Abstract
Exclusion diets are becoming increasingly popular in the management of irritable bowel syndrome (IBS). Several mechanisms exist by which food items might cause gastrointestinal symptoms, such as direct osmotic effects of food in the gut lumen, changes to the gut microbiota and immune activation. These effects have been demonstrated in animal models and in human studies, particularly in the case of gluten-free diets and diets low in fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs). Indeed, randomized controlled trials (RCTs) suggest that gluten-free diets and low-FODMAP diets improve IBS symptoms, and guidelines recommend the latter approach for treating symptoms in some patients with IBS. Designing such RCTs is challenging as participants need to eat so an ‘inert’ placebo is not an option. Blinding is also an issue with these studies; in the future, new exclusion diets should not advertise what the diet consists of until it is proved to reduce symptoms. In this Review, we outline the advantages and disadvantages of each choice of control group and emphasize the importance of collecting mechanistic data (regarding direct effects of food on the gut lumen, changes in gut microbiota and intestinal inflammation) as well as symptom data in RCTs of exclusion diets in IBS.
Key points
Interest in exclusion diets to treat irritable bowel syndrome (IBS) is resurging.
Randomized controlled trials (RCTs) have shown that some diets — such as a diet low in fermentable oligosaccharides, disaccharides, monosaccharides and polyols or a gluten-free diet — might reduce IBS symptoms.
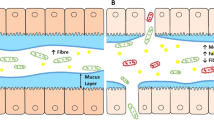
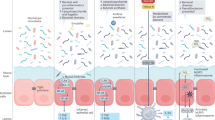
Diet might have an effect on IBS symptoms through various mechanisms, such as a direct effect of food, changing gut microbiota and immune activation.
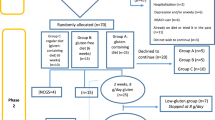
RCTs evaluating exclusion diets have unique issues relating to the choice of controls, which makes interpretation of the results challenging.
Future RCTs should collect data that provide mechanistic insights into how the diet might be exerting effects on symptoms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Skiadas, P. K. & Lascaratos, J. G. Dietetics in ancient Greek philosophy: Plato’s concepts of healthy diet. Eur. J. Clin. Nutr. 55, 532–537 (2001).
Keys, A. & Grande, F. Role of dietary fat in human nutrition. III. Diet and the epidemiology of coronary heart disease. Am. J. Public. Health Nations Health 47, 1520–1530 (1957).
Keys, A. Coronary heart disease in seven countries. Circulation 41, 186–195 (1970).
Simrén, M. et al. Food-related gastrointestinal symptoms in the irritable bowel syndrome. Digestion 63, 108–115 (2001).
Atkinson, W., Sheldon, T. A., Shaath, N. & Whorwell, P. J. Food elimination based on IgG antibodies in irritable bowel syndrome: a randomised controlled trial. Gut 53, 1459–1464 (2004).
Saito, Y. A., Locke, G. R., Weaver, A. L., Zinsmeister, A. R. & Talley, N. J. Diet and functional gastrointestinal disorders: a population-based case–control study. Am. J. Gastroenterol. 100, 2743–2748 (2005).
Verdu, E. F., Armstrong, D. & Murray, J. A. Between celiac disease and irritable bowel syndrome: the ‘no man’s land’ of gluten sensitivity. Am. J. Gastroenterol. 104, 1587–1594 (2009).
Gibson, P. R. & Shepherd, S. J. Evidence-based dietary management of functional gastrointestinal symptoms: the FODMAP approach. J. Gastroenterol. Hepatol. 25, 252–258 (2010).
Kris-Etherton, P. M. & Innis, S. Position of the American Dietetic Association and Dietitians of Canada: dietary fatty acids. J. Am. Dietetic Assoc. 107, 1599–1611 (2007).
Dehghan, M. et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): a prospective cohort study. Lancet 390, 2050–2062 (2017).
Bach Knudsen, K. E. & Hessov, I. Recovery of inulin from Jerusalem artichoke (Helianthus tuberosus) in the small intestine of man. Br. J. Nutr. 74, 101–113 (1995).
Macfarlane, G. T., Steed, H. & Macfarlane, S. Bacterial metabolism and health-related effects of galacto-oligosaccharides and other prebiotics. J. Appl. Microbiol. 104, 305–344 (2008).
Clausen, M. R., Jørgensen, J. & Mortensen, P. B. Comparison of diarrhea induced by ingestion of fructooligosaccharide Idolax and disaccharide lactulose: role of osmolarity versus fermentation of malabsorbed carbohydrate. Dig. Dis. Sci. 43, 2696–2707 (1998).
Kalantar-Zadeh, K., Berean, K. J., Burgell, R. E., Muir, J. G. & Gibson, P. R. Intestinal gases: influence on gut disorders and the role of dietary manipulations. Nat. Rev. Gastroenterol. Hepatol. 16, 733–747 (2019).
Troelsen, J. T. Adult-type hypolactasia and regulation of lactase expression. Biochim. Biophys. Acta 1723, 19–32 (2005).
Itan, Y., Jones, B. L., Ingram, C. J., Swallow, D. M. & Thomas, M. G. A worldwide correlation of lactase persistence phenotype and genotypes. BMC Evol. Biol. 10, 36 (2010).
Lomer, M. C., Parkes, G. C. & Sanderson, J. D. Review article: lactose intolerance in clinical practice — myths and realities. Aliment. Pharmacol. Ther. 27, 93–103 (2008).
Marriott, B. P., Cole, N. & Lee, E. National estimates of dietary fructose intake increased from 1977 to 2004 in the United States. J. Nutr. 139, 1228S–1235S (2009).
Jones, H. F., Butler, R. N. & Brooks, D. A. Intestinal fructose transport and malabsorption in humans. Am. J. Physiol. Gastrointest. Liver Physiol 300, G202–G206 (2011).
Yao, C. K. et al. Dietary sorbitol and mannitol: food content and distinct absorption patterns between healthy individuals and patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 27, 263–275 (2014).
Hyams, J. S. Sorbitol intolerance: an unappreciated cause of functional gastrointestinal complaints. Gastroenterology 84, 30–33 (1983).
Ong, D. K. et al. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J. Gastroenterol. Hepatol. 25, (1366–1373) (2010).
Barrett, J. S. et al. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates. Aliment. Pharmacol. Ther. 31, 874–882 (2010).
Murray, K. et al. Differential effects of FODMAPs (fermentable oligo, di-, mono-saccharides and polyols) on small and large intestinal contents in healthy subjects shown by MRI. Am. J. Gastroenterol. 109, 110–119 (2014).
Major, G. et al. Colon hypersensitivity to distension, rather than excessive gas production, produces carbohydrate-related symptoms in individuals with irritable bowel syndrome. Gastroenterology 152, 124–133.e2 (2017).
Simrén, M. et al. Visceral hypersensitivity is associated with GI symptom severity in functional GI disorders: consistent findings from five different patient cohorts. Gut. 67, 255–262 (2018).
Huaman, J. W. et al. Effects of prebiotics vs a diet low in FODMAPs in patients with functional gut disorders. Gastroenterology 155, 1004–1007 (2018).
Wilkinson-Smith, V. C. et al. Insights into the different effects of food on intestinal secretion using magnetic resonance imaging. J. Parenter. Enter. Nutr. 42, 1342–1348 (2018).
Pittayanon, R. et al. Gut microbiota in patients with irritable bowel syndrome — a systematic review. Gastroenterology 157, 97–108 (2019).
Halmos, E. P. et al. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 64, 93–100 (2015).
Hustoft, T. N. et al. Effects of varying dietary content of fermentable short-chain carbohydrates on symptoms, fecal microenvironment, and cytokine profiles in patients with irritable bowel syndrome. Neurogastroenterol. Motil. 29, e12969 (2017).
Staudacher, H. M. et al. A diet low in FODMAPs reduces symptoms in patients with irritable bowel syndrome and a probiotic restores Bifidobacterium species: a randomized controlled trial. Gastroenterology 153, 936–947 (2017).
McIntosh, K. et al. FODMAPs alter symptoms and the metabolome of patients with IBS: a randomised controlled trial. Gut. 66, 1241–1251 (2017).
Sloan, T. J. et al. A low FODMAP diet is associated with changes in the microbiota and reduction in breath hydrogen but not colonic volume in healthy subjects. PLoS One 26, e0201410 (2018).
Chumpitazi, B. P. et al. Randomised clinical trial: gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment. Pharmacol. Ther. 42, 418–427 (2015).
Bennet, S. M. P. et al. Multivariate modelling of faecal bacterial profiles of patients with IBS predicts responsiveness to a diet low in FODMAPs. Gut 67, 872–881 (2018).
Shi-Yi Zhou et al. FODMAP diet modulates visceral nociception by lipopolysaccharide-mediated intestinal inflammation and barrier dysfunction. J. Clin. Invest. 128, 267–280 (2018).
Waserman, S., Begin, P. & Watson, W. IgE-mediated food allergy. Allergy Asthma Clin. Immunology 14, 55 (2018).
Soares-Weiser, K. et al. The diagnosis of food allergy: a systematic review and meta-analysis. Allergy 69, 76–86 (2014).
Fritscher-Ravens, A. et al. Many patients with irritable bowel syndrome have atypical food allergies not associated with immunoglobulin E. Gastroenterology 157, 109–118 (2019).
Volta, U. et al. Dietary triggers in irritable bowel syndrome: is there a role for gluten? J. Neurogastroenterol. Motil. 22, 547–557 (2016).
Vazquez-Roque, M. I. et al. Association of HLA-DQ gene with bowel transit, barrier function, and inflammation in irritable bowel syndrome with diarrhea. Am. J. Physiol. Gastrointest. Liver Physiol 303, G1262–G1269 (2012).
Vazquez-Roque, M. I. et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhoea: effects on bowel frequency and intestinal function. Gastroenterology 144, 903–911 (2013).
Wahnschaffe, U. et al. Predictors of clinical response to gluten-free diet in patients diagnosed with diarrhoea-predominant irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 5, 844–850 (2007).
Verdu, E. F. et al. Gliadin-dependent neuromuscular and epithelial secretory responses in gluten-sensitive HLA-DQ8 transgenic mice. Am. J. Physiol. Gastrointest. Liver Physiol 294, G217–G225 (2008).
Chirdo F. G., Arranz E. in Advances in the Understanding of Gluten Related Pathology and the Evolution of Gluten-free Foods (eds Arranz E., Fernández-Bañares F., Rosell C. M., Rodrigo L., Peña A. S.) 141–162 (OmniaScience, 2015).
Zevallos, V. F. et al. Nutritional wheat amylase-trypsin inhibitors promote intestinal inflammation via activation of myeloid cells. Gastroenterology 52, 1100–1113 (2017).
Caminero, A. et al. Duodenal bacterial proteolytic activity determines sensitivity to dietary antigen through protease-activated receptor-2. Nat. Commun. 10, 1198 (2019).
Caminero, A. et al. Lactobacilli degrade wheat amylase trypsin inhibitors to reduce intestinal dysfunction induced by immunogenic wheat proteins. Gastroenterology 156, 2266–2280 (2019).
Spieth, P. M. et al. Randomized controlled trials — a matter of design. Neuropsychiatr. Dis. Treat. 12, 1341–1349 (2016).
Lovell, R. M. & Ford, A. C. Global prevalence of and risk factors for irritable bowel syndrome: a meta-analysis. Clin. Gastroenterol. Hepatol. 10, 712–721 (2012).
Sturdevant, S. G. & Lumley, T. Testing carryover effects after cessation of treatments: a design approach. BMC Med. Res. Methodol. 16, 92 (2016).
Hopewell, S., Dutton, S., Yu, L. M., Chan, A. W. & Altman, D. G. The quality of reports of randomised trials in 2000 and 2006: comparative study of articles indexed in PubMed. BMJ 340, c723 (2010).
Schulz, K. F. & Altman, D. G., Moher D for the CONSORT Group. CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. BMJ 340, c332 (2010).
Staudacher, H. M. et al. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J. Nutr. 142, 1510–1518 (2012).
Ford, A. C. & Moayyedi, P. Meta-analysis: factors affecting placebo response rate in irritable bowel syndrome. Aliment. Pharmacol. Ther. 32, 144–158 (2010).
Dionne, J. et al. A systematic review and meta-analysis evaluating the efficacy of a gluten-free diet and a low FODMAPs diet in treating symptoms of irritable bowel syndrome. Am. J. Gastroenterol. 113, 1290–1300 (2018).
Bohn, L. et al. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: a randomized controlled trial. Gastroenterology 149, 1399–1407 (2015).
National Institute for Health and Care Excellence. NICE Guidelines: Irritable Bowel Syndrome in Adults: Diagnosis and Management of Irritable Bowel Syndrome in Primary Care https://www.nice.org.uk/guidance/cg61 (NICE, 2017).
Ford, A. C., Harris, L. A., Lacy, B. E., Quigley, E. M. M. & Moayyedi, P. Systematic review with meta-analysis: the efficacy of prebiotics, probiotics, synbiotics and antibiotics in irritable bowel syndrome. Aliment. Pharmacol. Ther. 48, 1044–1060 (2018).
Kaptchuk, T. J. et al. Components of placebo effect: randomised controlled trial in patients with irritable bowel syndrome. BMJ 336, 999–1003 (2008).
Buhner, S. et al. Activation of human enteric neurons by supernatants of colonic biopsy specimens from patients with irritable bowel syndrome. Gastroenterology 137, 1425–1434 (2009).
Biesiekierski, J. R. et al. Gluten causes of gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo controlled trial. Am. J. Gastroenterol. 106, 508–514 (2011).
Shahhbazkhani, B. et al. Non-celiac gluten sensitivity has narrowed the spectrum of irritable bowel syndrome: a double-blind randomized placebo controlled trial. Nutrients 7, 4542–4554 (2015).
Hustoft, T. N. et al. Effects of varying dietary content of fermentable short-chain carbohydrates on symptoms, fecal microenvironment and cytokine profiles in patients with irritable bowel syndrome. Neurogastroenterol. Motil. 29, e12969 (2017).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Corring, T. The adaptation of digestive enzymes to the diet: its physiological significance. Reprod. Nutr. Dev. 20, 1217–1235 (1980).
Ford, A. C. et al. American College of Gastroenterology Monograph on Management of Irritable Bowel Syndrome. Am. J. Gastroenterol. 113, 1–18 (2018).
Moayyedi, P. et al. Canadian Association of Gastroenterology Clinical Practice Guideline for the Management of Irritable Bowel Syndrome (IBS). J. Can. Assoc. Gastroenterol. 2, 6029 (2019).
Moayyedi, P. et al. Irritable bowel syndrome diagnosis and management: a simplified algorithm for clinical practice. United European Gastroenterol. J. 5, 773–788 (2017).
Biesiekierski, J. R. et al. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 145, 320–328 (2013).
Hungin, A. P., Becher, A. & Cayley, B. Irritable bowel syndrome: an integrated explanatory model for clinical practice. Neurogastroenterol. Motil. 27, 750–763 (2015).
O’Connor, D. B., Jones, F., Connor, M., McMillan, B. & Ferguson, E. Effects of daily hassles and eating style on eating behavior. J. Health Psychol. 27, 20–31 (2008).
Werlang, M. E., Palmer, W. C. & Lacy, B. E. Irritable bowel syndrome and dietary interventions. Gastroenterol. Hepatol. 15, 16–26 (2019).
Caminero, A., Meisel, M., Jabri, B. & Verdu, E. F. Mechanisms by which gut microorganisms influence food sensitivities. Nat. Rev. Gastroenterol. Hepatol. 16, 7–18 (2019).
Acknowledgements
The work of P.M. and P.B. is funded by Canadian Institutes of Health Research (CIHR) (RN279389-358033). The Inflammation, Microbiome, and Alimentation: Gastro-Intestinal and Neuropsychiatric Effects: the IMAGINE network — a Strategy for Patient-Oriented Research CIHR Chronic Disease Network.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks Nick Talley and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Moayyedi, P., Simrén, M. & Bercik, P. Evidence-based and mechanistic insights into exclusion diets for IBS. Nat Rev Gastroenterol Hepatol 17, 406–413 (2020). https://doi.org/10.1038/s41575-020-0270-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-020-0270-3
This article is cited by
-
Healthcare Utilization Patterns: Irritable Bowel Syndrome, Inflammatory Bowel Disease, and Gastroesophageal Reflux Disease
Digestive Diseases and Sciences (2024)
-
Irritable bowel syndrome and microbiome; Switching from conventional diagnosis and therapies to personalized interventions
Journal of Translational Medicine (2022)
-
Genetics of irritable bowel syndrome: shifting gear via biobank-scale studies
Nature Reviews Gastroenterology & Hepatology (2022)
-
Food for thought about the immune drivers of gut pain
Nature (2021)
-
Leveraging diet to engineer the gut microbiome
Nature Reviews Gastroenterology & Hepatology (2021)