through activation of nuclear factor-κB (NFκB) and JNK pathways105. By contrast, the TH2 cell cytokines IL-4 and IL-13 have been shown to promote insulin sensitivity and glucose homeostasis, at least in part by triggering the JAK–STAT–PPAR axis in various metabolic organs106,107,108,109,110. In addition, the production of IL-10 and TGFβ by regulatory T (TReg) cells has been linked to protection against insulin resistance in mouse models of obesity111,112.
Modulation of immune cell functions
Some clinical studies have suggested beneficial effects of metformin on systemic inflammatory markers, such as serum levels of TNF, IL-6, PAI1 and the neutrophil-to-lymphocyte ratio, in patients with T2DM113,114,115 or polycystic ovary syndrome (PCOS)115 (Box 2). However, most of the data supporting anti-inflammatory properties of metformin came from in vivo studies in HFD-fed obese mice116,117,118,119 and a zebrafish model of obesity120, or in vitro and ex vivo experiments with supra-therapeutic concentrations of the drug in various immune cell types, particularly macrophages115,121,122,123,124,125,126,127. In line with this observation, metformin has been found to inhibit the lipopolysaccharide (LPS)-induced expression of pro-inflammatory cytokines (IL-6 and TNF) in both mouse peritoneal macrophages and human monocyte-derived macrophages121,124. At the mechanistic level, this anti-inflammatory effect was attributed, at least in part, to an AMPK-dependent decrease in NF-κB activity that is secondary to either inhibition of the JNK signalling pathway or activating transcription factor 3 induction (ATF3)121,124 (Fig. 4). Furthermore, metformin has been reported to inhibit monocyte-to-macrophage differentiation in the THP1 macrophage cell line via an AMPK-mediated inhibition of STAT3 activation, which might contribute to reducing atherosclerosis by lowering the inflammatory environment within the vessel wall124.
Obesity-associated meta-inflammation results partly from pro-inflammatory activation of tissue-resident macrophages, which secrete pro-inflammatory cytokines in metabolic organs (that is, liver and adipose tissue) and contributes to insulin resistance at least partly by inhibiting insulin signalling. Moreover, obesity is often associated with changes in microbiota composition (dysbiosis) that result in bacterial secretion of various immunomodulatory components (for example, short-chain fatty acids (SCFAs) or lipopolysaccharide (LPS)). Metformin could improve tissue-specific inflammation and insulin sensitivity by inducing regulatory T cells (TReg) and/or macrophages to polarize towards alternatively activated anti-inflammatory macrophages. These effects are thought to occur through direct and indirect pathways that involve changes in the gut microbiota (for example, increases in abundance of Akkermansia species) and both AMP-activated protein kinase (AMPK)-dependent and AMPK-independent mechanisms. ATF3, activating transcription factor 3; mTOR, mechanistic target of rapamycin; NF-κB, nuclear factor-κB; OXPHOS, oxidative phosphorylation; TGFβ, transforming growth factor-β; TLR4, toll-like receptor 4; TNF, tumour necrosis factor.
Metformin has also been shown to inhibit LPS-induced IL-1β secretion whilst potentiating IL-10 expression in mouse bone marrow-derived macrophages. However, the mechanism is thought to be independent of AMPK activation and might instead result from the specific inhibition of mitochondrial reactive oxygen species (ROS) production driven by reverse electron transport (RET) at the mitochondrial respiratory chain complex 1 (ref.122). Of note, it has also been proposed that this specific inhibition of RET-mediated ROS production contributes to the decrease in oxidative stress-related cell death induced by metformin46,56,58 and to its cardioprotective properties.
AMPK, as a central regulator of cellular metabolism, has a prominent role in both myeloid cell polarization and T cell effector responses128,129. Indeed, an emerging paradigm suggests that metabolic reprogramming and immune cell functions are intricately linked and that immune cell activation, proliferation and polarization are underpinned by profound changes in core metabolic pathways130. For example, pro-inflammatory macrophages and effector T cells mostly rely on glycolysis for the generation of ATP, whereas anti-inflammatory macrophages and TReg cells depend instead on AMPK-dependent fatty acid oxidation and mitochondrial OXPHOS128,129 (Fig. 4). So far, the contribution of AMPK-induced changes in cellular metabolism to the anti-inflammatory effects of metformin remains unknown and deserves dedicated investigation. Moreover, as the immunomodulatory properties of metformin have also been suggested to underlie some of its beneficial effects on autoimmune inflammatory diseases131,132,133,134 and cancer135,136,137,138, further studies are required to decipher the immune-mediated mode of action of metformin.
Metformin action in the intestine
Gut as the primary target of metformin
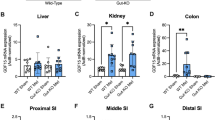
Although it is generally accepted that the anti-hyperglycaemic effect of metformin is primarily attributed to its action on the liver139, accumulating data suggest that the metabolic benefits of the drug might also be due to actions in the gut19,21,140 (Fig. 5). Early studies provided evidence that intravenous administration of metformin is less effective than oral dosing for inducing its glucose-lowering action141,142,143. In addition, metformin has been reported to inhibit intestinal glucose absorption in the proximal small intestine, which is seemingly associated with increased glucose utilization by the enterocytes in the intestine and increased lactate production through anaerobic metabolism19,144,145,146,147. Interestingly, imaging with PET–computed tomography (CT) in patients with T2DM treated with metformin revealed elevated intestinal glucose utilization as demonstrated by diffusely increased 18F-fluoro-deoxyglucose (FDG, a non-metabolized glucose analogue) uptake in the colon and to a minor extent in the small intestine148. Of note, metformin is considered to increase the intestinal utilization of glucose that is taken up from the circulation; however, increased uptake of glucose from the intestinal lumen could also be contributing to the metabolic benefits of metformin19,149. The mechanism of metformin stimulation of intestinal glucose utilization is debated, but might be related to the redistribution of GLUT2 at the apical membrane34,150,151 of enterocytes or increased expression of the sodium–glucose cotransporter 1 (SGLT1)152,153.
Metformin is taken up from the intestinal lumen in enterocytes by plasma membrane monoamine transporter (PMAT) and OCT3 and transported into the bloodstream by OCT1. Metformin stimulates secretion of the glucose-lowering hormone glucagon-like peptide 1 (GLP1) from enteroendocrine L cells by direct and indirect mechanisms. Of note, GLP1 secretion is directly controlled by muscarinic M3 receptor (M3R), Wnt signalling and AMPK activation in L cells. The GLP1 secretory effects directly induced by metformin are also mediated indirectly by modulation of the bile acid pool and gut microbiota composition. For example, the inhibitory effect of metformin on apical sodium-dependent bile acid transporter (ASBT) reduces the reabsorption of bile acids, leading to an increase in luminal concentrations of bile acids and subsequent stimulation of the bile acid receptor TGR5, as well as a decrease in intracellular bile acid concentrations limiting activation of farnesoid X receptor (FXR). In addition, metformin increases the abundance of short-chain fatty acid (SCFA)-producing bacteria and facilitates SCFA-induced GLP1 secretion via signalling through GPR41 and GPR43 in L cells. Increases in the abundance of Lactobacillus species increases release of GLP1 by a glucose–SGLT1-sensing mechanism. Decreases in the abundance of Bacteroides fragilis elevate the levels of the bile acid glycoursodeoxycholic acid (GUDCA, a potent endogenous antagonist of FXR) to modulate GLP1 secretion. GLP1 acts locally in the gut by activating a gut–brain–liver neuronal axis that contributes to the regulation of blood levels of glucose via a reduction in hepatic glucose production. SGLT1, sodium-coupled glucose transporter 1.
Metformin accumulates at very high concentrations in the small intestine and the large intestine (concentrations up to 30–300 times greater than in plasma)27,154, suggesting that the gastrointestinal tract is probably an important site of metformin action for the management of T2DM. Moreover, studies in rat models and humans with T2DM have linked gastrointestinal metformin exposure in the proximal and distal small intestine to suppression of hepatic glucose production and attenuated glycaemic responses to oral glucose155,156. In addition, oral administration of a delayed-release formulation of metformin, which is released slowly in the distal small intestine (ileum) where absorption is low (resulting in low plasma metformin exposure) has been shown to be as effective as the standard immediate-release formulation in lowering fasting plasma levels of glucose in individuals with T2DM20,157. In further support of a dissociation between metformin plasma exposure and gut-restricted metformin action, inhibition of the transporter MATE, which mediates hepatic and renal elimination of metformin, has little effect on blood levels of glucose despite an increase in metformin concentrations in plasma in humans158 or in the liver in mice28. Taken together, these findings not only indicate that the response to metformin is related to its systemic exposure but also illustrate the quantitative importance of the gastrointestinal tract in the glucose-lowering effect of metformin.
Effects of metformin on GLP1 release
One mechanism proposed to underlie the gut-mediated glucose-lowering effect of metformin relies on incretins. An increasing body of clinical evidence suggests that metformin increases the plasma levels of the gut incretin hormone, glucagon-like peptide 1 (GLP1) in patients with T2DM159,160. In addition, metformin has been reported to potentiate the responsiveness to incretin in mice by increasing the expression of pancreatic islet GLP1 receptor161. Of note, GLP1 has the capacity to improve blood glucose homeostasis in T2DM via pleiotropic actions, including feedback mechanisms that act through a gut–brain neuronal axis, the stimulation of glucose-stimulated insulin secretion, the inhibition of pancreatic glucagon secretion, the slowing of gastric emptying and a reduction in appetite and food intake.
The hormone GLP1 is secreted by intestinal enteroendocrine L cells in response to the presence of nutrients in the intestinal lumen. These cells are located mainly in the distal small intestinal mucosa and the colon. However, evidence suggests that the administration of metformin to the lower and upper gut is not critical to the capacity of the drug to increase plasma levels of GLP1 and achieve its glucose-lowering effect in humans156,162. Instead, a mechanism has been suggested in which metformin acts to decrease glucose absorption in the upper small intestine, which causes an increase in glucose concentrations in more distal regions of the gut. In turn, the increase in glucose concentrations then stimulates GLP1 secretion from the large population of enteroendocrine L cells located within the ileum145. The contribution of GLP1 signalling to the acute postprandial effect of metformin on plasma levels of glucose was confirmed in a 2018 study in patients with T2DM. Infusion of the GLP1 receptor antagonist exendin 9-39 lowered the effect of metformin on postprandial glucose levels, which suggests that the metformin-induced secretion of GLP1 contributes to the mode of action of the drug163. However, pharmacological inhibition or deletion of incretin receptor signalling showed no statistically significant effect on the capacity of metformin to improve oral glucose tolerance in diet-induced obese mice161. This finding suggests that in mice, GLP1 secretion is not the predominant mechanism for the glucose lowering effect of metformin. Of note, incretin-based therapies are approved for use as add-ons to first-line metformin monotherapy to further amplify clinical outcomes, which supports the contribution of GLP1-independent mechanisms to the glucose-lowering effect of metformin164.
Metformin has been hypothesized to increase both fasting and postprandial levels of GLP1 by decreasing its degradation by dipeptidyl peptidase 4 (DPP4) or by directly increasing GLP1 secretion from enteroendocine L cells; however, in vitro studies have yielded inconsistent results (reviewed in refs145,165). A range of potential mechanisms for acute metformin-induced GLP1 secretion have been proposed from in vitro and ex vivo studies using intestinal L cell lines or human colonic tissue sections163. These mechanisms include a role for muscarinic acetylcholine receptor166, Wnt signalling167 and AMPK activation155.
Metformin has also been found in in vitro studies to protect enteroendocrine GLP1-secreting L cells against lipoapoptosis via a sustained activation of AMPK in these cells168. This effect might explain the elevated plasma levels of GLP1 that are associated with chronic metformin therapy. However, supporting evidence shows that metformin exerts an indirect stimulatory effect by increasing GLP1 secretion in response to the availability of the bile acid pool in the intestine of patients with T2DM160,169. For example, metformin induces a decrease in the overall absorption of intestinal bile salts by strongly decreasing their absorption from the ileum (via an active process). This effect occurs despite a small increase in absorption of bile acids in the jejunum (via passive non‐saturable concentration‐dependent diffusion)170. Possible mechanisms for metformin-induced suppression of active bile acid reabsorption in the ileum are inhibition of the apical sodium-dependent bile acid transporter165 and modulation of the transcriptional activity of farnesoid X receptor (FXR, a bile acid receptor) via an AMPK-mediated mechanism171 in enterocytes (Fig. 5). Importantly, this process results in increased luminal concentration of bile acids in the ileum and colon, which increases GLP1 secretion via the stimulation of Takeda G protein-coupled receptor 5 (TGR5) on enteroendocrine L cells172,173,174. Furthermore, the higher concentration of bile acids now present in the intestinal lumen might simultaneously decrease the activation of FXR in L cells, and hence facilitate GLP1 production and secretion175. Thus, the combination of these intestinal effects might further stimulate metformin-induced secretion of GLP1 from L cells.
Metformin action on the gut–brain neuronal axis
Emerging evidence suggests the importance of nutrient sensing in the gut for the control of glucose homeostasis, through the activation of a gut–brain–liver negative feedback system to inhibit hepatic glucose production176. For example, glucose sensing in the upper small intestine is mediated through a SGLT1–GLP1 receptor (GLP1R)-dependent neuronal axis153. As such, the infusion of glucose into the upper small intestine stimulates intestinal GLP1 release, as revealed by increased portal vein active GLP1 levels; however, there is no effect on peripheral circulating levels of GLP1. These findings suggest that GLP1 acts locally in the gut by activating GLP1R on vagal afferent nerves that innervate the gut mucosa153,177.
The mechanism by which intestinal glucose contributes to the release of GLP1 suggests a role for SGLT1-mediated glucose uptake in enteroendocrine cells178,179. To evaluate the contribution of small-intestinal glucose-sensing mechanisms to the metabolic benefits of gut-restricted metformin, the effect of intraduodenal metformin infusion was assessed in rodents in response to HFD-induced impairments in intestinal glucose sensing. For example, 1-day treatment of metformin administered directly into the upper small intestine of HFD-fed rats was found to restore upper small-intestinal SGLT1-dependent glucose sensing, which resulted in the restoration of glucose-stimulated GLP1 release and suppression of glucose production153. In treated rats, increased expression of mucosal SGLT1 was dependent on changes in the composition of the upper small-intestine microbiota, as transplantation of the microbiota from metformin-treated rats into untreated HFD-fed rats restored the activity of the SGLT1-dependent glucose-sensing pathway180. However, future work is warranted to elucidate how metformin affects the composition of the microbiota (further discussed below) and modulates small intestinal glucose sensing.
Interestingly, in an obese diabetic rodent model, infusion of metformin in the upper small intestine activates AMPK in the duodenal mucosa layer to lower hepatic glucose production. This effect is independent of upper small-intestinal glucose sensing and acts through a gut–brain–liver neuronal network that triggers a GLP1R–PKA-dependent pathway155. The action of intraduodenal infusion of metformin on the inhibition of hepatic glucose production is abrogated by co-infusion with a GLP1R antagonist and through chemical inhibition of neurotransmission via afferent fibres in the gut. Moreover, this study highlights the importance of an intestinal GLP1-dependent mechanism that is relayed by the activation of GLP1R on vagal afferent neurons in mediating the acute effects of intraduodenal metformin155. However, the underlying mechanism linking duodenal AMPK activation and GLP1 signalling has not yet been fully elucidated. It remains uncertain whether metformin activates AMPK directly in GLP1-secreting cells or acts indirectly (for example, via bile acid accumulation) to stimulate the release of GLP1 for the subsequent stimulation of GLP1R on the vagus afferent nerve (Fig. 5).
The glucoregulatory role of duodenal AMPK activation has also been demonstrated in response to intraduodenal infusion of resveratrol (a natural polyphenolic compound known for its many beneficial effects to human health) or the AMPK activator A-769662, indicating the therapeutic relevance of targeting duodenal AMPK for gut-mediated antidiabetic therapies155,181,182. A 2018 study showed that inhibition of mTOR signalling is also required for the glucose-lowering effect of intraduodenal metformin but that mTOR acts independently of AMPK signalling177, thereby adding a novel potential gut target to potently lower glucose levels in patients with T2DM.
Modulation of gut microbiota
Evidence is accumulating that suggests a causal relationship between intestinal microbiota dysbiosis (defined as an imbalance in the normal populations of microorganisms present in the gastrointestinal tract) and metabolic diseases such as T2DM183,184. For example, metagenomic studies have shown a statistically significant effect on bacterial diversity present in faecal samples associated with metformin treatment in rodent models185,186,187 or individuals with T2DM22,183,188,189. These findings suggest that the changes in composition of gut microbiota might be a contributing factor to the glucose-lowering effect of metformin (Fig. 5). Accordingly, depletion of the gut microbiota by oral intake of antibiotics abrogates the glucose-lowering capacity of metformin in rodents186. Furthermore, faecal microbiota transplantation from metformin-treated donor rodents (which improved glucose tolerance) to HFD-fed recipient animals induced similar improvements in glucose tolerance in the recipients153,189.
Metformin treatment in patients with T2DM has been reported to alter the structure of gut bacterial communities by causing a substantial shift in the abundance of specific subsets of bacterial taxa, which partially restores faecal gut dysbiosis that is associated with T2DM22,160,183,188. However, these studies analysed the long-term therapeutic outcome in patients on a stable dose of metformin for more than 3 months and could be the result of changes in microbiota composition that are secondary to the subsequent metabolic improvements. However, evidence is now growing that metformin has rapid effects on the composition and function of the gut microbiota, which contribute to the antidiabetic effect of the drug. In a study in treatment-naive individuals with T2DM, a dramatic shift in the composition of the gut microbiota was found after metformin treatment for 2 and 4 months and was associated with increases in the abundance of Escherichia species and decreases in Intestinibacter species189. Moreover, another study of individuals with newly diagnosed T2DM showed decreased abundance of Bacteroides fragilis only 3 days after the first treatment with metformin180.
Although the aforementioned studies clearly document the effects of metformin on the distal gut microbiota, a 2018 study indicates that metformin also alters microbiota composition in the upper small intestine153, where metformin accumulates at high concentrations27,154. This effect could also affect tolerance of metformin (Box 1). Interestingly, administration of metformin in rodents in the upper small intestine for 1 day was associated with improved glucose sensing and rapid changes in upper small-intestinal microbiota composition, with increased abundance of Lactobacillus species153. Moreover, intestinal microbiota transplantation experiments from metformin-treated HFD-fed rats to recipient untreated HFD-fed rats confirmed the key role of changes in the upper small-intestinal microbiota in mediating metformin-induced restoration of glucose sensing153. Thus, metformin-induced changes of microbiota in both the upper and lower gastrointestinal tracts might be required to regulate glucose homeostasis.
In addition to the modulation of microbiome composition, metformin treatment has been reported to improve metabolic functions of the microbiome and interactions with host metabolism. This effect is consistent with the enrichment of short-chain fatty acid (SCFA)-producing bacteria including Akkermansia, Bifidobacterium, Blautia, Butyrivibrio, Lactobacillus, Megasphaera, Prevotella and Shewanella species in metformin-treated patients with T2DM, which results in elevated levels of SCFAs in the colon, with potential metabolic benefits for the host22,187,188,189,190. Interestingly, in silico analysis of the metabolism of prevalent bacteria from the human gut following metformin treatment using genome-scale metabolic models revealed important commensal and competing behaviour in the production of important SCFAs and their association with host cellular metabolism191. An increase in the colonic production of SCFAs in mice, especially butyrate and propionate, is known to trigger intestinal gluconeogenesis and induce metabolic benefits, including a decrease in hepatic glucose production192. In addition, manipulation of SCFA production through gut microbial fermentation of prebiotics (non-digestible dietary ingredients that induce the growth or activity of a limited number of beneficial bacterial species in the colon)193 or by delivering propionate specifically to the colon has been reported to increase plasma levels of GLP1 in humans, leading to a decrease in food intake and amelioration of T2DM features (for example, improved glycaemic response)194,195.
The gut microbiota also plays an important role in bile acid homeostasis, by generating unconjugated and secondary bile acids196. Moreover, metformin influences the ability of the microbiota to alter the bile acid pool by decreasing the abundance of B. fragilis and its bile salt hydrolase activity180. These changes are accompanied by increased levels of the bile acid glycoursodeoxycholic acid (GUDCA), which is a potent endogenous antagonist of FXR. Importantly, this effect contributes to elevated GLP1 production via an AMPK-independent mechanism180. Notably, metformin-induced improvements in glucose tolerance have been found to be transmitted by faecal microbiota transplantation from metformin-treated patients with T2DM into antibiotic-treated and microbiota-depleted HFD-fed mice. The action of metformin on a gut B. fragilis–GUDCA–intestinal FXR axis was further validated by the abrogation of the beneficial effects of metformin after supplementation of HFD-fed mice with B. fragilis or deletion of intestinal FXR180. These data highlight the crosstalk between microbiota and host cells and the potential therapeutic value of gut microbiota remodelling for the treatment of T2DM and related diseases (such as non-alcoholic fatty liver disease).
A number of studies have highlighted the higher abundance of the mucin-degrading bacteria Akkermansia muciniphila in the distal gut of metformin-treated humans with T2DM185,187,188,189 and metformin-treated HFD-fed mice186. Moreover, A. muciniphila has been reported to improve metabolic disorders possibly by increasing the endogenous production of endocannabinoids, which reduce inflammation and promote secretion of GLP1 (ref.197). Furthermore, the beneficial role of A. muciniphila in metabolic homeostasis has been demonstrated by its administration as a probiotic to HFD-fed mice, which resulted in improved glucose tolerance compared with untreated animals186. Interestingly, these effects were enhanced by treatment with pasteurized A. muciniphila and were partly recapitulated by oral administration of a specific membrane protein isolated from A. muciniphila, providing support for its future use in therapy198,199. However, data published in 2017 do not support a significant correlation between A. muciniphila abundance and a decrease in HbA1c in a cohort of treatment-naive individuals with T2DM who received metformin for 4 months189.
The mechanisms by which metformin selectively shapes the gut microbiota remain to be clarified but could be mediated by both direct and indirect actions of the drug. The effect of metformin on the abundance of A. muciniphila has been attributed to its action on intestinal mucin-producing goblet cells, which favours the production of mucus for the growth of the bacterium186. However, research has demonstrated that metformin can also directly promote the growth of A. muciniphila as well as Bifidobacterium adolescentis189, and inhibit the growth of B. fragilis189, Fusobacterium nucleatum and Clostridium perfringens200 in bacterial cultures. To explain the direct inhibitory effect of metformin on microbial growth, it has been hypothesized that metformin might inhibit the bacterial NADH–menaquinone oxidoreductase (NDH1) complex, which is closely related to the eukaryotic mitochondrial respiratory chain complex 1, and interfere with ATP production201. Interestingly, metformin was found to impair folate and methionine metabolism of intestinal microbiota in Caenorhabditis elegans202, via a mechanism that might also contribute to modulation of the function and growth of B. fragilis in the intestine of patients with T2DM treated with metformin189. In a 2017 study investigating the direct interactions between metformin and microbiota in a dynamic in vitro gut simulator that mimics the human gastrointestinal tract with a stabilized gut microbial community, it was shown that metformin can regulate many bacterial genes encoding metalloproteins and metal transporters189, which is possibly related to the metal-binding physicochemical properties of the drug60. Overall, these studies suggest that metformin creates an environment that favours the growth of a subset of beneficial gut bacterial species, while directly compromising the survival of potential pathogen-like bacteria to influence the physiology of the host.
Gut microbiota and host immune response
Alterations in gut microbiota are believed to play a role in meta-inflammation, notably through modulation of innate and adaptive immune systems203. Specific metabolites are generated by gut commensals, notably SCFAs. These metabolites can affect the local and systemic host immune response and contribute to impairments or improvements in metabolic homeostasis204. For instance, increases in the abundance of A. muciniphila in metformin-treated obese mice have been associated with increased activity of intestinal goblet cells and activation of adipose tissue-resident Treg cells, as well as decreased expression of the pro-inflammatory cytokines IL-1β and IL-6 in visceral adipose tissue186 (Fig. 4). Although goblet cells have been reported to induce tolerogenic dendritic cells and facilitate the development of Treg cells205, the underlying molecular mechanisms linking modulation of intestinal immune homeostasis to adipose tissue immunoregulatory responses is unclear. However, these preclinical studies suggest that metformin-induced changes in gut microbiota could increase a specific bacteria-derived immunomodulatory molecule (or molecules) that can promote regulatory immune responses both in the intestine and in peripheral metabolic organs, ultimately contributing to dampening of mucosal inflammation and/or meta-inflammation. Whether this gut–adipose tissue immunoregulatory axis also occurs in patients with T2DM treated with metformin remains to be determined.
Metformin and T1DM
Despite insulin treatment, many patients with type 1 diabetes mellitus (T1DM) do not achieve optimal glycaemic control206, which increases the risk of microvascular complications and cardiovascular diseases207. Furthermore, intensive insulin therapy increases the risk of hypoglycaemia and weight gain. Thus, several adjunctive therapies have been evaluated in combination with insulin in patients with T1DM to improve glycaemic control while decreasing the frequency of adverse events. Based on its therapeutic benefits in T2DM, metformin is thought to be a good candidate to improve blood levels of glucose and decrease insulin requirements in patients with T1DM, especially in those with overweight or obesity3,4. Metformin provides a safe oral option with a low risk of hypoglycaemia and the potential to reduce cardiovascular events3,4.
Several randomized placebo-controlled trials have evaluated the efficacy and safety of metformin therapy in T1DM, notably, the REMOVAL study, the largest and longest trial to date208. Unfortunately, as indicated by a meta-analysis published in 2018 (ref.209), these trials concluded that metformin as an adjunct to insulin therapy in patients with T1DM does not have a sustained effect on glycaemic control, as measured by HbA1c levels208,209,210. Moreover, the REMOVAL study showed that atherosclerosis progression as measured by the carotid intima–media thickness is not significantly decreased by metformin compared with placebo208. Nevertheless, metformin treatment resulted in minor benefits including decreases in insulin requirement, body weight, total cholesterol and low‐density lipoprotein cholesterol208,209. Given that metformin did not improve blood glucose control and its minor beneficial effects, the use of metformin in patients with T1DM is questionable.
Conclusions
The molecular mechanisms of metformin action continue to be a vigorous area of research 60 years after its introduction for the treatment of T2DM. Major advances have been made with respect to understanding the direct and indirect effects of the drug on metabolic tissues and the regulation of whole-body glucose homeostasis. Although it is widely accepted that the main physiological response to metformin is the inhibition of hepatic glucose production via direct action on the liver, evidence exists for indirect actions of the drug by modulation of the glucoregulatory neuronal network and/or enhancement of hepatic insulin sensitivity and meta-inflammation via effects on lipid metabolism and immune cells, respectively. The traditional liver-centred model for the glucose-lowering effect of metformin has also been challenged by the key contribution from the gut in increasing intestinal glucose utilization, stimulating GLP1 secretion and shaping the composition and function of the microbiome. At the molecular level, much work has focused on the inhibition of mitochondrial respiratory chain complex 1 and the subsequent decrease in cellular energy status and activation of AMPK. It is now clear that AMPK is dispensable for the direct inhibitory effect of metformin on gluconeogenesis, but it might play an indirect role in the long-term effects of the drug by improving hepatic insulin sensitivity.
Metformin decreases hepatic gluconeogenesis through various molecular mechanisms, including alterations in cellular energy charge, inhibition of adenylate cyclase and FBP1 secondary to increases in intracellular AMP levels mediated by mitochondria and modulation of the intracellular redox status as a consequence of direct inhibition of mG3PDH and/or mitochondria membrane depolarization. However, it remains to be demonstrated whether these signalling pathways are also relevant in the context of chronic metformin treatment and, more importantly, in patients with T2DM. The mechanisms by which metformin influences the gut microbiota are probably both direct and indirect due to its antimicrobial activities and inhibition of bile acid reabsorption and suggest the potential for the development of novel gut-targeted therapies.
There is considerable variation in the response to metformin at the individual level in terms of glycaemic response and gastrointestinal tolerance (Box 1); thus, a better understanding of the determinants underlying its clinical efficacy might help clinicians develop a personalized approach in metformin therapy. The use of modified-release or controlled-release formulations have already paved the way211,212. Besides its classic application as an anti-hyperglycaemic agent, the use of metformin has been significantly expanded with its use in the treatment of PCOS (Box 2). The long history and extensive clinical experience with metformin has also encouraged the repurposing of this drug for further therapeutic applications (Box 3), from cancer to the modulation of age-related diseases, highlighting the vast range of different possible actions of metformin. In addition, moving from the traditional oral use of metformin to a different route of administration213 or drug delivery systems (for example, microparticles or nanoparticles)214,215 might help accelerate the development of novel therapeutic applications for this multifaceted drug.
References
Davies, M. J. et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 41, 2669–2701 (2018).
Davies, M. J. et al. Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 61, 2461–2498 (2018).
United Kingdom Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352, 854–865 (1998).
United Kingdom Prospective Diabetes Study (UKPDS) Group. United Kingdom prospective diabetes study 24: a 6-year, randomized, controlled trial comparing sulfonylurea, insulin, and metformin therapy in patients with newly diagnosed type 2 diabetes that could not be controlled with diet therapy. United Kingdom Prospective Diabetes Study Group. Ann. Intern. Med. 128, 165–175 (1998).
Maruthur, N. M. et al. Diabetes medications as monotherapy or metformin-based combination therapy for type 2 diabetes: a systematic review and meta-analysis. Ann. Intern. Med. 164, 740–751 (2016).
Palmer, S. C. et al. Comparison of clinical outcomes and adverse events associated with glucose-lowering drugs in patients with type 2 diabetes: a meta-analysis. JAMA 316, 313–324 (2016).
Sanchez-Rangel, E. & Inzucchi, S. E. Metformin: clinical use in type 2 diabetes. Diabetologia 60, 1586–1593 (2017).
Howlett, H. C. & Bailey, C. J. A risk-benefit assessment of metformin in type 2 diabetes mellitus. Drug Saf. 20, 489–503 (1999).
Bailey, C. J. Metformin: historical overview. Diabetologia 60, 1566–1576 (2017).
Werner, E. & Bell, J. The preparation of methylguanidine, and of ββ-dimethylguanidine by interaction of dicyanodiamide, and methylammonium and dimethylammonium chlorides respectively. J. Am. Chem. Soc. 121, 1790–1794 (1922).
Nattrass, M. et al. Hyperlactatemia in diabetics with retinopathy during combined sulphonylurea and phenformin therapy. Diabete Metab. 4, 1–4 (1978).
Foretz, M., Guigas, B., Bertrand, L., Pollak, M. & Viollet, B. Metformin: from mechanisms of action to therapies. Cell Metab. 20, 953–966 (2014).
Hundal, R. S. et al. Mechanism by which metformin reduces glucose production in type 2 diabetes. Diabetes 49, 2063–2069 (2000).
Cusi, K., Consoli, A. & DeFronzo, R. A. Metabolic effects of metformin on glucose and lactate metabolism in noninsulin-dependent diabetes mellitus. J. Clin. Endocrinol. Metab. 81, 4059–4067 (1996).
Hother-Nielsen, O., Schmitz, O., Andersen, P. H., Beck-Nielsen, H. & Pedersen, O. Metformin improves peripheral but not hepatic insulin action in obese patients with type II diabetes. Acta Endocrinol. 120, 257–265 (1989).
Stumvoll, M., Nurjhan, N., Perriello, G., Dailey, G. & Gerich, J. E. Metabolic effects of metformin in non-insulin-dependent diabetes mellitus. N. Engl. J. Med. 333, 550–554 (1995).
DeFronzo, R. A., Barzilai, N. & Simonson, D. C. Mechanism of metformin action in obese and lean noninsulin-dependent diabetic subjects. J. Clin. Endocrinol. Metab. 73, 1294–1301 (1991).
Inzucchi, S. E. et al. Efficacy and metabolic effects of metformin and troglitazone in type II diabetes mellitus. N. Engl. J. Med. 338, 867–872 (1998).
Bailey, C. J., Mynett, K. J. & Page, T. Importance of the intestine as a site of metformin-stimulated glucose utilization. Br. J. Pharmacol. 112, 671–675 (1994).
Buse, J. B. et al. The primary glucose-lowering effect of metformin resides in the gut, not the circulation: results from short-term pharmacokinetic and 12-week dose-ranging studies. Diabetes Care 39, 198–205 (2016). This human study provides strong evidence for a gut-mediated mechanism in the blood glucose-lowering action of metformin.
McCreight, L. J., Bailey, C. J. & Pearson, E. R. Metformin and the gastrointestinal tract. Diabetologia 59, 426–435 (2016).
Forslund, K. et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 528, 262–266 (2015).
Hardie, D. G. AMPK-sensing energy while talking to other signaling pathways. Cell Metab. 20, 939–952 (2014).
Cao, J. et al. Low concentrations of metformin suppress glucose production in hepatocytes through AMP-activated protein kinase (AMPK). J. Biol. Chem. 289, 20435–20446 (2014).
Graham, G. G. et al. Clinical pharmacokinetics of metformin. Clin. Pharmacokinet. 50, 81–98 (2011).
Tucker, G. T. et al. Metformin kinetics in healthy subjects and in patients with diabetes mellitus. Br. J. Clin. Pharmacol. 12, 235–246 (1981).
Wilcock, C. & Bailey, C. J. Accumulation of metformin by tissues of the normal and diabetic mouse. Xenobiotica 24, 49–57 (1994).
Jensen, J. B. et al. [11C]-Labeled metformin distribution in the liver and small intestine using dynamic positron emission tomography in mice demonstrates tissue-specific transporter dependency. Diabetes 65, 1724–1730 (2016).
Gormsen, L. C. et al. In vivo imaging of human 11C-metformin in peripheral organs: dosimetry, biodistribution, and kinetic analyses. J. Nucl. Med. 57, 1920–1926 (2016).
Liang, X. & Giacomini, K. M. Transporters involved in metformin pharmacokinetics and treatment response. J. Pharm. Sci. 106, 2245–2250 (2017).
Todd, J. N. & Florez, J. C. An update on the pharmacogenomics of metformin: progress, problems and potential. Pharmacogenomics 15, 529–539 (2014).
Dujic, T. et al. Variants in pharmacokinetic transporters and glycemic response to metformin: a Metgen meta-analysis. Clin. Pharmacol. Ther. 101, 763–772 (2017).
Zhou, K. et al. Variation in the glucose transporter gene SLC2A2 is associated with glycemic response to metformin. Nat. Genet. 48, 1055–1059 (2016).
Ait-Omar, A. et al. GLUT2 accumulation in enterocyte apical and intracellular membranes: a study in morbidly obese human subjects and ob/ob and high fat-fed mice. Diabetes 60, 2598–2607 (2011).
Jablonski, K. A. et al. Common variants in 40 genes assessed for diabetes incidence and response to metformin and lifestyle intervention in the diabetes prevention program. Diabetes 59, 2672–2681 (2010).
Hollunger, G. Guanidines and oxidative phosphorylations. Acta Pharmacol. Toxicol. 11, 1–84 (1955).
Schafer, G. Site-specific uncoupling and inhibition of oxidative phosphorylation by biguanides. II. Biochim. Biophys. Acta 172, 334–337 (1969).
El-Mir, M. Y. et al. Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J. Biol. Chem. 275, 223–228 (2000).
Owen, M. R., Doran, E. & Halestrap, A. P. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem. J. 348, 607–614 (2000). This study together with El-Mir et al. (2000) were the first to report specific inhibition of metformin on the mitochondrial respiratory chain complex 1.
Degli Esposti, M. Inhibitors of NADH-ubiquinone reductase: an overview. Biochim. Biophys. Acta 1364, 222–235 (1998).
Fontaine, E. Metformin-induced mitochondrial complex I inhibition: facts, uncertainties, and consequences. Front. Endocrinol. 9, 753 (2018).
Vial, G., Detaille, D. & Guigas, B. Role of mitochondria in the mechanism(s) of action of metformin. Front. Endocrinol. 10, 294 (2019).
Stephenne, X. et al. Metformin activates AMP-activated protein kinase in primary human hepatocytes by decreasing cellular energy status. Diabetologia 54, 3101–3110 (2011). The is the first study to show that metformin inhibits mitochondrial respiratory chain complex 1 and activates AMPK in primary human hepatocytes.
Andrzejewski, S., Gravel, S. P., Pollak, M. & St-Pierre, J. Metformin directly acts on mitochondria to alter cellular bioenergetics. Cancer Metab. 2, 12 (2014).
Bridges, H. R., Jones, A. J., Pollak, M. N. & Hirst, J. Effects of metformin and other biguanides on oxidative phosphorylation in mitochondria. Biochem. J. 462, 475–487 (2014). This elegant study dissects the mechanism by which metformin inhibits the mitochondrial respiratory chain complex 1.
Guigas, B. et al. Metformin inhibits mitochondrial permeability transition and cell death: a pharmacological in vitro study. Biochem. J. 382, 877–884 (2004).
Wheaton, W. W. et al. Metformin inhibits mitochondrial complex I of cancer cells to reduce tumorigenesis. eLife 3, e02242 (2014).
Zannella, V. E. et al. Reprogramming metabolism with metformin improves tumor oxygenation and radiotherapy response. Clin. Cancer Res. 19, 6741–6750 (2013).
Gui, D. Y. et al. Environment dictates dependence on mitochondrial complex I for NAD+ and aspartate production and determines cancer cell sensitivity to metformin. Cell Metab. 24, 716–727 (2016).
Cheng, G. et al. Mitochondria-targeted analogues of metformin exhibit enhanced antiproliferative and radiosensitizing effects in pancreatic cancer cells. Cancer Res. 76, 3904–3915 (2016).
Boukalova, S. et al. Mitochondrial targeting of metformin enhances its activity against pancreatic cancer. Mol. Cancer Ther. 15, 2875–2886 (2016).
Guo, Z. et al. Heme binding biguanides target cytochrome P450-dependent cancer cell mitochondria. Cell Chem. Biol. 24, 1314–1275.e6 (2017).
Thakur, S. et al. Metformin targets mitochondrial glycerophosphate dehydrogenase to control rate of oxidative phosphorylation and growth of thyroid cancer in vitro and in vivo. Clin. Cancer Res. 24, 4030–4043 (2018).
Fontaine, E. Metformin and respiratory chain complex I: the last piece of the puzzle? Biochem. J. 463, e3–e5 (2014).
Wilcock, C., Wyre, N. D. & Bailey, C. J. Subcellular distribution of metformin in rat liver. J. Pharm. Pharmacol. 43, 442–444 (1991).
Detaille, D. et al. Metformin prevents high-glucose-induced endothelial cell death through a mitochondrial permeability transition-dependent process. Diabetes 54, 2179–2187 (2005). This is the first study demonstrating that metformin can reduce oxidative stress through its mitochondrial action.
Detaille, D., Guigas, B., Leverve, X., Wiernsperger, N. & Devos, P. Obligatory role of membrane events in the regulatory effect of metformin on the respiratory chain function. Biochem. Pharmacol. 63, 1259–1272 (2002).
El-Mir, M. Y. et al. Neuroprotective role of antidiabetic drug metformin against apoptotic cell death in primary cortical neurons. J. Mol. Neurosci. 34, 77–87 (2008).
Bridges, H. R., Sirvio, V. A., Agip, A. N. & Hirst, J. Molecular features of biguanides required for targeting of mitochondrial respiratory complex I and activation of AMP-kinase. BMC Biol. 14, 65 (2016).
Logie, L. et al. Cellular responses to the metal-binding properties of metformin. Diabetes 61, 1423–1433 (2012).
Hirst, J. Mitochondrial complex I. Annu. Rev. Biochem. 82, 551–575 (2013).
Zickermann, V. et al. Structural biology. Mechanistic insight from the crystal structure of mitochondrial complex I. Science 347, 44–49 (2015).
Saheki, T. et al. Citrin/mitochondrial glycerol-3-phosphate dehydrogenase double knock-out mice recapitulate features of human citrin deficiency. J. Biol. Chem. 282, 25041–25052 (2007).
Shu, Y. et al. Effect of genetic variation in the organic cation transporter 1 (OCT1) on metformin action. J. Clin. Invest. 117, 1422–1431 (2007).
He, L. & Wondisford, F. E. Metformin action: concentrations matter. Cell Metab. 21, 159–162 (2015).
Christensen, M. M. et al. The pharmacogenetics of metformin and its impact on plasma metformin steady-state levels and glycosylated hemoglobin A1c. Pharmacogenet. Genomics 21, 837–850 (2011).
Lalau, J. D., Lemaire-Hurtel, A. S. & Lacroix, C. Establishment of a database of metformin plasma concentrations and erythrocyte levels in normal and emergency situations. Clin. Drug Investig. 31, 435–438 (2011).
Foretz, M. et al. Metformin inhibits hepatic gluconeogenesis in mice independently of the LKB1/AMPK pathway via a decrease in hepatic energy state. J. Clin. Invest. 120, 2355–2369 (2010). This paper provides genetic evidence of AMPK-independent action of metformin in the inhibition of hepatic glucose production.
Fullerton, M. D. et al. Single phosphorylation sites in Acc1 and Acc2 regulate lipid homeostasis and the insulin-sensitizing effects of metformin. Nat. Med. 19, 1649–1654 (2013). This paper provides genetic evidence of AMPK-mediated phosphorylation of both ACC1 and ACC2 in metformin regulation of lipid metabolism and insulin sensitivity.
Hunter, R. W. et al. Metformin reduces liver glucose production by inhibition of fructose-1-6-bisphosphatase. Nat. Med. 24, 1395–1406 (2018). This study provides genetic evidence of metformin inhibition of hepatic glucose production through allosteric inhibition of fructose-1-6-bisphosphatase via metformin-induced increases of intracellular AMP levels.
Miller, R. A. et al. Biguanides suppress hepatic glucagon signalling by decreasing production of cyclic AMP. Nature 494, 256–260 (2013). This paper describes the suppression of hepatic glucagon signalling in response to metformin by inhibition of adenylate cyclase via metformin-induced increases of intracellular AMP levels.
Clarke, J. D. et al. Mechanism of altered metformin distribution in nonalcoholic steatohepatitis. Diabetes 64, 3305–3313 (2015).
Scheen, A. J. Clinical pharmacokinetics of metformin. Clin. Pharmacokinet. 30, 359–371 (1996).
Alshawi, A. & Agius, L. Low metformin causes a more oxidized mitochondrial NADH/NAD redox state in hepatocytes and inhibits gluconeogenesis by a redox-independent mechanism. J. Biol. Chem. 294, 2839–2853 (2019).
Madiraju, A. K. et al. Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature 510, 542–546 (2014). This paper describes a redox-dependent mechanism to account for the inhibition of hepatic gluconeogenesis by metformin.
Al-Oanzi, Z. H. et al. Opposite effects of a glucokinase activator and metformin on glucose-regulated gene expression in hepatocytes. Diabetes Obes. Metab. 19, 1078–1087 (2017).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001). This is the first paper showing AMPK activation by metformin.
Hawley, S. A. et al. Use of cells expressing gamma subunit variants to identify diverse mechanisms of AMPK activation. Cell Metab. 11, 554–565 (2010).
Zhang, C. S. et al. The lysosomal v-ATPase-ragulator complex is a common activator for AMPK and mTORC1, acting as a switch between catabolism and anabolism. Cell Metab. 20, 526–540 (2014). This study reports an AMP-independent mechanism for glucose sensing by AMPK through the lysosomal pathway.
Li, M. et al. Transient receptor potential V channels are essential for glucose sensing by aldolase and AMPK. Cell Metab. https://doi.org/10.1016/j.cmet.2019.05.018 (2019).
Zhang, C. S. et al. Metformin activates AMPK through the lysosomal pathway. Cell Metab. 24, 521–522 (2016).
Shaw, R. J. et al. The kinase LKB1 mediates glucose homeostasis in liver and therapeutic effects of metformin. Science 310, 1642–1646 (2005).
He, L. et al. Metformin and insulin suppress hepatic gluconeogenesis through phosphorylation of CREB binding protein. Cell 137, 635–646 (2009).
Lee, J. M. et al. AMPK-dependent repression of hepatic gluconeogenesis via disruption of CREB.CRTC2 complex by orphan nuclear receptor small heterodimer partner. J. Biol. Chem. 285, 32182–32191 (2010).
Caton, P. W. et al. Metformin suppresses hepatic gluconeogenesis through induction of SIRT1 and GCN5. J. Endocrinol. 205, 97–106 (2010).
Boudaba, N. et al. AMPK re-activation suppresses hepatic steatosis but its downregulation does not promote fatty liver development. EBioMedicine 28, 194–209 (2018). This paper provides genetic evidence of AMPK-dependent effects of metformin on lipogenesis inhibition and fatty acid oxidation stimulation.
Cokorinos, E. C. et al. Activation of skeletal muscle AMPK promotes glucose disposal and glucose lowering in non-human primates and mice. Cell Metab. 25, 1147–1159.e10 (2017). This study provides pharmacological and genetic evidence of the blood glucose-lowering effect of AMPK activation in skeletal muscle.
Madiraju, A. K. et al. Metformin inhibits gluconeogenesis via a redox-dependent mechanism in vivo. Nat. Med. 24, 1384–1394 (2018).
Ringler, R. L. & Singer, T. P. Studies on the mitochondrial alpha-glycerophosphate dehydrogenase. I. Reaction of the dehydrogenase with electron acceptors and the respiratory chain. J. Biol. Chem. 234, 2211–2217 (1959).
Lee, Y. P. & Lardy, H. A. Influence of thyroid hormones on L-α-glycerophosphate dehydrogenases and other dehydrogenases in various organs of the rat. J. Biol. Chem. 240, 1427–1436 (1965).
Lin, E. C. Glycerol utilization and its regulation in mammals. Annu. Rev. Biochem. 46, 765–795 (1977).
Brisson, D., Vohl, M. C., St-Pierre, J., Hudson, T. J. & Gaudet, D. Glycerol: a neglected variable in metabolic processes? Bioessays 23, 534–542 (2001).
Baur, J. A. & Birnbaum, M. J. Control of gluconeogenesis by metformin: does redox trump energy charge? Cell Metab. 20, 197–199 (2014).
Cool, B. et al. Identification and characterization of a small molecule AMPK activator that treats key components of type 2 diabetes and the metabolic syndrome. Cell Metab. 3, 403–416 (2006). This is the first paper describing a small-molecule direct AMPK activator.
Esquejo, R. M. et al. Activation of liver AMPK with PF-06409577 corrects NAFLD and lowers cholesterol in rodent and primate preclinical models. EBioMedicine 31, 122–132 (2018).
Mazza, A. et al. The role of metformin in the management of NAFLD. Exp. Diabetes Res. 2012, 716404 (2012).
Hotamisligil, G. S. Inflammation, metaflammation and immunometabolic disorders. Nature 542, 177–185 (2017).
Brestoff, J. R. & Artis, D. Immune regulation of metabolic homeostasis in health and disease. Cell 161, 146–160 (2015).
Lackey, D. E. & Olefsky, J. M. Regulation of metabolism by the innate immune system. Nat. Rev. Endocrinol. 12, 15–28 (2016).
McNelis, J. C. & Olefsky, J. M. Macrophages, immunity, and metabolic disease. Immunity 41, 36–48 (2014).
Lumeng, C. N., Bodzin, J. L. & Saltiel, A. R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117, 175–184 (2007).
Huang, W. et al. Depletion of liver Kupffer cells prevents the development of diet-induced hepatic steatosis and insulin resistance. Diabetes 59, 347–357 (2010).
Lanthier, N. et al. Kupffer cell activation is a causal factor for hepatic insulin resistance. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G107–G116 (2010).
Talukdar, S. et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat. Med. 18, 1407–1412 (2012).
Cai, D. et al. Local and systemic insulin resistance resulting from hepatic activation of IKK-β and NF-κB. Nat. Med. 11, 183–190 (2005).
Kang, K. et al. Adipocyte-derived Th2 cytokines and myeloid PPARδ regulate macrophage polarization and insulin sensitivity. Cell Metab. 7, 485–495 (2008).
Odegaard, J. I. et al. Alternative M2 activation of Kupffer cells by PPARδ ameliorates obesity-induced insulin resistance. Cell Metab. 7, 496–507 (2008).
Ricardo-Gonzalez, R. R. et al. IL-4/STAT6 immune axis regulates peripheral nutrient metabolism and insulin sensitivity. Proc. Natl Acad. Sci. USA 107, 22617–22622 (2010).
Stanya, K. J. et al. Direct control of hepatic glucose production by interleukin-13 in mice. J. Clin. Invest. 123, 261–271 (2013).
Odegaard, J. I. et al. Macrophage-specific PPARγ controls alternative activation and improves insulin resistance. Nature 447, 1116–1120 (2007).
Ilan, Y. et al. Induction of regulatory T cells decreases adipose inflammation and alleviates insulin resistance in ob/ob mice. Proc. Natl Acad. Sci. USA 107, 9765–9770 (2010).
Feuerer, M. et al. Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters. Nat. Med. 15, 930–939 (2009).
Evia-Viscarra, M. L. et al. The effects of metformin on inflammatory mediators in obese adolescents with insulin resistance: controlled randomized clinical trial. J. Pediatr. Endocrinol. Metab. 25, 41–49 (2012).
Fidan, E. et al. The effects of rosiglitazone and metformin on inflammation and endothelial dysfunction in patients with type 2 diabetes mellitus. Acta Diabetol. 48, 297–302 (2011).
Cameron, A. R. et al. Anti-inflammatory effects of metformin irrespective of diabetes status. Circ. Res. 119, 652–665 (2016). This study shows some beneficial anti-inflammatory effects of metformin in humans, irrespective of their diabetic status.
Lu, C. H., Hung, Y. J. & Hsieh, P. S. Additional effect of metformin and celecoxib against lipid dysregulation and adipose tissue inflammation in high-fat fed rats with insulin resistance and fatty liver. Eur. J. Pharmacol. 789, 60–67 (2016).
Xue, W. et al. Alkannin inhibited hepatic inflammation in diabetic Db/Db mice. Cell. Physiol. Biochem. 45, 2461–2470 (2018).
Shen, C. L. et al. Annatto-extracted tocotrienols improve glucose homeostasis and bone properties in high-fat diet-induced type 2 diabetic mice by decreasing the inflammatory response. Sci. Rep. 8, 11377 (2018).
Jing, Y. et al. Metformin improves obesity-associated inflammation by altering macrophages polarization. Mol. Cell. Endocrinol. 461, 256–264 (2018).
de Oliveira, S. et al. Metformin modulates innate immune-mediated inflammation and early progression of NAFLD-associated hepatocellular carcinoma in zebrafish. J. Hepatol. 70, 710-721(2018).
Kim, J. et al. Metformin suppresses lipopolysaccharide (LPS)-induced inflammatory response in murine macrophages via activating transcription factor-3 (ATF-3) induction. J. Biol. Chem. 289, 23246–23255 (2014).
Kelly, B., Tannahill, G. M., Murphy, M. P. & O’Neill, L. A. Metformin inhibits the production of reactive oxygen species from NADH:ubiquinone oxidoreductase to limit induction of interleukin-1β (IL-1β) and boosts interleukin-10 (IL-10) in lipopolysaccharide (LPS)-activated macrophages. J. Biol. Chem. 290, 20348–20359 (2015).
Moiseeva, O. et al. Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-κB activation. Aging Cell 12, 489–498 (2013).
Vasamsetti, S. B. et al. Metformin inhibits monocyte-to-macrophage differentiation via AMPK-mediated inhibition of STAT3 activation: potential role in atherosclerosis. Diabetes 64, 2028–2041 (2015). This study shows the inhibition of monocyte-to-macrophage differentiation and atheromatous plaque formation by metformin in ApoE −/− mice through an AMPK–STAT3-dependent mechanism.
Yan, Z. et al. Metformin suppresses UHMWPE particle-induced osteolysis in the mouse calvaria by promoting polarization of macrophages to an anti-inflammatory phenotype. Mol. Med. 24, 20 (2018).
Buldak, L. et al. Metformin affects macrophages’ phenotype and improves the activity of glutathione peroxidase, superoxide dismutase, catalase and decreases malondialdehyde concentration in a partially AMPK-independent manner in LPS-stimulated human monocytes/macrophages. Pharmacol. Rep. 66, 418–429 (2014).
Arai, M. et al. Metformin, an antidiabetic agent, suppresses the production of tumor necrosis factor and tissue factor by inhibiting early growth response factor-1 expression in human monocytes in vitro. J. Pharmacol. Exp. Ther. 334, 206–213 (2010).
Stienstra, R., Netea-Maier, R. T., Riksen, N. P., Joosten, L. A. B. & Netea, M. G. Specific and complex reprogramming of cellular metabolism in myeloid cells during innate immune responses. Cell Metab. 26, 142–156 (2017).
Geltink, R. I. K., Kyle, R. L. & Pearce, E. L. Unraveling the complex interplay between T cell metabolism and function. Annu. Rev. Immunol. 36, 461–488 (2018).
Buck, M. D., Sowell, R. T., Kaech, S. M. & Pearce, E. L. Metabolic instruction of immunity. Cell 169, 570–586 (2017).
Negrotto, L., Farez, M. F. & Correale, J. Immunologic effects of metformin and pioglitazone treatment on metabolic syndrome and multiple sclerosis. JAMA Neurol. 73, 520–528 (2016).
Lee, S. Y. et al. Metformin suppresses systemic autoimmunity in Roquin(san/san) mice through inhibiting B cell differentiation into plasma cells via regulation of AMPK/mTOR/STAT3. J. Immunol. 198, 2661–2670 (2017).
Son, H. J. et al. Metformin attenuates experimental autoimmune arthritis through reciprocal regulation of Th17/Treg balance and osteoclastogenesis. Mediators Inflamm. 2014, 973986 (2014).
Lee, S. Y. et al. Metformin ameliorates inflammatory bowel disease by suppression of the STAT3 signaling pathway and regulation of the between Th17/treg balance. PLOS ONE 10, e0135858 (2015).
Eikawa, S. et al. Immune-mediated antitumor effect by type 2 diabetes drug, metformin. Proc. Natl Acad. Sci. USA 112, 1809–1814 (2015).
Kunisada, Y. et al. Attenuation of CD4+CD25+ regulatory T cells in the tumor microenvironment by metformin, a type 2 diabetes drug. EBioMedicine 25, 154–164 (2017).
Li, L. et al. Metformin-induced reduction of CD39 and CD73 blocks myeloid-derived suppressor cell activity in patients with ovarian cancer. Cancer Res. 78, 1779–1791 (2018).
Pereira, F. V. et al. Metformin exerts antitumor activity via induction of multiple death pathways in tumor cells and activation of a protective immune response. Oncotarget 9, 25808–25825 (2018).
Natali, A. & Ferrannini, E. Effects of metformin and thiazolidinediones on suppression of hepatic glucose production and stimulation of glucose uptake in type 2 diabetes: a systematic review. Diabetologia 49, 434–441 (2006).
Wu, T., Horowitz, M. & Rayner, C. K. New insights into the anti-diabetic actions of metformin: from the liver to the gut. Expert Rev. Gastroenterol. Hepatol. 11, 157–166 (2017).
Bonora, E. et al. Lack of effect of intravenous metformin on plasma concentrations of glucose, insulin, C-peptide, glucagon and growth hormone in non-diabetic subjects. Curr. Med. Res. Opin. 9, 47–51 (1984).
Stepensky, D., Friedman, M., Raz, I. & Hoffman, A. Pharmacokinetic-pharmacodynamic analysis of the glucose-lowering effect of metformin in diabetic rats reveals first-pass pharmacodynamic effect. Drug Metab. Dispos. 30, 861–868 (2002).
Sum, C. F. et al. The effect of intravenous metformin on glucose metabolism during hyperglycaemia in type 2 diabetes. Diabet. Med. 9, 61–65 (1992).
Koffert, J. P. et al. Metformin treatment significantly enhances intestinal glucose uptake in patients with type 2 diabetes: results from a randomized clinical trial. Diabetes Res. Clin. Pract. 131, 208–216 (2017).
Wu, T. et al. Metformin reduces the rate of small intestinal glucose absorption in type 2 diabetes. Diabetes Obes. Metab. 19, 290–293 (2017).
Bailey, C. J., Wilcock, C. & Day, C. Effect of metformin on glucose metabolism in the splanchnic bed. Br. J. Pharmacol. 105, 1009–1013 (1992).
Penicaud, L., Hitier, Y., Ferre, P. & Girard, J. Hypoglycaemic effect of metformin in genetically obese (fa/fa) rats results from an increased utilization of blood glucose by intestine. Biochem. J. 262, 881–885 (1989).
Gontier, E. et al. High and typical 18F-FDG bowel uptake in patients treated with metformin. Eur. J. Nucl. Med. Mol. Imaging 35, 95–99 (2008).
Ikeda, T., Iwata, K. & Murakami, H. Inhibitory effect of metformin on intestinal glucose absorption in the perfused rat intestine. Biochem. Pharmacol. 59, 887–890 (2000).
Sakar, Y. et al. Metformin-induced regulation of the intestinal D-glucose transporters. J. Physiol. Pharmacol. 61, 301–307 (2010).
Walker, J. et al. 5-Aminoimidazole-4-carboxamide riboside (AICAR) enhances GLUT2-dependent jejunal glucose transport: a possible role for AMPK. Biochem. J. 385, 485–491 (2005).
Lenzen, S., Lortz, S. & Tiedge, M. Effect of metformin on SGLT1, GLUT2, and GLUT5 hexose transporter gene expression in small intestine from rats. Biochem. Pharmacol. 51, 893–896 (1996).
Bauer, P. V. et al. Metformin alters upper small intestinal microbiota that impact a glucose-SGLT1-sensing glucoregulatory pathway. Cell Metab. 27, 101–117.e5 (2018). This study in rodents shows the effect of metformin on upper small intestinal microbiota and glucose sensing.
Bailey, C. J., Wilcock, C. & Scarpello, J. H. Metformin and the intestine. Diabetologia 51, 1552–1553 (2008).
Duca, F. A. et al. Metformin activates a duodenal Ampk-dependent pathway to lower hepatic glucose production in rats. Nat. Med. 21, 506–511 (2015). This paper presents studies in rodent models that show that metformin lowers blood levels of glucose by inhibiting hepatic glucose production through a neuron-mediated gut–brain–liver axis.
Borg, M. J. et al. Comparative effects of proximal and distal small intestinal administration of metformin on plasma glucose and glucagon-like peptide-1, and gastric emptying after oral glucose, in type 2 diabetes. Diabetes Obes. Metab. 21, 640–647 (2019).
Henry, R. R. et al. Improved glycemic control with minimal systemic metformin exposure: effects of metformin delayed-release (metformin DR) targeting the lower bowel over 16 weeks in a randomized trial in subjects with type 2 diabetes. PLOS ONE 13, e0203946 (2018).
Oh, J. et al. Inhibition of the multidrug and toxin extrusion (MATE) transporter by pyrimethamine increases the plasma concentration of metformin but does not increase antihyperglycaemic activity in humans. Diabetes Obes. Metab. 18, 104–108 (2016).
Mannucci, E. et al. Effects of metformin on glucagon-like peptide-1 levels in obese patients with and without type 2 diabetes. Diabetes Nutr. Metab. 17, 336–342 (2004).
Napolitano, A. et al. Novel gut-based pharmacology of metformin in patients with type 2 diabetes mellitus. PLOS ONE 9, e100778 (2014).
Maida, A., Lamont, B. J., Cao, X. & Drucker, D. J. Metformin regulates the incretin receptor axis via a pathway dependent on peroxisome proliferator-activated receptor-α in mice. Diabetologia 54, 339–349 (2011).
DeFronzo, R. A. et al. Once-daily delayed-release metformin lowers plasma glucose and enhances fasting and postprandial GLP-1 and PYY: results from two randomised trials. Diabetologia 59, 1645–1654 (2016).
Bahne, E. et al. Metformin-induced glucagon-like peptide-1 secretion contributes to the actions of metformin in type 2 diabetes. JCI Insight 3, 93936 (2018). This paper presents human studies that analyse the role of GLP1 release in the glucose-lowering effect of metformin.
Li, M. et al. Efficacy and safety of liraglutide versus sitagliptin both in combination with metformin in patients with type 2 diabetes: a systematic review and meta-analysis. Medicine 96, e8161 (2017).
Bahne, E. et al. Involvement of glucagon-like peptide-1 in the glucose-lowering effect of metformin. Diabetes Obes. Metab. 18, 955–961 (2016).
Mulherin, A. J. et al. Mechanisms underlying metformin-induced secretion of glucagon-like peptide-1 from the intestinal L cell. Endocrinology 152, 4610–4619 (2011).
Kim, M. H. et al. Metformin enhances glucagon-like peptide 1 via cooperation between insulin and Wnt signaling. J. Endocrinol. 220, 117–128 (2014).
Kappe, C., Patrone, C., Holst, J. J., Zhang, Q. & Sjoholm, A. Metformin protects against lipoapoptosis and enhances GLP-1 secretion from GLP-1-producing cells. J. Gastroenterol. 48, 322–332 (2013).
Bronden, A. et al. Single-dose metformin enhances bile acid-induced glucagon-like peptide-1 secretion in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 102, 4153–4162 (2017).
Carter, D., Howlett, H. C., Wiernsperger, N. F. & Bailey, C. J. Differential effects of metformin on bile salt absorption from the jejunum and ileum. Diabetes Obes. Metab. 5, 120–125 (2003).
Lien, F. et al. Metformin interferes with bile acid homeostasis through AMPK-FXR crosstalk. J. Clin. Invest. 124, 1037–1051 (2014).
Hansen, M. et al. Effect of chenodeoxycholic acid and the bile acid sequestrant colesevelam on glucagon-like peptide-1 secretion. Diabetes Obes. Metab. 18, 571–580 (2016).
Wu, T. et al. Effects of rectal administration of taurocholic acid on glucagon-like peptide-1 and peptide YY secretion in healthy humans. Diabetes Obes. Metab. 15, 474–477 (2013).
Thomas, C. et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metab. 10, 167–177 (2009).
Trabelsi, M. S. et al. Farnesoid X receptor inhibits glucagon-like peptide-1 production by enteroendocrine L cells. Nat. Commun. 6, 7629 (2015).
Duca, F. A., Bauer, P. V., Hamr, S. C. & Lam, T. K. Glucoregulatory relevance of small intestinal nutrient sensing in physiology, bariatric surgery, and pharmacology. Cell Metab. 22, 367–380 (2015).
Waise, T. M. Z., Dranse, H. J. & Lam, T. K. T. The metabolic role of vagal afferent innervation. Nat. Rev. Gastroenterol. Hepatol. 15, 625–636 (2018).
Kuhre, R. E., Frost, C. R., Svendsen, B. & Holst, J. J. Molecular mechanisms of glucose-stimulated GLP-1 secretion from perfused rat small intestine. Diabetes 64, 370–382 (2015).
Parker, H. E. et al. Predominant role of active versus facilitative glucose transport for glucagon-like peptide-1 secretion. Diabetologia 55, 2445–2455 (2012).
Sun, L. et al. Gut microbiota and intestinal FXR mediate the clinical benefits of metformin. Nat. Med. 24, 1919–1929 (2018).
Cote, C. D. et al. Resveratrol activates duodenal Sirt1 to reverse insulin resistance in rats through a neuronal network. Nat. Med. 21, 498–505 (2015).
Waise, T. M. Z. et al. Inhibition of upper small intestinal mTOR lowers plasma glucose levels by inhibiting glucose production. Nat. Commun. 10, 714 (2019).
Karlsson, F. H. et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 498, 99–103 (2013).
Qin, J. et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 (2012).
Lee, H. & Ko, G. Effect of metformin on metabolic improvement and gut microbiota. Appl. Environ. Microbiol. 80, 5935–5943 (2014).
Shin, N. R. et al. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut 63, 727–735 (2014). These rodent studies show that metformin induces a profound shift in the faecal microbial community profile in HFD-fed mice, with higher abundance of the mucin-degrading bacterium Akkermansia accompanied by an increase in the number of mucin-producing goblet cells and adipose tissue-resident regulatory T cells.
Zhang, X. et al. Modulation of gut microbiota by berberine and metformin during the treatment of high-fat diet-induced obesity in rats. Sci. Rep. 5, 14405 (2015).
de la Cuesta-Zuluaga, J. et al. Metformin is associated with higher relative abundance of mucin-degrading Akkermansia muciniphila and several short-chain fatty acid-producing microbiota in the gut. Diabetes Care 40, 54–62 (2017). These studies in humans show that metformin shifts gut microbiota composition through enrichment of SCFA-producing and mucin-degrading bacteria.
Wu, H. et al. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat. Med. 23, 850–858 (2017). This study in humans shows that altered gut microbiota mediates some of metformin’s antidiabetic effects in treatment-naive patients with type 2 diabetes mellitus.
Tong, X. et al. Structural alteration of gut microbiota during the amelioration of human type 2 diabetes with hyperlipidemia by metformin and a traditional chinese herbal formula: a multicenter, randomized, open label clinical trial. mBio. 9, e02392-17 (2018).
Rosario, D. et al. Understanding the representative gut microbiota dysbiosis in metformin-treated type 2 diabetes patients using genome-scale metabolic modeling. Front. Physiol. 9, 775 (2018).
De Vadder, F. et al. Microbiota-generated metabolites promote metabolic benefits via gut-brain neural circuits. Cell 156, 84–96 (2014).
Gibson, G. R. & Roberfroid, M. B. Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J. Nutr. 125, 1401–1412 (1995).
Cani, P. D. et al. Gut microbiota fermentation of prebiotics increases satietogenic and incretin gut peptide production with consequences for appetite sensation and glucose response after a meal. Am. J. Clin. Nutr. 90, 1236–1243 (2009).
Chambers, E. S. et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 64, 1744–1754 (2015).
Ridlon, J. M., Kang, D. J. & Hylemon, P. B. Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 47, 241–259 (2006).
Everard, A. et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl Acad. Sci. USA 110, 9066–9071 (2013).
Plovier, H. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 23, 107–113 (2017).
Depommier, C. et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat. Med. 25, 1096–1103 (2019).
Maier, L. et al. Extensive impact of non-antibiotic drugs on human gut bacteria. Nature 555, 623–628 (2018).
Vashisht, R. & Brahmachari, S. K. Metformin as a potential combination therapy with existing front-line antibiotics for tuberculosis. J. Transl. Med. 13, 83 (2015).
Cabreiro, F. et al. Metformin retards aging in C. elegans by altering microbial folate and methionine metabolism. Cell 153, 228–239 (2013).
Belkaid, Y. & Harrison, O. J. Homeostatic immunity and the microbiota. Immunity 46, 562–576 (2017).
Postler, T. S. & Ghosh, S. Understanding the holobiont: how microbial metabolites affect human health and shape the immune system. Cell Metab. 26, 110–130 (2017).
McDole, J. R. et al. Goblet cells deliver luminal antigen to CD103+ dendritic cells in the small intestine. Nature 483, 345–349 (2012).
Weisman, A., Bai, J. W., Cardinez, M., Kramer, C. K. & Perkins, B. A. Effect of artificial pancreas systems on glycaemic control in patients with type 1 diabetes: a systematic review and meta-analysis of outpatient randomised controlled trials. Lancet Diabetes Endocrinol. 5, 501–512 (2017).
Wood, J. R. et al. Most youth with type 1 diabetes in the T1D Exchange Clinic Registry do not meet American Diabetes Association or International Society for Pediatric and Adolescent Diabetes clinical guidelines. Diabetes Care 36, 2035–2037 (2013).
Petrie, J. R. et al. Cardiovascular and metabolic effects of metformin in patients with type 1 diabetes (REMOVAL): a double-blind, randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 5, 597–609 (2017).
Meng, H. et al. Effect of metformin on glycaemic control in patients with type 1 diabetes: a meta-analysis of randomized controlled trials. Diabetes Metab. Res. Rev. 34, e2983 (2018).
Lund, S. S. et al. Effect of adjunct metformin treatment in patients with type-1 diabetes and persistent inadequate glycaemic control. A randomized study. PLOS ONE 3, e3363 (2008).
Scheen, A. J. Will delayed release metformin provide better management of diabetes type 2? Expert Opin. Pharmacother. 17, 627–630 (2016).
Fujita, Y. & Inagaki, N. Metformin: new preparations and nonglycemic benefits. Curr. Diab. Rep. 17, 5 (2017).
Berstein, L. M. Metformin: not only per os. Expert Rev. Endocrinol. Metab. 13, 63–65 (2018).
Cetin, M. & Sahin, S. Microparticulate and nanoparticulate drug delivery systems for metformin hydrochloride. Drug Deliv. 23, 2796–2805 (2016).
Zhao, Y. et al. Polymetformin combines carrier and anticancer activities for in vivo siRNA delivery. Nat. Commun. 7, 11822 (2016).
Bouchoucha, M., Uzzan, B. & Cohen, R. Metformin and digestive disorders. Diabetes Metab. 37, 90–96 (2011).
Bonnet, F. & Scheen, A. Understanding and overcoming metformin gastrointestinal intolerance. Diabetes Obes. Metab. 19, 473–481 (2017).
McCreight, L. J. et al. Pharmacokinetics of metformin in patients with gastrointestinal intolerance. Diabetes Obes. Metab. 20, 1593–1601 (2018).
Dujic, T. et al. Association of organic cation transporter 1 with intolerance to metformin in type 2 diabetes: a GoDARTS study. Diabetes 64, 1786–1793 (2015).
Dujic, T., Zhou, K., Tavendale, R., Palmer, C. N. & Pearson, E. R. Effect of serotonin transporter 5-HTTLPR polymorphism on gastrointestinal intolerance to metformin: a GoDARTS study. Diabetes Care 39, 1896–1901 (2016).
Hoffmann, I. S., Roa, M., Torrico, F. & Cubeddu, L. X. Ondansetron and metformin-induced gastrointestinal side effects. Am. J. Ther. 10, 447–451 (2003).
Scarpello, J. H., Hodgson, E. & Howlett, H. C. Effect of metformin on bile salt circulation and intestinal motility in type 2 diabetes mellitus. Diabet. Med. 15, 651–656 (1998).
Elbere, I. et al. Association of metformin administration with gut microbiome dysbiosis in healthy volunteers. PLOS ONE 13, e0204317 (2018).
Greenway, F., Wang, S. & Heiman, M. A novel cobiotic containing a prebiotic and an antioxidant augments the glucose control and gastrointestinal tolerability of metformin: a case report. Benef. Microbes 5, 29–32 (2014).
Burton, J. H. et al. Addition of a gastrointestinal microbiome modulator to metformin improves metformin tolerance and fasting glucose levels. J. Diabetes Sci. Technol. 9, 808–814 (2015).
Escobar-Morreale, H. F. Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat. Rev. Endocrinol. 14, 270–284 (2018).
Burt Solorzano, C. M. et al. Neuroendocrine dysfunction in polycystic ovary syndrome. Steroids 77, 332–337 (2012).
Ovalle, F. & Azziz, R. Insulin resistance, polycystic ovary syndrome, and type 2 diabetes mellitus. Fertil. Steril. 77, 1095–1105 (2002).
Carmina, E. & Lobo, R. A. Use of fasting blood to assess the prevalence of insulin resistance in women with polycystic ovary syndrome. Fertil. Steril. 82, 661–665 (2004).
Adashi, E. Y., Hsueh, A. J. & Yen, S. S. Insulin enhancement of luteinizing hormone and follicle-stimulating hormone release by cultured pituitary cells. Endocrinology 108, 1441–1449 (1981).
Nestler, J. E. et al. Insulin stimulates testosterone biosynthesis by human thecal cells from women with polycystic ovary syndrome by activating its own receptor and using inositolglycan mediators as the signal transduction system. J. Clin. Endocrinol. Metab. 83, 2001–2005 (1998).
Carmina, E. et al. The contributions of oestrogen and growth factors to increased adrenal androgen secretion in polycystic ovary syndrome. Hum. Reprod. 14, 307–311 (1999).
Tosi, F. et al. Insulin enhances ACTH-stimulated androgen and glucocorticoid metabolism in hyperandrogenic women. Eur. J. Endocrinol. 164, 197–203 (2011).
Wild, R. A. et al. Assessment of cardiovascular risk and prevention of cardiovascular disease in women with the polycystic ovary syndrome: a consensus statement by the Androgen Excess and Polycystic Ovary Syndrome (AE-PCOS) Society. J. Clin. Endocrinol. Metab. 95, 2038–2049 (2010).
Morin-Papunen, L. et al. Metformin reduces serum C-reactive protein levels in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 88, 4649–4654 (2003).
Tang, T. et al. Combined lifestyle modification and metformin in obese patients with polycystic ovary syndrome. A randomized, placebo-controlled, double-blind multicentre study. Hum. Reprod. 21, 80–89 (2006).
Naderpoor, N. et al. Metformin and lifestyle modification in polycystic ovary syndrome: systematic review and meta-analysis. Hum. Reprod. Update 21, 560–574 (2015).
Orio, F. Jr. et al. Improvement in endothelial structure and function after metformin treatment in young normal-weight women with polycystic ovary syndrome: results of a 6-month study. J. Clin. Endocrinol. Metab. 90, 6072–6076 (2005).
Nestler, J. E., Jakubowicz, D. J., Evans, W. S. & Pasquali, R. Effects of metformin on spontaneous and clomiphene-induced ovulation in the polycystic ovary syndrome. N. Engl. J. Med. 338, 1876–1880 (1998).
Lord, J. M., Flight, I. H. & Norman, R. J. Insulin-sensitising drugs (metformin, troglitazone, rosiglitazone, pioglitazone, D-chiro-inositol) for polycystic ovary syndrome. Cochrane Database Syst. Rev. 3, CD003053 (2003).
Kashyap, S., Wells, G. A. & Rosenwaks, Z. Insulin-sensitizing agents as primary therapy for patients with polycystic ovarian syndrome. Hum. Reprod. 19, 2474–2483 (2004).
Legro, R. S. et al. Clomiphene, metformin, or both for infertility in the polycystic ovary syndrome. N. Engl. J. Med. 356, 551–566 (2007).
Morley, L. C., Tang, T., Yasmin, E., Norman, R. J. & Balen, A. H. Insulin-sensitising drugs (metformin, rosiglitazone, pioglitazone, D-chiro-inositol) for women with polycystic ovary syndrome, oligo amenorrhoea and subfertility. Cochrane Database Syst. Rev. 11, CD003053 (2017).
Balen, A. H. et al. The management of anovulatory infertility in women with polycystic ovary syndrome: an analysis of the evidence to support the development of global WHO guidance. Hum. Reprod. Update 22, 687–708 (2016).
Heckman-Stoddard, B. M., DeCensi, A., Sahasrabuddhe, V. V. & Ford, L. G. Repurposing metformin for the prevention of cancer and cancer recurrence. Diabetologia 60, 1639–1647 (2017).
Kurelac, I. et al. Inducing cancer indolence by targeting mitochondrial complex I is potentiated by blocking macrophage-mediated adaptive responses. Nat. Commun. 10, 903 (2019).
Rotermund, C., Machetanz, G. & Fitzgerald, J. C. The therapeutic potential of metformin in neurodegenerative diseases. Front. Endocrinol. 9, 400 (2018).
Anisimov, V. N. et al. Metformin slows down aging and extends life span of female SHR mice. Cell Cycle 7, 2769–2773 (2008).
Barzilai, N., Crandall, J. P., Kritchevsky, S. B. & Espeland, M. A. Metformin as a tool to target aging. Cell Metab. 23, 1060–1065 (2016).
Justice, J. N. et al. A framework for selection of blood-based biomarkers for geroscience-guided clinical trials: report from the TAME Biomarkers Workgroup. Geroscience 40, 419–436 (2018).
Degner, N. R., Wang, J. Y., Golub, J. E. & Karakousis, P. C. Metformin use reverses the increased mortality associated with diabetes mellitus during tuberculosis treatment. Clin. Infect. Dis. 66, 198–205 (2018).
Singhal, A. et al. Metformin as adjunct antituberculosis therapy. Sci. Transl. Med. 6, 263ra159 (2014). This study provides evidence for the benefits of metformin treatment as host-adjunctive therapy for improving the effective treatment of tuberculosis.
Rangarajan, S. et al. Metformin reverses established lung fibrosis in a bleomycin model. Nat. Med. 24, 1121–1127 (2018).
Sato, N. et al. Metformin attenuates lung fibrosis development via NOX4 suppression. Respir. Res. 17, 107 (2016).
Acknowledgements
The authors acknowledge the support of grants from Inserm, CNRS, Université Paris Descartes, Agence Nationale de la Recherche (ANR), Société Francophone du Diabète (SFD), Fondation pour la Recherche Médicale (FRM), the Dutch Organization for Scientific Research (ZonMW) and DiabetesFonds.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Lactic acidosis
-
A medical condition characterized by excessively low pH in the bloodstream due to excess lactate production by glycolytic tissues, inadequate lactate utilization by gluconeogenic tissues, or varying combinations of these two processes.
- Pharmacokinetics
-
The study of the transit of a dosed drug in body fluids and tissues over time, as defined by its rate of absorption, distribution, metabolism and excretion.
- Pharmacodynamics
-
The study of the action of a drug in the body, and its biochemical and physiological effects.
- Half-maximal inhibitory concentration
-
(IC50). The concentration of an inhibitor required to decrease the response of the target by 50%.
- Pyruvate tolerance
-
A measure of glycaemic excursion in response to an intraperitoneal or intravenous injection of pyruvate, used to assess hepatic gluconeogenesis.
- Cytosolic redox potential
-
Cytoplasmic oxidation state of the cell, which is assessed by the ratio of reduced to oxidized intracellular metabolite redox couples (for example, lactate/pyruvate ratio).
- Type 2 immune cells
-
Cells involved in type 2 immune responses, such as type 2 innate lymphoid cells, eosinophils, T helper 2 cells, mast cells, basophils and alternatively-activated macrophages.
- Reverse electron transport
-
(RET). The transport of electrons from ubiquinol back to respiratory complex 1, generating a substantial amount of reactive oxygen species.
- Incretins
-
Incretins are gut hormones that are secreted after nutrient intake and stimulate glucose-stimulated insulin secretion.
- Lipoapoptosis
-
A non-canonical form of programmed cell death, which is the result of fatty acid over-accumulation that occurs in diseases associated with over-nutrition and ageing.
- Short-chain fatty acid
-
(SCFA). A fatty acid with fewer than six carbon atoms (for example, acetate, propionate and butyrate) that is the end-product of fermentation of dietary fibres by the anaerobic intestinal microbiota and acts as a signal molecule in the control of mammalian energy metabolism.
Rights and permissions
About this article
Cite this article
Foretz, M., Guigas, B. & Viollet, B. Understanding the glucoregulatory mechanisms of metformin in type 2 diabetes mellitus. Nat Rev Endocrinol 15, 569–589 (2019). https://doi.org/10.1038/s41574-019-0242-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-019-0242-2
This article is cited by
-
Silver nanoparticles in diabetes mellitus: therapeutic potential and mechanistic insights
Bulletin of the National Research Centre (2024)
-
Effects of exercise, metformin, and combination treatments on type 2 diabetic mellitus-induced muscle atrophy in db/db mice: Crosstalk between autophagy and the proteasome
Journal of Physiology and Biochemistry (2024)
-
An update on the bioactivities and health benefits of two plant-derived lignans, phyllanthin and hypophyllanthin
Advances in Traditional Medicine (2024)
-
Inflammation and immunological disarrays are associated with acute exercise in type 2 diabetes
Journal of Diabetes & Metabolic Disorders (2024)
-
Research Progress on the Use of Metformin in Leukemia Treatment
Current Treatment Options in Oncology (2024)