Abstract
Tertiary lymphoid structures (TLSs) are ectopic lymphoid tissues that drive antigen-specific immune responses at sites of chronic inflammation. Unlike secondary lymphoid organs such as lymph nodes, TLSs lack capsules and have their own unique characteristics and functions. The presumed influence of TLSs on the disease course has led to widespread interest in obtaining a better understanding of their biology and function. Studies using single-cell analyses have suggested heterogeneity in TLS composition and phenotype, and consequently, functional correlates with disease progression are sometimes conflicting. The presence of TLSs correlates with a favourable disease course in cancer and infection. Conversely, in autoimmune diseases and chronic age-related inflammatory diseases including chronic kidney disease, the presence of TLSs is associated with a more severe disease course. However, the detailed mechanisms that underlie these clinical associations are not fully understood. To what extent the mechanisms of TLS development and maturation are shared across organs and diseases is also still obscure. Improved understanding of TLS development and function at the cellular and molecular levels may enable the exploitation of these structures to improve therapies for chronic diseases, including chronic kidney disease.
Key points
-
Tertiary lymphoid structures (TLSs) are organized lymphoid aggregates that develop in perivascular areas in response to disturbed tissue homeostasis.
-
TLSs serve as local immune niches to promote adaptive immunity; their unencapsulated structure enables direct exposure to diverse stimuli from an inflamed environment.
-
The development of TLSs in different organs involves common mechanisms that are presumably regulated by tissue-specific cues.
-
The presence of TLSs correlates with a favourable disease course in many types of cancer and infection.
-
In autoimmunity, chronic inflammation and ageing, the presence of TLSs correlates with pathological conditions and a more severe disease course.
-
Functional characterization of TLSs in human diseases and the development of interventions to induce or reduce TLSs could lead to promising therapeutic avenues.
Similar content being viewed by others
Introduction
The prevalence of age-related chronic diseases, including cardiovascular disease, cancer, infections and chronic kidney disease (CKD) is increasing owing to the increase in mean global life expectancy1. Age-related diseases are characterized by chronic inflammation, which is a key contributor to the pathogenesis of these diseases and an important therapeutic target1. Inflammation is a protective response that can restore tissue homeostasis following tissue injury or infection; however, sustained or unresolved inflammation has detrimental effects that can contribute to the development and progression of chronic diseases.
Tertiary lymphoid structures (TLSs) (also known as tertiary lymphoid tissues, tertiary lymphoid organs or ectopic lymphoid tissues) develop in non-lymphoid organs during chronic inflammatory conditions, including cancer, infection, autoimmunity and age-related diseases2,3,4,5. TLSs are organized lymphoid aggregates with a network of specialized fibroblasts that share many functional and structural characteristics with secondary lymphoid organs (SLOs), particularly lymph nodes. For example, TLSs and SLOs can drive antigen-specific immune responses5 and are equipped with specialized blood vessels known as high endothelial venules (HEVs) that facilitate transmigration of lymphocytes from the blood into lymphoid tissues6. However, TLSs are less organized than SLOs and develop in response to cues that are associated with disturbed homeostasis. For example, tissue-resident cells can transdifferentiate into TLS cellular components in response to microenvironmental stimuli during tissue inflammation7. The advent of single-cell sequencing techniques has led to new insights into cellular diversity and molecular heterogeneity during the various stages of TLS development.
Interest in TLSs is increasing because of their presence in and presumed contribution to chronic inflammatory conditions and ageing8,9,10,11,12,13. The role of TLSs in these diseases is context-dependent and can be beneficial or detrimental. In cancer, for example, antitumour immune responses are generated within TLSs that are located near to tumours14,15 and the presence of TLSs is associated with improved response to therapies in some, but not all, types of cancer16. Similarly, TLSs induced by infections generate anti-pathogen immune responses that are beneficial for the host17,18. Conversely, in autoimmunity, TLSs promote activation of autoreactive lymphocytes, resulting in autoantibody production, and the presence of TLSs is associated with a poor prognosis19. The development of TLSs also contributes to the progression of kidney diseases such as acute kidney injury (AKI) in elderly people13,20. Age-dependent TLS formation has been reported in the kidney, lung and other organs and has a role in age-related chronic inflammatory diseases7,8,13,21,22,23. These findings suggest that TLSs are important sites for modulating local immunity and are of critical importance when considering therapeutic strategies to prevent the progression of chronic diseases.
In this Review, we discuss TLS development, maintenance and function, with a focus on the roles of TLSs in autoimmune disease, cancer, infection and chronic age-related inflammatory diseases, including CKD. We also discuss the clinical implications and therapeutic potential of TLSs in various tissues and diseases.
Characteristics of TLSs and SLOs
TLSs and SLOs are mainly composed of lymphocytes with the support of specialized fibroblasts and are associated with HEVs. Molecules that are essential for SLO maintenance and function, including homeostatic chemokines and lymphotoxin, are also detected in TLSs5. In addition, both structures promote induction of adaptive immune responses and can contain germinal centres that are essential for cognate T cell–B cell interactions, consecutive antibody somatic hypermutation and affinity maturation3,24. Activation of the adaptive immune responses distinguishes TLSs from simple inflammatory cell infiltrates.
TLSs also have some unique characteristics and functions, particularly with respect to local tissue responses (Fig. 1, Table 1). The homeostatic chemokines CXCL13, CCL19 and CCL21 have crucial roles in the development and maintenance of SLOs, but CCL21 is not involved in age-dependent TLS formation in the kidney, bladder, or liver7,22,23. Development of TLSs is induced after birth (discussed further below) and depends on CD4 T cells or other types of immune cells7,25,26, whereas SLO development depends on specialized embryonic lymphoid tissue inducer (LTi) cells that mediate the transition of embryonic mesenchymal lymphoid tissue organizer cells into CXCL13-producing follicular dendritic cells (FDCs) and CCL19/CCL21-producing fibroblastic reticular cells in a lymphoid tissue-dependent manner5. LTi cells promote lymphoid organogenesis via lymphotoxin and tumour necrosis factor (TNF)27 and FDCs are specialized mesenchymal cells that support B cell activation28. After birth, other cell types such as T helper 17 (Th17) cells or B cells take over the function of LTi cells, and parenchymal and mesenchymal cells become lymphoid tissue organizer cells in adult organs during lymphoid neogenesis25,26,29,30.
a, In lymph nodes, immune cell subpopulations are distributed in a regular pattern and form sub-compartments. B cells form B cell follicles and occasionally develop germinal centres during antigen stimulation, whereas most T cells reside in the T cell area adjacent to B cell follicles. A fibrous capsule surrounds these distinct cellular compartments. b, In tertiary lymphoid structures (TLSs), T cells and B cells are mostly intermingled with each other but occasionally form distinct B cell and T cell compartments similar to those that are present in lymph nodes. TLSs lack a capsule and are exposed to local antigens, danger-associated molecular patterns, cytokines and various ions. HEV, high endothelial venule.
In SLOs, autoreactive B cells are eliminated via a process that depends on competition between cells with different specificities31. A key feature of this censoring mechanism is the selective exclusion of self-antigen-binding B cells from the normal migration route into B cell follicles, which leads to their premature death (B cells that are excluded from follicles have a half-life of less than a day)31,32. By contrast, autoreactive B cells can enter the follicular niche, receive survival factors and survive in TLSs33. In autoimmunity, disease-specific autoantibodies are produced in TLSs in target organs34,35. Autoantibody production in TLSs has also been reported in aged, injured kidneys20.
SLOs are encapsulated and thereby physically separated from the inflammatory environment, whereas TLSs are directly exposed to antigens, cytokines, damage-associated molecular patterns (DAMPs) and other inflammatory factors6,24. This difference makes the immune response that is generated in TLSs more effective than that generated in SLOs. For example, TLSs in the lungs, termed inducible bronchus-associated lymphoid tissue (iBALT), develop in response to infection and contribute to protective immunity, even in the absence of SLOs36. Germinal centres in iBALT comprise plasma cells and memory B cells with greater cross-protective potential than those in the lymph nodes and spleen18. B cells in iBALT also proliferate more upon antigen encounter than those in lymph nodes or the spleen18.
Heterogeneity and development of TLSs
TLSs vary in their cellular composition from loose T cell–B cell clusters to highly organized structures with distinct T cell zones and B cell areas that harbour germinal centres. TLS development is thought to be a gradual process, but detailed analysis of TLS phenotypes and the clinical relevance of TLS maturation stages is challenging. Tissue samples from patients with chronic inflammatory diseases are limited and the time of disease initiation in these samples is unknown, so establishing a correlation between TLS status and disease progression is difficult. In addition, methods for qualitative and quantitative TLS evaluation are not standardized, which might partly explain conflicting results regarding the clinical relevance of TLSs in the kidney and other organs37,38,39,40.
Stages of maturation
To stratify the definition of TLSs, we analysed surgically resected kidney samples from patients with complicated pyelonephritis and from older patients (≥60 years) with renal cell carcinoma (RCC) as well as aged mouse kidneys with ischaemia–reperfusion injury-induced TLSs21. We defined TLSs as clusters of lymphocytes with signs of proliferation and showed that TLS development in human and murine kidneys occurs through distinct stages21 (Fig. 2a). Similar TLS maturation stages have been found in patients with various types of cancer and in mouse lungs after experimental TLS induction41,42. These data suggest that a common developmental sequence mediates lymphoid neogenesis across different organs.
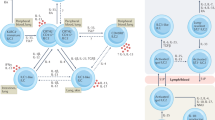
a, In the kidney and most other organs, arteries, veins and lymphatic vessels run together. Tertiary lymphoid structures (TLSs) first appear as small aggregates containing mainly T cells and B cells at perivascular sites, and then expand and mature into clusters with distinct B cell and T cell areas. In immature TLSs, antigen presentation and intimate T cell–B cell interactions drive activation of lymphocytes and TLS maturation. Mature TLSs contain high endothelial venules (HEVs), which are specialized blood vessels that are adapted for lymphocyte trafficking. Some of the B cell areas (follicles) contain germinal centres, which are histologically defined as clusters of proliferating B cells. The germinal centres support B cell affinity maturation, class switching and somatic hypermutation. b, In parallel to TLS maturation, resident fibroblasts transdifferentiate into several distinct phenotypes and orchestrate TLS development. First, in response to IL-17, IL-22 and retinoic acid, perivascular fibroblasts acquire the ability to produce homeostatic chemokines such as CXC-chemokine ligand 13 (CXCL13) and CC-chemokine ligand 19 (CCL19) and recruit immune cells. These immunofibroblasts then further transdifferentiate into follicular dendritic cells (FDCs) and follicular reticular cells (FRCs), which form and support the B cell and T cell areas, respectively. Lymphotoxin (LT) and tumour necrosis factor (TNF) promote the transdifferentiation of immunofibroblasts into FDCs50, but the signalling pathways that drive FRC differentiation are unknown. c, Within TLSs, dendritic cells and B cells may present local antigens to T cells, leading to T cell activation and proliferation. d, CD4 T cells with B cell helper functions, T follicular helper-like cells (Tfh-like cells) and T peripheral helper (Tph) cells187, interact with B cells in TLSs via several co-stimulation molecules, including ICOS–ICOSL, CD154–CD40 and CD153–CD30, in synergy with cytokines such as IL-21 and IL-10 (ref. 13). These interactions promote B cell transdifferentiation into germinal centre B cells.
TLS can be induced in an antigen-dependent or antigen-independent manner (Box 1). They initially appear as small aggregates containing mainly T cells and B cells and then expand and mature into clusters with B cell areas supported by CD21+ FDCs with or without distinct T cell areas13,21,41. Finally, a germinal centre reaction can be activated in the B cell areas of TLSs. We found that TLS development arrests before germinal centre development in mildly injured kidneys, whereas TLSs in severely injured kidneys are fully mature. This finding suggests that TLS maturation is associated with the severity of inflammation, kidney injury and kidney dysfunction21.
A single-cell RNA sequencing analysis of B cells derived from human tumours identified unswitched IgD+ B cells, switched IgG+ B cells and a wide range of B cells that expressed genes that are associated with immature or mature germinal centres11,12. Analysis of B cell receptor sequences from the B cells further confirmed their clonal expansion11. These data support in situ B cell proliferation and maturation within TLSs in human cancer tissues.
Consistent with the observation that TLSs disappear when inflammation is resolved17,43, TLS maturation can be reversed with immunosuppressive treatments21, including corticosteroids28,41. In a retrospective study of kidney biopsy samples from clinically stable transplanted renal allografts, we found TLS s in 50% of the allografts 1 month after transplantation. Mature TLS, defined as those with FDCs, were present in 19% of the allografts 1 year after transplantation. The presence of mature TLSs, but not immature TLS, in 1-year biopsy samples was associated with poor graft function44. These results suggest that classification of TLSs into distinct maturation stages might improve their prognostic power in inflammatory kidney diseases, including kidney transplant rejection.
Involvement of perivascular cells
In most organs, TLSs develop in perivascular areas21,29,45,46, which are rich in extracellular matrix components, small blood vessels, lymphatic vessels and neurons. Perivascular areas are conserved across organs and can act as a niche for tissue-resident immune cells47,48. Several types of unique cells that are involved in TLS formation and maintenance reside in the perivascular area.
Perivascular fibroblasts have a pivotal role in TLS formation7,49,50 (Fig. 2b). These cells transdifferentiate into specialized fibroblasts that produce homeostatic chemokines and provide cues for optimal spatial immune cell organization7,9,51,52,53,54. The molecular cues that induce this phenotypic change are context dependent. For example, in lung fibroblasts, CXCL13 expression was induced by type I interferon (IFN) signalling52, IL-17A, or TNF25, whereas in salivary gland fibroblasts CXCL13 expression was induced by IL-22 (ref. 55).
The anatomical relationship between TLSs and the vasculature suggests a functional connection between blood vessels and lymphoid neogenesis. In the kidney, arteries, veins and lymphatic vessels run in parallel and TLSs are detected in perivascular, periglomerular, and subcapsular areas and in the renal pelvis, all of which contain arterial circuits21,51,56. Loss of Notch signalling in vascular endothelial cells, but not in lymphatic vessel endothelial cells, led to TLS formation in mouse kidneys57. In this model, arteries located within TLSs acquire the HEV phenotype and promote lymphocyte recruitment.
Lymphatic vessels might also have an important role in TLS induction and maintenance. These vessels spread along the arteries and veins within organs and form a dense network throughout the body6,58. They serve as conduits for various immune cells, including lymphocytes and dendritic cells, to traffic from peripheral non-lymphoid organs to SLOs59. In a model of chronic ileitis caused by TNF overabundance, TLS develop in the mesentery at the sites of lymphatic valves60. Lymphatic vessels, particularly lymphatic endothelial cells, produce IL-7, which is a non-redundant regulator of T cell homeostasis in SLOs and TLSs61,62 and contributes to TLS maintenance. Genetic deletion or pharmacological neutralization of IL-7-producing lymphatic endothelial cells resulted in defective TLS formation and a decrease in the number of CD4+ tissue-resident memory T (TRM) cells in the mouse lung61,63. Thus, interaction between immune cells and non-immune cells in the perivascular area is vital for TLS development across organs including the kidney and lung.
TLSs in pathological conditions
The clinical relevance of TLSs in diseases including autoimmune conditions, cancer, infection, chronic inflammatory diseases and in ageing is context dependent. Even in the same organ, TLSs can be beneficial or detrimental depending on their composition and the aetiology and phase of the disease.
Autoimmune diseases
The formation of TLSs has been reported in almost all organ-specific human autoimmune diseases, including rheumatoid arthritis64, lupus nephritis65, type 1 diabetes mellitus66, Crohn disease67 and Sjogren syndrome33. The reported prevalence of TLSs in these diseases ranges from almost 100% in patients with Hashimoto thyroiditis68 to almost 20% in patients with juvenile dermatomyositis69. Most, but not all, clinical studies have shown a direct correlation between the presence of TLS and disease activity34. The reasons for this inconsistency are unknown but may include differences in TLS definition, disease stage and treatment history as well as sampling techniques (for example, biopsy or surgical resection) and errors.
TLSs may promote breaking of self-tolerance in autoimmune disease. This hypothesis is exemplified by the finding that transplantation of tissue containing TLSs from patients with rheumatoid arthritis and myasthenia gravis into immunodeficient mice resulted in sustained production of anti-cyclic citrullinated peptide (CCP) antibodies and anti-acetylcholine receptor antibodies19,70. The fact that TLSs can also be detected in seronegative patients with arthritis, such as those with spondyloarthropathies71,72, suggests that TLS can contribute to autoimmune disease pathogenesis via multiple mechanisms.
Lupus nephritis affects 30–60% of patients with SLE and is one of the most serious complications of this disease73. Although lupus nephritis is characterized by heterogeneous glomerulonephritis, tubulointerstitial inflammation is common and determines the kidney prognosis74. Several studies have reported TLS formation in lupus nephritis65,75. An analysis of micro-dissected TLSs from patient kidney biopsy samples detected clonal B cells with a high degree of somatic hypermutation65. These findings indicate in situ germinal centre responses and the generation of autoantibodies, which could potentially contribute to kidney disease progression by binding proximal tubular cells and stimulating cytokine production76. The presence of follicular T cell-like CD4 T cells in kidney biopsy samples was associated with lower estimated glomerular filtration rate in patients with lupus nephritis75, suggesting a pathogenic contribution of TLSs in this disease77.
Interestingly, TLSs are also detected in the central nervous system in patients with SLE78. Similarly, in patients with rheumatoid arthritis, TLSs are detected not only in synovial tissues, but also in lung79 and bone marrow80. These findings suggest the involvement of circulating cells or humoral factors in TLS development in autoimmune diseases.
Cancer
Inflammation and cancer are often accompanied by TLSs81 but their density varies between tumour types and between individuals with the same disease (Fig. 3; Supplementary Table 1). The reasons for this heterogeneity are unclear but tumour-intrinsic molecular features may contribute. For example, higher mutational burden is associated with higher TLS density in colorectal82,83,84, bladder85 and pancreatic cancers86. HER2 loss and hormone receptor-positivity in breast cancer87,88,89, and ALK rearrangements in non-small-cell lung cancer90, are associated with reduced TLS density, whereas the expression of viral antigens in head and neck squamous cell carcinoma shows conflicting data concerning correlation with TLS density91,92. A positive correlation between TLSs and the survival of patients with bladder cancer was first reported in 1970 (ref. 93) and similar findings have been reported for many other cancer types42. The presence of TLSs in the tumour microenvironment correlates with increased infiltration of adaptive immune cells42, and evidence suggests that B and T cells in TLSs are tumour specific15,94,95,96,97,98,99. T cell priming may take place in TLSs independently of lymph nodes, suggesting a unique local function for TLSs in the tumour microenvironment.
The proportion of tertiary lymphoid structure (TLS)-positive tumours was extracted from 57 published studies reporting histological assessment of TLSs in most common tumour types and was compared across organs by one-way ANOVA test. As TLSs mainly develop in the tumour periphery, studies that only performed TLS analysis in intratumoural areas were excluded. Treated cohorts were also excluded because neoadjuvant therapy impairs TLS development. In total, 72 cohorts were selected for analysis (see Supplementary Table 1 for details and references). Each independent cohort is represented by a separate symbol. Diverse patient characteristics were plotted as distinct cohorts where those data were provided. TLS development differs significantly between organs as well as within each organ, which may be due to differences between molecular tumour subtypes, as well as study-specific histological definitions of TLS (ranging from lymphocytic aggregates of any maturation stage to clusters with specific composition containing high endothelial venules, mature dendritic cells or organized B and T cell compartments). The proportion of TLS-positive tumours detected by H&E or immunostaining is similar, suggesting that both methods of histological assessment are reliable. BC, breast cancer; CRC, colorectal cancer; DCIS, ductal carcinoma in situ; GACA, gastric cancer; H&E, haematoxylin and eosin; HCC, hepatocellular carcinoma; HNSCC, head and neck squamous cell carcinoma; HPV, human papilloma virus; HR, hormone receptor; IBC, invasive breast cancer (cohorts containing different molecular subtypes of breast cancer); MIBC, muscle invasive bladder cancer; NonMIBC, non-muscle invasive bladder cancer; NSCLC, non-small-cell lung cancer; OVCA, ovarian cancer; PDAC, pancreatic ductal adenocarcinoma; RCC, renal cell carcinoma; TMBhi, tumour mutational burden high (microsatellite instable CRC, or tumours with a mutational burden greater than the cohort median in MIBC and PDAC); TMBlo, tumour mutational burden low (microsatellite stable CRC or tumours with a mutation load equal to or below the cohort median in MIBC and PDAC); TNBC, triple-negative breast cancer.
The maturation stage of TLSs in cancer varies from B cell aggregates to organized structures with germinal centres42,96. In untreated lung40,41, colorectal83 and bladder cancer100, mature, germinal centre-positive TLSs are a positive prognosticator of survival, whereas immature TLSs have either no association or a weak association with survival. In hepatocellular carcinoma, mature TLSs correlate with improved survival101,102,103, whereas immature TLSs serve as survival niches for tumour progenitor cells and confer a worse prognosis104. Similarly, in kidney cancer, TLSs are mostly immature and a higher density of these TLSs correlates with a dismal prognosis100, a phenomenon that is replicated in RCC-derived lung metastases105. Thus, TLSs correlate with improved outcome in many but not all cancer types, and differences in the TLS composition or maturation may explain this discrepancy.
Based on their presumed role in B and T cell priming, a high TLS density was expected to be associated with response to immunotherapies including immune checkpoint inhibitors (ICI). Some but not all studies have reported such an association. Methodological differences, small cohort sizes and other limitations might underlie these conflicting results, underscoring the need to establish standardized TLS definitions and quantification methods for future studies.
Two studies that used transcriptomic data to quantify TLSs in tumour samples reported that these structures were associated with improved response to ICI therapy in patients with metastatic kidney cancer and melanoma11,12. However, a large fraction of the samples that were analysed at baseline were lymph node metastases, which may make it difficult to attribute the transcriptomic data specifically to TLSs.
A study in patients with sarcoma who received anti-PD1 therapy reported an objective response rate of 30% in those who were selected based on baseline TLS positivity compared with 2.4% in an all-comer cohort106,107. However, the sarcoma histological subtypes differed significantly between the TLS-positive and the all-comer cohorts107, which could be a confounding factor108,109. In patients with high-risk bladder cancer, TLS density predicted response to ICI in a US phase I trial110 but not in the phase Ib NABUCCO trial in the Netherlands28; all clinical variables in these trials were comparable. TLS density is highly variable even among patients with the same disease; thus, the small sample sizes of these phase I trials lack sufficient statistical power to analyse the biomarker potential of TLS and may explain the conflicting results. Retrospective analyses of patients with gastric cancer111 and other tumour types including RCC112 reported that the presence of mature TLSs before ICI treatment was associated with improved responses to this therapy. Together, these findings suggest that the composition of TLSs is a more reliable predictive or prognostic factor than their density. Large prospective cohort studies are needed to determine whether TLSs are associated with a positive ICI response in kidney cancer and, if so, whether the discrepancy between this positive response and the negative prognostic association of TLSs in untreated patients100,105 can be attributed to the quality of the TLSs.
A meta-analysis showed that tumour mutation burden and CXCL13 expression by tumour-infiltrating T cells were the main positive predictors of the response to ICIs across seven different cancer types113. Similarly, CXCL13-expressing PD-1+CD8+ T cells predicted the clinical response to ICI in lung cancer114. The relationship between CXCL13-expressing T cells and TLSs remains to be investigated. Analyses of tumour samples from patients who have received ICI therapy or the GVAX tumour vaccine have consistently shown that those from responders have higher TLS density or higher levels of TLS-related transcripts than do those of non-responders11,28,85,115,116. This finding suggests that TLSs develop in the tumour microenvironment in response to an ongoing immune response.
No data are currently available from sufficiently large cohorts to determine whether TLSs are a cause or a consequence of tumour-specific immunity3. However, evidence suggests that TLS induction might have therapeutic potential as a stand-alone treatment or in combination with other (immune) therapies. In a mouse model of ovarian cancer, administration of recombinant CXCL13 induced TLSs and had antitumour efficacy117. In a peritoneal melanoma model, TLS development was increased by ICI therapy and correlated with tumour reduction53. Several studies have reported that treatment with ICIs in combination with intratumoural CXCL13 injection118, administration of stimulator of interferon genes (STING) agonist119 or TLR9 agonist120, targeted delivery of LIGHT (also known as TNSF14), which is a ligand of the lymphotoxin β-receptor (LTβR)121,122, or injection of lymph node-derived stromal cells123, resulted in TLS development and improved control of experimental tumours compared with monotherapies (ICI or the second component alone). Synergistic effects were also observed when chemotherapy was combined with intratumoural CXCL13 and CCL21 injection to induce TLS development in a pancreatic cancer model124. In a chemotherapy-resistant colorectal cancer model, the combination of antiangiogenic agents with agonistic anti-CD40 antibody induced vascular normalization, TLS development and T cell-mediated tumour control125. However, in experimental glioma, agonistic anti-CD40 antibody induced immature TLSs, which correlated with an impaired response to ICI126. In a model of autochthonous pancreatic ductal adenocarcinoma, tumour antigen-specific DNA vaccination induced expansion of antigen-specific T cells and the development of mature TLSs but the effects on tumour growth were not reported127. For all of these treatments, TLS induction was just one of many immunostimulatory effects; thus, further studies are needed to investigate the specific contributions of TLSs to spontaneous or therapy-induced antitumour immunity.
HEVs could have crucial roles in TLS induction and represent a predecessor to TLSs. They develop in inflamed tissues, can promote the formation of T cell aggregates128,129 and are major sites of lymphocyte entry into tumours130. In patients with metastatic melanoma, the presence of tumour-associated HEVs was associated with a greater clinical response to treatment with ICIs, including improved survival130. Administration of an agonistic antibody to LTβR increased the presence of HEVs in experimental sarcoma, resulting in improved effector T cell function and a reduction in tumour growth130. Furthermore, combined treatment with anti-VEGFR2 and anti-PDL1 antibodies stimulated tumour immunity by inducing HEVs in murine models131. Similarly, in a mouse metastatic breast cancer model, targeted delivery of an anti-neoplastic drug to tumours using a monoclonal antibody against surface molecules on HEVs improved tumour control132. Although the presence of HEVs is insufficient to drive TLS development, their role in this process warrants further investigation.
In summary, the available data suggest that mature TLSs are associated with an antitumour immune response and improved survival in multiple cancer types. However, whether TLS development is a consequence of, or a prerequisite for, effective antitumour immunity is unknown.
Infection
The roles of TLSs in the context of infection have mostly been investigated using pulmonary models. The lung is constantly exposed to pathogenic bacteria, viruses, allergens and harmful particles that are present in inhaled air133. In the lung, TLSs develop quickly in response to viral or bacterial infections and drive an effective immune response including antigen-specific T cells and B cells that provides protection against harmful pathogens134,135. Bacterial infection with Mycobacterium tuberculosis, Pseudomonas aeruginosa or Staphylococcus aureus induces TLS formation in the lung136,137. Intratracheal mucosal vaccination against M. tuberculosis also results in TLS formation and impairment of this process ameliorated vaccine-induced immunity against the bacteria, demonstrating that TLS formation is important for the induction of protective immune responses138,139. Mucosal vaccine-induced immunity against M. tuberculosis is dependent on IL-17-producing Th17 cells, which are crucial for TLS development in the lung138.
Infection with influenza virus52 or severe acute respiratory syndrome corona virus (SARS-CoV)140 is also often accompanied by TLS development in the lung. In response to infection with influenza virus, mice that lacked SLOs developed TLSs, which mediated clearance of the infection and improved survival compared with mice with SLOs17. In mice with influenza virus infection, depletion of CD11chi dendritic cells after clearance of the virus from the lung resulted in disintegration of TLSs and a reduction in antibodies to the virus141. These results indicate protective roles of TLSs against viral infection.
The memory T cell population is heterogeneous and includes TRM cells with high expression of C-type lectin CD69 and/or the integrin CD103 (ref. 142). TRM reside in non-lymphoid tissues and govern local immunity in mucosal tissue in locations such as the lung, skin and gut142,143. Influenza virus infection induces heterogeneous CD4+ TRM cells, including T resident helper (TRH) cells that reside within TLSs and promote local antibody production that protects against influenza reinfection144. Mucosal vaccination with heat-killed Klebsiella pneumonia induces CD4+ TRM cells, which have an important role in the bacterial clearance of K. pneumonia infection63. SARS-CoV-2 infection and mRNA COVID-19 vaccination also generate TRM cells145,146 and the number of SARS-CoV-2-specific TRM cells in the airway correlates with survival in patients with severe COVID-19 (ref. 147), suggesting that TRM cells and TLSs are also involved in protective immunity against SARS-CoV-2. Thus, TLSs with TRM cells in the lung are a key target for the development of novel mucosal vaccines against pathogens that cause severe acute respiratory diseases.
Patients with severe complicated pyelonephritis who are resistant to antibiotics and require surgical resections exhibit many TLSs in the renal cortex and pelvis21,51, suggesting that renal TLSs also generate anti-pathogen adaptive immune responses. How the cellular and molecular components, functions and trigger of TLSs differ between kidneys and other mucosal tissues is unknown and requires further investigation.
Chronic inflammatory diseases and ageing
TLSs are induced in chronic inflammatory diseases of the lung, kidney and other organs. Ageing is also associated with chronic inflammation, which contributes to age-associated morbidity and mortality. Targeting this age-dependent non-specific inflammation, termed inflammaging, is an important strategy for preventing age-related diseases and extending the healthy lifespan1. Ageing is also gaining attention as a cause of TLS formation in various organs. Emerging evidence suggests that TLSs and their molecular machinery are involved in impaired tissue regeneration capacity in older people and in age-related diseases7,8,13,148,149.
Lung
Allergens, inhaled particles, DAMPs and self-antigens can trigger TLS formation in the lung. In contrast to their protective role in lung infections, TLSs have pathogenic roles in chronic inflammatory lung diseases such as asthma and chronic obstructive pulmonary disease (COPD), which is an age-related disease induced by chronic exposure to cigarette smoke61,150,151. In patients with COPD, TLS number is positively associated with disease severity152,153. Type 3 innate lymphoid cells (ILC3s) that express neuropilin-1 have LTi activity and are involved in the initiation of TLS development via the production of IL-17A and IL-22 in patients with COPD154. Lymphotoxin β-receptor (LTβR) signalling promotes TLS development and LTβR ligand expression is enhanced in immune cells from these patients151. In mice that were chronically exposed to cigarette smoke, inhibition of LTβR signalling disrupted smoking-related TLSs in the lung and induced regeneration of lung tissue151, indicating that TLSs contribute to shaping the pathogenesis of lung diseases during chronic inflammation.
A subpopulation of memory Th2 cells, memory pathogenic Th2 (Tpath2) cells, that produce large amounts of inflammatory cytokines, such as IL-5, IL-13 and amphiregulin, are involved in the pathogenesis of various inflammatory diseases155,156. Memory Tpath2 cells show the characteristics of TRM cells and are maintained within lung TLSs61,157. Unique lymphatic endothelial cells (LECs) that express Thy1 are crucial for memory Tpath2 cell survival in TLSs in the inflamed lung, possibly by providing the key survival cytokine IL-7 (ref. 61). Thy1+ LECs also produce IL-33 and the T cell-attracting chemokines CCL21 and CCL19. IL-33 induces enhanced production of IL-5, IL-13 and amphiregulin by memory Tpath2 cells. Thus, Thy1+ LECs generate an inflammatory niche and regulate the quantity and quality of TLS-residing memory Tpath2 cells during chronic inflammation155. These data suggest that memory Tpath2 cells and Thy1+ LECs, which are TLS pathogenic components in the inflamed lung, could be potent therapeutic targets for intractable inflammatory diseases.
Differences in the cellular components of lung TLSs may contribute to their opposite roles in infection and chronic inflammation. However, further studies are needed to determine the critical factors that dictate the composition of lung TLS under different inflammatory conditions.
Kidney
TLSs can be induced in CKDs, including IgA nephropathy158,159,160, lupus nephritis65, IgG4-related kidney diseases161, interstitial nephritis162,163, membranous nephropathy164 and antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis165,166 as well as in kidney allografts37,39,44,167,168,169,170. Clinical studies have demonstrated that TLS formation is associated with poor kidney outcomes in these diseases (Supplementary Table 2).
Studies in patients with IgA nephropathy have reported that TLSs were detected in 30–40% of participants and were associated with higher levels of serum creatinine, proteinuria and blood pressure, greater severity of glomerulonephritis and future disease progression158,159,160. A study in patients with membranous nephropathy reported that TLSs were detected in 34.2% of participants and were associated with higher levels of serum creatinine, proteinuria and blood pressure, lower serum albumin concentrations and lower remission rates164. Notably, a higher proportion of patients with membranous nephropathy who had renal TLSs had anti-PLA2R autoantibodies than those without TLSs (72.5% versus 47.4%)164. However, most of these studies were observational and the cellular and molecular characteristics of TLSs as well as the mechanisms that underlie the observed associations remain unclear.
In elderly people, AKI often leads to kidney failure149,171 but the mechanism is unclear. In mice, we found that aged (1-year-old) but not young kidneys (2-month-old) develop TLSs after injury and that TLS development was associated with impaired regenerative capacity in the aged, injured kidney7,21. To define the cellular and molecular basis for TLSs in the kidney, we established an inducible kidney TLS model using 1-year-old mice. Using this model, which enables various maturational stages of TLSs to be induced by changing the severity of injury, we performed a detailed analysis of the roles of stromal and haematopoietic cells in age-dependent TLS development7,13,21. We found that in aged, injured kidneys, fibroblasts have crucial roles in TLS formation and maturation54. In young, injured kidneys, resident fibroblasts transdifferentiate into scar-producing myofibroblasts at the cost of physiological erythropoietin production, leading to fibrosis and renal anaemia, which are common pathological conditions in CKD172,173,174. In aged, injured kidneys, fibroblasts also transdifferentiate into TLS-related heterogeneous fibroblasts with distinct phenotypes7 (Fig. 2b). In the early phase of TLS formation, fibroblasts surrounding TLSs produce retinoic acid, which promotes transdifferentiation of fibroblasts inside TLSs into neural crest marker p75NTR-expressing fibroblasts, some of which produce CXCL13 and CCL19. Subsequently, some of the fibroblasts inside TLSs lose p75NTR expression and mature into CXCL13-producing FDCs. Most of these TLS-associated fibroblasts are derived from a single lineage of resident fibroblasts7. These observations indicate that paracrine interactions between heterogeneous fibroblasts orchestrate TLS formation in the kidney.
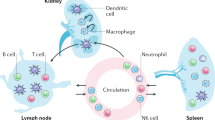
Intimate cellular interactions also occur in the TLS haematopoietic compartment. We showed the accumulation of two unique age-dependent lymphocytes, senescence-associated T (SAT) cells and age-associated B cells (ABCs) in aged, injured kidneys13. SAT cells are unique CD4 T cells that exhibit defective proliferation and production of T cell cytokines in response to T cell receptor stimulation, but secrete abundant atypical humoral factors such as osteopontin175,176. ABCs are defined as Tbx21+CD11b+/CD11c+ B cells and act as antigen-presenting cells177,178 (Fig. 2c). In aged, injured kidneys, SAT cells and ABCs reside within TLSs in close contact with each other. SAT cells produce ABC-inducing factors, such as IL-21 and IFNγ179 and expand together with ABCs after injury13. In addition to IFNγ and IL-21, unbiased receptor ligand analysis identified CD153–CD30 (Tnfsf8–Tnfrsf8) signalling between SAT cells and ABCs. In mice, genetic deficiency of CD153 or CD30 impaired functional SAT induction, resulting in a reduction in ABC numbers and attenuation of TLS formation with improved kidney function and a reduction in fibrosis and inflammation13. These results indicate that CD153–CD30 signalling is required for functional SAT cell induction and TLS formation and that TLS formation is maladaptive in aged, injured kidneys (Fig. 2d). SAT cells and ABCs are also induced in mouse lupus nephritis models and show similar molecular interactions to those that occur in aged, injured kidneys180.
Cellular and molecular components of age-dependent TLS formation are similar in mice and humans, suggesting that this phenomenon is conserved across species7,13,21. Elderly (2-year-old) mice without kidney injury and healthy aged people without CKD spontaneously develop TLSs in the kidney7,21,181. Further studies are needed to identify the mechanisms that lead to this spontaneous TLS formation and the involvement of SAT cells and ABCs in this process.
Other organs
Spontaneous age-dependent TLS formation has also been observed in organs such as the liver and bladder22,23,181. SAT cells and ABC development are observed in age-dependent TLSs in the bladder and spleen22,180,182 and in kidneys and visceral adipose tissues in conditions that accelerate immune ageing, such as autoimmune diseases180,183,184 and obesity185,186 in mouse models. Studies that investigated the effects of loss of function of SAT cells and ABCs demonstrated their pathogenic potentials in these contexts13,180,183,184,186, which is consistent with the findings in aged, injured kidneys that are described above.
A subset of peripheral T helper cells with B cell helper functions that express PD1, CD4 and CD153 (ref. 187) and ABCs that express CD30 was identified in joint tissue from patients with rheumatoid arthritis, suggesting a role of CD153–CD30 signalling in T cell–B cell interactions in human TLSs13. The presence of an age-dependent human CD153+PD1+CD4+ T cell population with B cell helper functions should be investigated in future studies.
Another potential cellular driver of age-dependent TLS formation is vascular endothelial cells. Loss of Notch signalling in endothelial cells results in TLS formation in the kidneys57. Notch signalling components and Notch-dependent vascular networks in bone decrease with age188, but whether a similar age-dependent decrease in Notch signalling occurs in endothelial cells and contributes to age-dependent TLS formation remains to be investigated. Age-dependent phenotypic changes in other cell populations such as fibroblasts and pericytes could also potentially contribute to TLS formation, but this hypothesis requires further investigation. TLSs that are induced in aged organs and in autoimmune diseases might be maladaptive and are a potential novel therapeutic target7,8,13,148.
Conclusions and future research
In the past two decades, knowledge of TLSs has substantially expanded. Molecular and phenotypic heterogeneity within TLSs is increasingly recognized in mice and humans and is associated with the disease course in autoimmune diseases, cancer, infection and chronic inflammatory diseases. Ageing has also emerged as a cause of TLS formation and the molecular details and therapeutic potential are beginning to be unravelled.
The unique contribution of TLSs to local immunity in chronic inflammatory conditions might result from their optimal adaptation to the local tissue microenvironment. However, the mechanisms by which TLSs adapt to tissue and generate tailored immune responses in situ are largely unknown and many unanswered questions remain (Box 2). For example, the initial event that leads to TLS induction and the factors that influence susceptibility to TLS development in different organs and diseases remain to be determined.
In addition to multistep interactions between immune cells and non-immune cells, factors that shape the tissue microenvironment may influence immune responses generated within TLSs. For example, several studies have shown that the ionic microenvironment directly affects a variety of immune cells. In the tumour microenvironment, extracellular potassium released by necrotic cells suppresses T cell effector functions189. Furthermore, increased sodium in inflammatory tissues promotes IL-17-producing CD4+ T cell development and inhibits regulatory T cell differentiation, exacerbating inflammation190,191. The effects of the injured tissue microenvironment on immune responses in TLSs should be further investigated.
Even in the same disease, the clinical influence of TLSs varies depending on the disease phase and the cellular composition of the TLSs. Furthermore, how TLSs influence responses to therapy and how therapy changes the function of TLSs are poorly understood. The opposing clinical correlations of TLSs in different diseases indicate that the optimal approach for targeting these structures must be defined in a context-dependent manner. It is therefore important to determine the conditions that promote the development or induce the resolution of TLSs.
Chronic inflammation is a common feature of all forms of CKD and AKI149,192,193,194 and TLSs could potentially drive CKD progression by maintaining and accelerating inflammation irrespective of the aetiology. Therapeutic interventions that target TLS formation could potentially be beneficial in CKD and other chronic inflammatory and autoimmune diseases. As CKD can progress without overt symptoms, the development of non-invasive biomarkers for TLS detection will also be necessary. Furthermore, the identification of essential pathways for TLS formation will enable selective targeting to induce TLS development or resolution as required to obtain clinical benefits in different diseases. Accumulation of basic and clinical evidence, particularly in human TLSs, using standardized quantification approaches is necessary to guide the future development of novel therapeutic approaches to TLS-related chronic diseases.
References
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Antonioli, L. et al. Ectopic lymphoid organs and immune-mediated diseases: molecular basis for pharmacological approaches. Trends Mol. Med. 26, 1021–1033 (2020).
Schumacher, T. N. & Thommen, D. S. Tertiary lymphoid structures in cancer. Science 375, eabf9419 (2022).
Sato, Y. & Yanagita, M. Immunology of the ageing kidney. Nat. Rev. Nephrol. 15, 625–640 (2019).
Gago da Graca, C., van Baarsen, L. G. M. & Mebius, R. E. Tertiary lymphoid structures: diversity in their development, composition, and role. J. Immunol. 206, 273–281 (2021).
Ruddle, N. H. Lymphatic vessels and tertiary lymphoid organs. J. Clin. Invest. 124, 953–959 (2014).
Sato, Y. et al. Heterogeneous fibroblasts underlie age-dependent tertiary lymphoid tissues in the kidney. JCI Insight 1, e87680 (2016).
Camell, C. D. et al. Aging induces an Nlrp3 inflammasome-dependent expansion of adipose B cells that impairs metabolic homeostasis. Cell Metab. 30, 1024–1039 e1026 (2019).
Nayar, S. et al. Immunofibroblasts are pivotal drivers of tertiary lymphoid structure formation and local pathology. Proc. Natl Acad. Sci. USA 116, 13490–13497 (2019).
Petitprez, F. et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 577, 556–560 (2020).
Helmink, B. A. et al. B cells and tertiary lymphoid structures promote immunotherapy response. Nature 577, 549–555 (2020).
Cabrita, R. et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature 577, 561–565 (2020).
Sato, Y. et al. CD153/CD30 signaling promotes age-dependent tertiary lymphoid tissue expansion and kidney injury. J. Clin. Invest. 132, e146071 (2022).
Mazor, R. D. et al. Tumor-reactive antibodies evolve from non-binding and autoreactive precursors. Cell 185, 1208–1222 e1221 (2022).
Meylan, M. et al. Tertiary lymphoid structures generate and propagate anti-tumor antibody-producing plasma cells in renal cell cancer. Immunity 55, 527–541 e525 (2022).
Fridman, W. H. et al. B cells and tertiary lymphoid structures as determinants of tumour immune contexture and clinical outcome. Nat. Rev. Clin. Oncol. 19, 441–457 (2022).
Moyron-Quiroz, J. E. et al. Role of inducible bronchus associated lymphoid tissue (iBALT) in respiratory immunity. Nat. Med. 10, 927–934 (2004).
Adachi, Y. et al. Distinct germinal center selection at local sites shapes memory B cell response to viral escape. J. Exp. Med. 212, 1709–1723 (2015).
Humby, F. et al. Ectopic lymphoid structures support ongoing production of class-switched autoantibodies in rheumatoid synovium. PLoS Med. 6, e1 (2009).
Cippa, P. E. et al. A late B lymphocyte action in dysfunctional tissue repair following kidney injury and transplantation. Nat. Commun. 10, 1157 (2019).
Sato, Y. et al. Developmental stages of tertiary lymphoid tissue reflect local injury and inflammation in mouse and human kidneys. Kidney Int. 98, 448–463 (2020).
Ligon, M. M. et al. Single cell and tissue-transcriptomic analysis of murine bladders reveals age- and TNFα-dependent but microbiota-independent tertiary lymphoid tissue formation. Mucosal Immunol. 13, 908–918 (2020).
Singh, P. et al. Lymphoid neogenesis and immune infiltration in aged liver. Hepatology 47, 1680–1690 (2008).
Sato, Y., Tamura, M. & Yanagita, M. Tertiary lymphoid tissues: a regional hub for kidney inflammation. Nephrol. Dial. Transpl. 38, 26–33 (2023).
Rangel-Moreno, J. et al. The development of inducible bronchus-associated lymphoid tissue depends on IL-17. Nat. Immunol. 12, 639–646 (2011).
Marinkovic, T. et al. Interaction of mature CD3+CD4+ T cells with dendritic cells triggers the development of tertiary lymphoid structures in the thyroid. J. Clin. Invest. 116, 2622–2632 (2006).
Cupedo, T. et al. Human fetal lymphoid tissue-inducer cells are interleukin 17-producing precursors to RORC+ CD127+ natural killer-like cells. Nat. Immunol. 10, 66–74 (2009).
van Dijk, N. et al. Preoperative ipilimumab plus nivolumab in locoregionally advanced urothelial cancer: the NABUCCO trial. Nat. Med. 26, 1839–1844 (2020).
Pikor, N. B. et al. Integration of Th17- and lymphotoxin-derived signals initiates meningeal-resident stromal cell remodeling to propagate neuroinflammation. Immunity 43, 1160–1173 (2015).
Furtado, G. C. et al. TNFα-dependent development of lymphoid tissue in the absence of RORγt+ lymphoid tissue inducer cells. Mucosal Immunol. 7, 602–614 (2014).
Cyster, J. G., Hartley, S. B. & Goodnow, C. C. Competition for follicular niches excludes self-reactive cells from the recirculating B-cell repertoire. Nature 371, 389–395 (1994).
Cyster, J. G. & Goodnow, C. C. Antigen-induced exclusion from follicles and anergy are separate and complementary processes that influence peripheral B cell fate. Immunity 3, 691–701 (1995).
Le Pottier, L. et al. Ectopic germinal centers are rare in Sjogren’s syndrome salivary glands and do not exclude autoreactive B cells. J. Immunol. 182, 3540–3547 (2009).
Bombardieri, M., Lewis, M. & Pitzalis, C. Ectopic lymphoid neogenesis in rheumatic autoimmune diseases. Nat. Rev. Rheumatol. 13, 141–154 (2017).
Akama-Garren, E. H. & Carroll, M. C. T cell help in the autoreactive germinal center. Scand. J. Immunol. 95, e13192 (2022).
Moyron-Quiroz, J. E. et al. Persistence and responsiveness of immunologic memory in the absence of secondary lymphoid organs. Immunity 25, 643–654 (2006).
Sarwal, M. et al. Molecular heterogeneity in acute renal allograft rejection identified by DNA microarray profiling. N. Engl. J. Med. 349, 125–138 (2003).
Thaunat, O. et al. Lymphoid neogenesis in chronic rejection: evidence for a local humoral alloimmune response. Proc. Natl Acad. Sci. USA 102, 14723–14728 (2005).
Bagnasco, S. M. et al. CD20-positive infiltrates in renal allograft biopsies with acute cellular rejection are not associated with worse graft survival. Am. J. Transpl. 7, 1968–1973 (2007).
Scheepstra, C. et al. B cells in cluster or in a scattered pattern do not correlate with clinical outcome of renal allograft rejection. Transplantation 86, 772–778 (2008).
Silina, K. et al. Germinal centers determine the prognostic relevance of tertiary lymphoid structures and are impaired by corticosteroids in lung squamous cell carcinoma. Cancer Res. 78, 1308–1320 (2018).
Sautes-Fridman, C., Petitprez, F., Calderaro, J. & Fridman, W. H. Tertiary lymphoid structures in the era of cancer immunotherapy. Nat. Rev. Cancer 19, 307–325 (2019).
Buckley, C. D., Barone, F., Nayar, S., Benezech, C. & Caamano, J. Stromal cells in chronic inflammation and tertiary lymphoid organ formation. Annu. Rev. Immunol. 33, 715–745 (2015).
Lee, Y. H. et al. Advanced tertiary lymphoid tissues in protocol biopsies are associated with progressive graft dysfunction in kidney transplant recipients. J. Am. Soc. Nephrol. 33, 186–200 (2022).
Masum, M. A. et al. Vasculature-associated lymphoid tissue: a unique tertiary lymphoid tissue correlates with renal lesions in lupus nephritis mouse model. Front. Immunol. 11, 595672 (2020).
Grabner, R. et al. Lymphotoxin β receptor signaling promotes tertiary lymphoid organogenesis in the aorta adventitia of aged ApoE−/− mice. J. Exp. Med. 206, 233–248 (2009).
Dahlgren, M. W. & Molofsky, A. B. Adventitial cuffs: regional hubs for tissue immunity. Trends Immunol. 40, 877–887 (2019).
Dahlgren, M. W. et al. Adventitial stromal cells define group 2 innate lymphoid cell tissue niches. Immunity 50, 707–722 e706 (2019).
Arai, H., Sato, Y. & Yanagita, M. Fibroblast heterogeneity and tertiary lymphoid tissues in the kidney. Immunol. Rev. 302, 196–210 (2021).
Krautler, N. J. et al. Follicular dendritic cells emerge from ubiquitous perivascular precursors. Cell 150, 194–206 (2012).
Ichii, O. et al. Close association between altered urine-urothelium barrier and tertiary lymphoid structure formation in the renal pelvis during nephritis. J. Am. Soc. Nephrol. 33, 88–107 (2022).
Denton, A. E. et al. Type I interferon induces CXCL13 to support ectopic germinal center formation. J. Exp. Med. 216, 621–637 (2019).
Rodriguez, A. B. et al. Immune mechanisms orchestrate tertiary lymphoid structures in tumors via cancer-associated fibroblasts. Cell Rep. 36, 109422 (2021).
Sato, Y. & Yanagita, M. Functional heterogeneity of resident fibroblasts in the kidney. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 95, 468–478 (2019).
Barone, F. et al. IL-22 regulates lymphoid chemokine production and assembly of tertiary lymphoid organs. Proc. Natl Acad. Sci. USA 112, 11024–11029 (2015).
Yamamoto, S. & Yanagita, M. A novel pathological mechanism of tertiary lymphoid structure formation in the renal pelvis. J. Am. Soc. Nephrol. 33, 4–6 (2022).
Fleig, S. et al. Loss of vascular endothelial notch signaling promotes spontaneous formation of tertiary lymphoid structures. Nat. Commun. 13, 2022 (2022).
Petrova, T. V. & Koh, G. Y. Biological functions of lymphatic vessels. Science 369, eaax4063 (2020).
Randolph, G. J., Angeli, V. & Swartz, M. A. Dendritic-cell trafficking to lymph nodes through lymphatic vessels. Nat. Rev. Immunol. 5, 617–628 (2005).
Czepielewski, R. S. et al. Ileitis-associated tertiary lymphoid organs arise at lymphatic valves and impede mesenteric lymph flow in response to tumor necrosis factor. Immunity 54, 2795–2811.e9 (2021).
Shinoda, K. et al. Thy1+IL-7+ lymphatic endothelial cells in iBALT provide a survival niche for memory T-helper cells in allergic airway inflammation. Proc. Natl Acad. Sci. USA 113, E2842–E2851 (2016).
Miller, C. N. et al. IL-7 production in murine lymphatic endothelial cells and induction in the setting of peripheral lymphopenia. Int. Immunol. 25, 471–483 (2013).
Amezcua Vesely, M. C. et al. Effector TH17 cells give rise to long-lived TRM cells that are essential for an immediate response against bacterial infection. Cell 178, 1176–1188.e15 (2019).
Takemura, S. et al. Lymphoid neogenesis in rheumatoid synovitis. J. Immunol. 167, 1072–1080 (2001).
Chang, A. et al. In situ B cell-mediated immune responses and tubulointerstitial inflammation in human lupus nephritis. J. Immunol. 186, 1849–1860 (2011).
Korpos, E. et al. Identification and characterisation of tertiary lymphoid organs in human type 1 diabetes. Diabetologia 64, 1626–1641 (2021).
Gomez-Nguyen, A. et al. Chronic stress induces colonic tertiary lymphoid organ formation and protection against secondary injury through IL-23/IL-22 signaling. Proc. Natl Acad. Sci. USA 119, e2208160119 (2022).
Armengol, M. P. et al. Thyroid autoimmune disease: demonstration of thyroid antigen-specific B cells and recombination-activating gene expression in chemokine-containing active intrathyroidal germinal centers. Am. J. Pathol. 159, 861–873 (2001).
Lopez De Padilla, C. M., Vallejo, A. N., Lacomis, D., McNallan, K. & Reed, A. M. Extranodal lymphoid microstructures in inflamed muscle and disease severity of new-onset juvenile dermatomyositis. Arthritis Rheum. 60, 1160–1172 (2009).
Schonbeck, S., Padberg, F., Hohlfeld, R. & Wekerle, H. Transplantation of thymic autoimmune microenvironment to severe combined immunodeficiency mice. A new model of myasthenia gravis. J. Clin. Invest. 90, 245–250 (1992).
Cantaert, T. et al. B lymphocyte autoimmunity in rheumatoid synovitis is independent of ectopic lymphoid neogenesis. J. Immunol. 181, 785–794 (2008).
Canete, J. D. et al. Ectopic lymphoid neogenesis in psoriatic arthritis. Ann. Rheum. Dis. 66, 720–726 (2007).
Anders, H. J. et al. Lupus nephritis. Nat. Rev. Dis. Prim. 6, 7 (2020).
Yu, F. et al. Tubulointerstitial lesions of patients with lupus nephritis classified by the 2003 International Society of Nephrology and Renal Pathology Society system. Kidney Int. 77, 820–829 (2010).
Liarski, V. M. et al. Cell distance mapping identifies functional T follicular helper cells in inflamed human renal tissue. Sci. Transl. Med. 6, 230ra246 (2014).
Yung, S., Tsang, R. C., Sun, Y., Leung, J. K. & Chan, T. M. Effect of human anti-DNA antibodies on proximal renal tubular epithelial cell cytokine expression: implications on tubulointerstitial inflammation in lupus nephritis. J. Am. Soc. Nephrol. 16, 3281–3294 (2005).
Jamaly, S., Rakaee, M., Abdi, R., Tsokos, G. C. & Fenton, K. A. Interplay of immune and kidney resident cells in the formation of tertiary lymphoid structures in lupus nephritis. Autoimmun. Rev. 20, 102980 (2021).
Stock, A. D. et al. Tertiary lymphoid structures in the choroid plexus in neuropsychiatric lupus. JCI Insight 4, 124203 (2019).
Rangel-Moreno, J. et al. Inducible bronchus-associated lymphoid tissue (iBALT) in patients with pulmonary complications of rheumatoid arthritis. J. Clin. Invest. 116, 3183–3194 (2006).
Bugatti, S. et al. Involvement of subchondral bone marrow in rheumatoid arthritis: lymphoid neogenesis and in situ relationship to subchondral bone marrow osteoclast recruitment. Arthritis Rheum. 52, 3448–3459 (2005).
Luo, S. et al. Chronic inflammation: a common promoter in tertiary lymphoid organ neogenesis. Front. Immunol. 10, 2938 (2019).
Giannakis, M. et al. Genomic correlates of immune-cell infiltrates in colorectal carcinoma. Cell Rep. 15, 857–865 (2016).
Posch, F. et al. Maturation of tertiary lymphoid structures and recurrence of stage II and III colorectal cancer. Oncoimmunology 7, e1378844 (2018).
Rozek, L. S. et al. Tumor-infiltrating lymphocytes, Crohn’s-like lymphoid reaction, and survival from colorectal cancer. J. Natl Cancer Inst. 108, djw027 (2016).
Pagliarulo, F. et al. Molecular, immunological, and clinical features associated with lymphoid neogenesis in muscle invasive bladder cancer. Front. Immunol. 12, 793992 (2021).
A, J. G. et al. Germinal center reactions in tertiary lymphoid structures associate with neoantigen burden, humoral immunity and long-term survivorship in pancreatic cancer. Oncoimmunology 10, 1900635 (2021).
Figenschau, S. L., Fismen, S., Fenton, K. A., Fenton, C. & Mortensen, E. S. Tertiary lymphoid structures are associated with higher tumor grade in primary operable breast cancer patients. BMC Cancer 15, 101 (2015).
Lee, H. J. et al. Prognostic significance of tumor-infiltrating lymphocytes and the tertiary lymphoid structures in HER2-positive breast cancer treated with adjuvant trastuzumab. Am. J. Clin. Pathol. 144, 278–288 (2015).
Lee, S. J. et al. Expression of myxovirus resistance A (MxA) is associated with tumor-infiltrating lymphocytes in human epidermal growth factor receptor 2 (HER2)-positive breast cancers. Cancer Res. Treat. 49, 313–321 (2017).
Zou, Y., Zhao, J., Huang, F., Xiang, X. & Xia, Y. Decreased tertiary lymphoid structures in lung adenocarcinomas with ALK rearrangements. J. Clin. Med. 11, 5935 (2022).
Ruffin, A. T. et al. B cell signatures and tertiary lymphoid structures contribute to outcome in head and neck squamous cell carcinoma. Nat. Commun. 12, 3349 (2021).
Wirsing, A. M., Rikardsen, O. G., Steigen, S. E., Uhlin-Hansen, L. & Hadler-Olsen, E. Characterisation and prognostic value of tertiary lymphoid structures in oral squamous cell carcinoma. BMC Clin. Pathol. 14, 38 (2014).
Sarma, K. P. The role of lymphoid reaction in bladder cancer. J. Urol. 104, 843–849 (1970).
Jiao, S. et al. Intratumor expanded T cell clones can be non-sentinel lymph node derived in breast cancer revealed by single-cell immune profiling. J. Immunother. Cancer 10, e003325 (2022).
Poschke, I. et al. Identification of a tumor-reactive T-cell repertoire in the immune infiltrate of patients with resectable pancreatic ductal adenocarcinoma. Oncoimmunology 5, e1240859 (2016).
Coronella-Wood, J. A. & Hersh, E. M. Naturally occurring B-cell responses to breast cancer. Cancer Immunol. Immunother. 52, 715–738 (2003).
Nzula, S., Going, J. J. & Stott, D. I. Antigen-driven clonal proliferation, somatic hypermutation, and selection of B lymphocytes infiltrating human ductal breast carcinomas. Cancer Res. 63, 3275–3280 (2003).
Coronella, J. A. et al. Antigen-driven oligoclonal expansion of tumor-infiltrating B cells in infiltrating ductal carcinoma of the breast. J. Immunol. 169, 1829–1836 (2002).
Zhu, W. et al. A high density of tertiary lymphoid structure B cells in lung tumors is associated with increased CD4+ T cell receptor repertoire clonality. Oncoimmunology 4, e1051922 (2015).
Masuda, T. et al. Unique characteristics of tertiary lymphoid structures in kidney clear cell carcinoma: prognostic outcome and comparison with bladder cancer. J. Immunother. Cancer 10, e003883 (2022).
Li, H. et al. Existence of intratumoral tertiary lymphoid structures is associated with immune cells infiltration and predicts better prognosis in early-stage hepatocellular carcinoma. Aging 12, 3451–3472 (2020).
Wada, Y., Nakashima, O., Kutami, R., Yamamoto, O. & Kojiro, M. Clinicopathological study on hepatocellular carcinoma with lymphocytic infiltration. Hepatology 27, 407–414 (1998).
Calderaro, J. et al. Intra-tumoral tertiary lymphoid structures are associated with a low risk of early recurrence of hepatocellular carcinoma. J. Hepatol. 70, 58–65 (2019).
Finkin, S. et al. Ectopic lymphoid structures function as microniches for tumor progenitor cells in hepatocellular carcinoma. Nat. Immunol. 16, 1235–1244 (2015).
Remark, R. et al. Characteristics and clinical impacts of the immune environments in colorectal and renal cell carcinoma lung metastases: influence of tumor origin. Clin. Cancer Res. 19, 4079–4091 (2013).
Toulmonde, M. et al. Use of PD-1 targeting, macrophage infiltration, and IDO pathway activation in sarcomas: a phase 2 clinical trial. JAMA Oncol. 4, 93–97 (2018).
Italiano, A. et al. Pembrolizumab in soft-tissue sarcomas with tertiary lymphoid structures: a phase 2 PEMBROSARC trial cohort. Nat. Med. 28, 1199–1206 (2022).
Saerens, M. et al. Immune checkpoint inhibitors in treatment of soft-tissue sarcoma: a systematic review and meta-analysis. Eur. J. Cancer 152, 165–182 (2021).
Chen, L. et al. The immunosuppressive niche of soft-tissue sarcomas is sustained by tumor-associated macrophages and characterized by intratumoral tertiary lymphoid structures. Clin. Cancer Res. 26, 4018–4030 (2020).
Gao, J. et al. Neoadjuvant PD-L1 plus CTLA-4 blockade in patients with cisplatin-ineligible operable high-risk urothelial carcinoma. Nat. Med. 26, 1845–1851 (2020).
Mori, T. et al. Tertiary lymphoid structures show infiltration of effective tumor-resident T cells in gastric cancer. Cancer Sci. 112, 1746–1757 (2021).
Vanhersecke, L. et al. Mature tertiary lymphoid structures predict immune checkpoint inhibitor efficacy in solid tumors independently of PD-L1 expression. Nat. Cancer 2, 794–802 (2021).
Litchfield, K. et al. Meta-analysis of tumor- and T cell-intrinsic mechanisms of sensitization to checkpoint inhibition. Cell 184, 596–614 e514 (2021).
Thommen, D. S. et al. A transcriptionally and functionally distinct PD-1+ CD8+ T cell pool with predictive potential in non-small-cell lung cancer treated with PD-1 blockade. Nat. Med. 24, 994–1004 (2018).
Sweeney, K. J. et al. Tertiary lymphoid structures with overlapping histopathologic features of cutaneous marginal zone lymphoma during neoadjuvant cemiplimab therapy are associated with antitumor response. J. Cutan. Pathol. 48, 674–679 (2021).
Groeneveld, C. S. et al. Tertiary lymphoid structures marker CXCL13 is associated with better survival for patients with advanced-stage bladder cancer treated with immunotherapy. Eur. J. Cancer 148, 181–189 (2021).
Ukita, M. et al. CXCL13-producing CD4+ T cells accumulate in the early phase of tertiary lymphoid structures in ovarian cancer. JCI Insight 7, 157215 (2022).
Yang, M. et al. CXCL13 shapes immunoactive tumor microenvironment and enhances the efficacy of PD-1 checkpoint blockade in high-grade serous ovarian cancer. J. Immunother. Cancer 9, e001136 (2021).
Filderman, J. N., Appleman, M., Chelvanambi, M., Taylor, J. L. & Storkus, W. J. STINGing the tumor microenvironment to promote therapeutic tertiary lymphoid structure development. Front. Immunol. 12, 690105 (2021).
Gallotta, M. et al. Inhaled TLR9 agonist renders lung tumors permissive to PD-1 blockade by promoting optimal CD4+ and CD8+ T-cell interplay. Cancer Res. 78, 4943–4956 (2018).
Huang, Y. et al. Dual-mechanism based CTLs infiltration enhancement initiated by Nano-sapper potentiates immunotherapy against immune-excluded tumors. Nat. Commun. 11, 622 (2020).
Johansson-Percival, A. et al. De novo induction of intratumoral lymphoid structures and vessel normalization enhances immunotherapy in resistant tumors. Nat. Immunol. 18, 1207–1217 (2017).
Zhu, G. et al. Induction of tertiary lymphoid structures with antitumor function by a lymph node-derived stromal cell line. Front. Immunol. 9, 1609 (2018).
Delvecchio, F. R. et al. Pancreatic cancer chemotherapy is potentiated by induction of tertiary lymphoid structures in mice. Cell Mol. Gastroenterol. Hepatol. 12, 1543–1565 (2021).
Ragusa, S. et al. Antiangiogenic immunotherapy suppresses desmoplastic and chemoresistant intestinal tumors in mice. J. Clin. Invest. 130, 1199–1216 (2020).
van Hooren, L. et al. Agonistic CD40 therapy induces tertiary lymphoid structures but impairs responses to checkpoint blockade in glioma. Nat. Commun. 12, 4127 (2021).
Castino, G. F. et al. Spatial distribution of B cells predicts prognosis in human pancreatic adenocarcinoma. Oncoimmunology 5, e1085147 (2016).
Blanchard, L. & Girard, J. P. High endothelial venules (HEVs) in immunity, inflammation and cancer. Angiogenesis 24, 719–753 (2021).
Hua, Y. et al. Cancer immunotherapies transition endothelial cells into HEVs that generate TCF1+ T lymphocyte niches through a feed-forward loop. Cancer Cell 40, 1600–1618.e10 (2022).
Asrir, A. et al. Tumor-associated high endothelial venules mediate lymphocyte entry into tumors and predict response to PD-1 plus CTLA-4 combination immunotherapy. Cancer Cell 40, 318–334.e9 (2022).
Allen, E. et al. Combined antiangiogenic and anti-PD-L1 therapy stimulates tumor immunity through HEV formation. Sci. Transl. Med. 9, eaak9679 (2017).
Jiang, L. et al. Simultaneous targeting of primary tumor, draining lymph node, and distant metastases through high endothelial venule-targeted delivery. Nano Today 36, 101045 (2021).
Hewitt, R. J. & Lloyd, C. M. Regulation of immune responses by the airway epithelial cell landscape. Nat. Rev. Immunol. 21, 347–362 (2021).
Marin, N. D., Dunlap, M. D., Kaushal, D. & Khader, S. A. Friend or foe: the protective and pathological roles of inducible bronchus-associated lymphoid tissue in pulmonary diseases. J. Immunol. 202, 2519–2526 (2019).
Silva-Sanchez, A. & Randall, T. D. Role of iBALT in respiratory immunity. Curr. Top. Microbiol. Immunol. 426, 21–43 (2020).
Frija-Masson, J. et al. Bacteria-driven peribronchial lymphoid neogenesis in bronchiectasis and cystic fibrosis. Eur. Respir. J. 49, 1601873 (2017).
Orme, I. M., Robinson, R. T. & Cooper, A. M. The balance between protective and pathogenic immune responses in the TB-infected lung. Nat. Immunol. 16, 57–63 (2015).
Gopal, R. et al. Interleukin-17-dependent CXCL13 mediates mucosal vaccine-induced immunity against tuberculosis. Mucosal Immunol. 6, 972–984 (2013).
Kaushal, D. et al. Mucosal vaccination with attenuated Mycobacterium tuberculosis induces strong central memory responses and protects against tuberculosis. Nat. Commun. 6, 8533 (2015).
Channappanavar, R., Fett, C., Zhao, J., Meyerholz, D. K. & Perlman, S. Virus-specific memory CD8 T cells provide substantial protection from lethal severe acute respiratory syndrome coronavirus infection. J. Virol. 88, 11034–11044 (2014).
GeurtsvanKessel, C. H. et al. Dendritic cells are crucial for maintenance of tertiary lymphoid structures in the lung of influenza virus-infected mice. J. Exp. Med. 206, 2339–2349 (2009).
Mueller, S. N. & Mackay, L. K. Tissue-resident memory T cells: local specialists in immune defence. Nat. Rev. Immunol. 16, 79–89 (2016).
Gray, J. I. & Farber, D. L. Tissue-resident immune cells in humans. Annu. Rev. Immunol. 40, 195–220 (2022).
Swarnalekha, N. et al. T resident helper cells promote humoral responses in the lung. Sci. Immunol. 6, eabb6808 (2021).
Poon, M. M. L. et al. SARS-CoV-2 infection generates tissue-localized immunological memory in humans. Sci. Immunol. 6, eabl9105 (2021).
Ssemaganda, A. et al. Expansion of cytotoxic tissue-resident CD8+ T cells and CCR6+CD161+ CD4+ T cells in the nasal mucosa following mRNA COVID-19 vaccination. Nat. Commun. 13, 3357 (2022).
Szabo, P. A. et al. Longitudinal profiling of respiratory and systemic immune responses reveals myeloid cell-driven lung inflammation in severe COVID-19. Immunity 54, 797–814.e6 (2021).
Zhou, L. et al. Reversible CD8 T cell-neuron cross-talk causes aging-dependent neuronal regenerative decline. Science 376, eabd5926 (2022).
Sato, Y. & Yanagita, M. Immune cells and inflammation in AKI to CKD progression. Am. J. Physiol. Renal Physiol. 315, F1501–F1512 (2018).
Kuroda, E. et al. Inhaled fine particles induce alveolar macrophage death and interleukin-1α release to promote inducible bronchus-associated lymphoid tissue formation. Immunity 45, 1299–1310 (2016).
Conlon, T. M. et al. Inhibition of LTβR signalling activates WNT-induced regeneration in lung. Nature 588, 151–156 (2020).
Hogg, J. C. et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 350, 2645–2653 (2004).
Sullivan, J. L. et al. B Cell-adaptive immune profile in emphysema-predominant chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 200, 1434–1439 (2019).
Shikhagaie, M. M. et al. Neuropilin-1 is expressed on lymphoid tissue residing LTi-like group 3 innate lymphoid cells and associated with ectopic lymphoid aggregates. Cell Rep. 18, 1761–1773 (2017).
Nakayama, T. et al. Th2 cells in health and disease. Annu. Rev. Immunol. 35, 53–84 (2017).
Morimoto, Y. et al. Amphiregulin-producing pathogenic memory T helper 2 cells instruct eosinophils to secrete osteopontin and facilitate airway fibrosis. Immunity 49, 134–150.e6 (2018).
Ichikawa, T. et al. CD103hi Treg cells constrain lung fibrosis induced by CD103lo tissue-resident pathogenic CD4 T cells. Nat. Immunol. 20, 1469–1480 (2019).
Pei, G. et al. Renal interstitial infiltration and tertiary lymphoid organ neogenesis in IgA nephropathy. Clin. J. Am. Soc. Nephrol. 9, 255–264 (2014).
Togashi, R. et al. Tubulointerstitial B-cell infiltration and tertiary lymphoid tissue in adult-onset immunoglobulin A vasculitis with nephritis. Int. Urol. Nephrol. 55, 749–758 (2023).
Luo, R. et al. Tertiary lymphoid organs are associated with the progression of kidney damage and regulated by interleukin-17A. Theranostics 11, 117–131 (2021).
Miyanaga, T. et al. Tertiary lymphoid tissue in early-stage IgG4-related tubulointerstitial nephritis incidentally detected with a tumor lesion of the ureteropelvic junction: a case report. BMC Nephrol. 22, 34 (2021).
Singh, S. et al. Tertiary lymphoid structure signatures are associated with immune checkpoint inhibitor related acute interstitial nephritis. JCI Insight https://doi.org/10.1172/jci.insight.165108 (2022).
Heller, F. et al. The contribution of B cells to renal interstitial inflammation. Am. J. Pathol. 170, 457–468 (2007).
Wang, Z. F. et al. Characteristics and prognostic value of tertiary lymphoid organs in membranous nephropathy: a retrospective study. Front. Med. 8, 803929 (2021).
Steinmetz, O. M. et al. Analysis and classification of B-cell infiltrates in lupus and ANCA-associated nephritis. Kidney Int. 74, 448–457 (2008).
Brix, S. R. et al. Organisation of lymphocytic infiltrates in ANCA-associated glomerulonephritis. Histopathology 72, 1093–1101 (2018).
Filippone, E. J. & Farber, J. L. The implications of B-lineage cells in kidney allografts. Transplantation 104, 2011–2023 (2020).
Thaunat, O. et al. Chronic rejection triggers the development of an aggressive intragraft immune response through recapitulation of lymphoid organogenesis. J. Immunol. 185, 717–728 (2010).
Kayler, L. K. et al. Acute cellular rejection with CD20-positive lymphoid clusters in kidney transplant patients following lymphocyte depletion. Am. J. Transpl. 7, 949–954 (2007).
Hwang, H. S. et al. Clinical impacts of CD38+ B cells on acute cellular rejection with CD20+ B cells in renal allograft. Transplantation 89, 1489–1495 (2010).
Ishani, A. et al. Acute kidney injury increases risk of ESRD among elderly. J. Am. Soc. Nephrol. 20, 223–228 (2009).
Sato, Y. & Yanagita, M. Renal anemia: from incurable to curable. Am. J. Physiol. Renal Physiol. 305, F1239–F1248 (2013).
Asada, N. et al. Dysfunction of fibroblasts of extrarenal origin underlies renal fibrosis and renal anemia in mice. J. Clin. Invest. 121, 3981–3990 (2011).
Kaneko, K. et al. Lineage tracing analysis defines erythropoietin-producing cells as a distinct subpopulation of resident fibroblasts with unique behaviors. Kidney Int. 102, 280–292 (2022).
Tahir, S. et al. A CD153+CD4+ T follicular cell population with cell-senescence features plays a crucial role in lupus pathogenesis via osteopontin production. J. Immunol. 194, 5725–5735 (2015).
Fukushima, Y., Minato, N. & Hattori, M. The impact of senescence-associated T cells on immunosenescence and age-related disorders. Inflamm. Regen. 38, 24 (2018).
Rubtsov, A. V. et al. Toll-like receptor 7 (TLR7)-driven accumulation of a novel CD11c+ B-cell population is important for the development of autoimmunity. Blood 118, 1305–1315 (2011).
Hao, Y., O’Neill, P., Naradikian, M. S., Scholz, J. L. & Cancro, M. P. A B-cell subset uniquely responsive to innate stimuli accumulates in aged mice. Blood 118, 1294–1304 (2011).
Manni, M. et al. Regulation of age-associated B cells by IRF5 in systemic autoimmunity. Nat. Immunol. 19, 407–419 (2018).
Fukushima, Y. et al. cis interaction of CD153 with TCR/CD3 is crucial for the pathogenic activation of senescence-associated T cells. Cell Rep. 40, 111373 (2022).
Huang, Y. et al. Identification of novel genes associated with renal tertiary lymphoid organ formation in aging mice. PLoS One 9, e91850 (2014).
Elyahu, Y. et al. Aging promotes reorganization of the CD4 T cell landscape toward extreme regulatory and effector phenotypes. Sci. Adv. 5, eaaw8330 (2019).
Rubtsova, K. et al. B cells expressing the transcription factor T-bet drive lupus-like autoimmunity. J. Clin. Invest. 127, 1392–1404 (2017).
Nickerson, K. M. et al. Age-associated B cells are heterogeneous and dynamic drivers of autoimmunity in mice. J. Exp. Med. 220, e20221346 (2023).
Shirakawa, K. et al. Obesity accelerates T cell senescence in murine visceral adipose tissue. J. Clin. Invest. 126, 4626–4639 (2016).
Yoshida, S. et al. The CD153 vaccine is a senotherapeutic option for preventing the accumulation of senescent T cells in mice. Nat. Commun. 11, 2482 (2020).
Rao, D. A. et al. Pathologically expanded peripheral T helper cell subset drives B cells in rheumatoid arthritis. Nature 542, 110–114 (2017).
Kusumbe, A. P. et al. Age-dependent modulation of vascular niches for haematopoietic stem cells. Nature 532, 380–384 (2016).
Eil, R. et al. Ionic immune suppression within the tumour microenvironment limits T cell effector function. Nature 537, 539–543 (2016).
Binger, K. J. et al. High salt reduces the activation of IL-4- and IL-13-stimulated macrophages. J. Clin. Invest. 125, 4223–4238 (2015).
Kleinewietfeld, M. et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 496, 518–522 (2013).
Rabb, H. et al. Inflammation in AKI: current understanding, key questions, and knowledge gaps. J. Am. Soc. Nephrol. 27, 371–379 (2016).
Nangaku, M. Chronic hypoxia and tubulointerstitial injury: a final common pathway to end-stage renal failure. J. Am. Soc. Nephrol. 17, 17–25 (2006).
Sato, Y., Takahashi, M. & Yanagita, M. Pathophysiology of AKI to CKD progression. Semin. Nephrol. 40, 206–215 (2020).
Pitzalis, C., Jones, G. W., Bombardieri, M. & Jones, S. A. Ectopic lymphoid-like structures in infection, cancer and autoimmunity. Nat. Rev. Immunol. 14, 447–462 (2014).
Drayton, D. L., Liao, S., Mounzer, R. H. & Ruddle, N. H. Lymphoid organ development: from ontogeny to neogenesis. Nat. Immunol. 7, 344–353 (2006).
Author information
Authors and Affiliations
Contributions
Y.S. and K.S. researched the data for the article. All authors contributed substantially to discussion of the content, and wrote the article. M.Y. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.Y. has received research grants from Mitsubishi Tanabe Pharma and Boehringer Ingelheim. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Nephrology thanks Reza Abdi, Kristin Fenton, A. Richard Kitching and George Tsokos for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sato, Y., Silina, K., van den Broek, M. et al. The roles of tertiary lymphoid structures in chronic diseases. Nat Rev Nephrol 19, 525–537 (2023). https://doi.org/10.1038/s41581-023-00706-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-023-00706-z
This article is cited by
-
Tertiary lymphoid structural heterogeneity determines tumour immunity and prospects for clinical application
Molecular Cancer (2024)
-
Multi-resolution deep learning characterizes tertiary lymphoid structures and their prognostic relevance in solid tumors
Communications Medicine (2024)
-
The immunoregulatory roles of non-haematopoietic cells in the kidney
Nature Reviews Nephrology (2024)