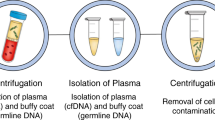
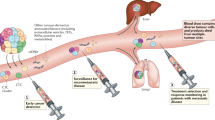
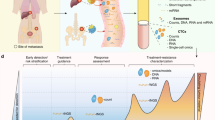
Abstract
In the era of precision oncology, liquid biopsy techniques, especially the use of plasma circulating tumour DNA (ctDNA) analysis, represent a paradigm shift in the use of genomic biomarkers with considerable implications for clinical practice. Compared with tissue-based tumour DNA analysis, plasma ctDNA is more convenient to test, more readily accessible, faster to obtain and less invasive, minimizing procedure-related risks and offering the opportunity to perform serial monitoring. Additionally, genomic profiles of ctDNA have been shown to reflect tumour heterogeneity, which has important implications for the identification of resistant clones and selection of targeted therapy well before clinical and radiographic changes occur. Moreover, plasma ctDNA testing can also be applied to cancer screening, risk stratification and quantification of minimal residual disease. These features provide an unprecedented opportunity for early treatment of patients, improving the chances of treatment success.
Key points
-
Precision medicine entails the targeting of therapy according to the presence of specific genomic biomarkers; thus, the ability to detect such biomarkers is of paramount importance.
-
Liquid biopsies are non-invasive and fast and can be repeatedly performed throughout a disease course and during treatment to provide information on tumour heterogeneity and to capture the emergence of resistant clones.
-
Circulating tumour DNA (ctDNA) analysis offers the opportunity to identify treatment failures well before clinical and radiographic progression, enabling early interventions and possibly increasing the chances of treatment success.
-
Characterization of a tumour’s genomic landscape using ctDNA analysis can identify predictive biomarkers for patient selection and enrolment in clinical trials.
-
ctDNA yield depends on various factors, including time of blood sample acquisition relative to treatment and response, tumour mutational and disease burden, and the amount of tumour-derived cell-free DNA (cfDNA), which should be considered when ordering and interpreting the test.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Weinberg, R. The Biology of Cancer2nd edn. (Taylor & Francis, 2013).
Mok, T. S. et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med. 361, 947–957 (2009).
Motzer, R. J. et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 27, 3584–3590 (2009).
Motzer, R. J. et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet 372, 449–456 (2008).
Choueiri, T. K. et al. Cabozantinib versus everolimus in advanced renal-cell carcinoma. N. Engl. J. Med. 373, 1814–1823 (2015).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001).
Swanton, C. Intratumour heterogeneity: evolution through space and time. Cancer Res. 72, 4875–4882 (2012).
Faltas, B. M. et al. Clonal evolution of chemotherapy-resistant urothelial carcinoma. Nat. Genet. 48, 1490–1499 (2016).
Gundem, G. et al. The evolutionary history of lethal metastatic prostate cancer. Nature 520, 353–357 (2015).
Lanman, R. B. et al. Analytical and clinical validation of a digital sequencing panel for quantitative, highly accurate evaluation of cell-free circulating tumor DNA. PloS One10, e0140712 (2015).
Mok, T. S. et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N. Engl. J. Med. 376, 629–640 (2017).
Mandel, P. & Metais, P. Les acides nucléiques du plasma sanguin chez l’homme. C. R. Seances Soc. Biol. Fil. 142, 241–243 (1948).
Siravegna, G., Marsoni, S., Siena, S. & Bardelli, A. Integrating liquid biopsies into the management of cancer. Nat. Rev. Clin. Oncol. 14, 531–548 (2017).
Jahr, S. et al. DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res. 61, 1659–1665 (2001).
Stroun, M., Lyautey, J., Lederrey, C., Olson-Sand, A. & Anker, P. About the possible origin and mechanism of circulating DNA apoptosis and active DNA release. Clin. Chim. Acta. 313, 139–142 (2001).
Leon, S. A., Shapiro, B., Sklaroff, D. M. & Yaros, M. J. Free DNA in the serum of cancer patients and the effect of therapy. Cancer Res. 37, 646–650 (1977).
Bettegowda, C. et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl Med. 6, 224ra24 (2014).
Diehl, F. et al. Detection and quantification of mutations in the plasma of patients with colorectal tumors. Proc. Natl Acad. Sci. USA 102, 16368–16373 (2005).
Gormally, E., Caboux, E., Vineis, P. & Hainaut, P. Circulating free DNA in plasma or serum as biomarker of carcinogenesis: practical aspects and biological significance. Mutat. Res. 635, 105–117 (2007).
Mouliere, F. et al. Enhanced detection of circulating tumor DNA by fragment size analysis. Sci. Transl Med. 10, eaat4921 (2018).
Jiang, P. et al. Preferred end coordinates and somatic variants as signatures of circulating tumor DNA associated with hepatocellular carcinoma. Proc. Natl Acad. Sci. USA 115, E10925–E10933 (2018).
Wang, B. G. et al. Increased plasma DNA integrity in cancer patients. Cancer Res. 63, 3966–3968 (2003).
Gao, Y. J. et al. Increased integrity of circulating cell-free DNA in plasma of patients with acute leukemia. Clin. Chem. Lab. Med. 48, 1651–1656 (2010).
Yi, X. et al. The feasibility of using mutation detection in ctDNA to assess tumor dynamics. Int. J. Cancer 140, 2642–2647 (2017).
Lo, Y. M. et al. Rapid clearance of fetal DNA from maternal plasma. Am. J. Hum. Genet. 64, 218–224 (1999).
Diehl, F. et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 14, 985–990 (2008).
Scholer, L. V. et al. Clinical implications of monitoring circulating tumor DNA in patients with colorectal cancer. Clin. Cancer Res. 23, 5437–5445 (2017).
Overman, M. J. et al. Use of research biopsies in clinical trials: are risks and benefits adequately discussed? J. Clin. Oncol. 31, 17–22 (2013).
Rothe, F. et al. Plasma circulating tumor DNA as an alternative to metastatic biopsies for mutational analysis in breast cancer. Ann. Oncol. 25, 1959–1965 (2014).
Younossi, Z. M., Teran, J. C., Ganiats, T. G. & Carey, W. D. Ultrasound-guided liver biopsy for parenchymal liver disease: an economic analysis. Dig. Dis. Sci. 43, 46–50 (1998).
Kelly, R. J. et al. Complications and economic burden associated with obtaining tissue for diagnosis and molecular analysis in patients with non-small-cell lung cancer in the United States. J. Oncol. Pract. 15, e717–e727 (2019).
Arnaud, A. Costs and outcomes comparison of tissue and blood based biopsies for the purpose of biomarker testing. Value Health 19, A143–A144 (2016).
Khleif, S. N., Doroshow, J. H. & Hait, W. N. AACR-FDA-NCI cancer biomarkers collaborative consensus report: advancing the use of biomarkers in cancer drug development. Clin. Cancer Res. 16, 3299–3318 (2010).
Ileana Dumbrava, E., Meric-Bernstam, F. & Yap, T. A. Challenges with biomarkers in cancer drug discovery and development. Expert. Opin. Drug. Discov. 13, 685–690 (2018).
Hilton, J. F. et al. Acquisition of metastatic tissue from patients with bone metastases from breast cancer. Breast Cancer Res. Treat. 129, 761–765 (2011).
Holmes, M. G. et al. CT-guided bone biopsies in metastatic castration-resistant prostate cancer: factors predictive of maximum tumor yield. J. Vasc. Interv. Radiol. 28, 1073–1081.e1 (2017).
Wyatt, A. W. et al. Genomic alterations in cell-free DNA and enzalutamide resistance in castration-resistant prostate cancer. JAMA Oncol. 2, 1598–1606 (2016).
De Mattos-Arruda, L. et al. Capturing intra-tumor genetic heterogeneity by de novo mutation profiling of circulating cell-free tumor DNA: a proof-of-principle. Ann. Oncol. 25, 1729–1735 (2014).
Pal, S. K. et al. Evolution of circulating tumor DNA profile from first-line to subsequent therapy in metastatic renal cell carcinoma. Eur. Urol. 72, 557–564 (2017).
Oxnard, G. R. et al. Noninvasive detection of response and resistance in EGFR-mutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin. Cancer Res. 20, 1698–1705 (2014).
Goodall, J. et al. Circulating free DNA to guide prostate cancer treatment with PARP inhibition. Cancer Discov. 7, 1006–1017 (2017).
El Messaoudi, S., Rolet, F., Mouliere, F. & Thierry, A. R. Circulating cell free DNA: preanalytical considerations. Clin. Chim. Acta 424, 222–230 (2013).
Grolz, D. et al. Liquid biopsy preservation solutions for standardized pre-analytical workflows–venous whole blood and plasma. Curr. Pathobiol. Rep. 6, 275–286 (2018).
Okada, S., Yoshimori, M. & Kakizoe, T. Pancreatic cancer: medical aspects. Pancreas 16, 349–354 (1998).
Jen, J., Johnson, C. & Levin, B. Molecular approaches for colorectal cancer screening. Eur. J. Gastroenterol. Hepatol. 10, 213–217 (1998).
Ignatiadis, M. & Dawson, S. J. Circulating tumor cells and circulating tumor DNA for precision medicine: dream or reality? Ann. Oncol. 25, 2304–2313 (2014).
Vogelstein, B. & Kinzler, K. W. Digital PCR. Proc. Natl Acad. Sci. USA 96, 9236–9241 (1999).
Oh, J. E. et al. Detection of low-level KRAS mutations using PNA-mediated asymmetric PCR clamping and melting curve analysis with unlabeled probes. J. Mol. Diagn. 12, 418–424 (2010).
Watanabe, K. et al. EGFR mutation analysis of circulating tumor DNA using an improved PNA-LNA PCR clamp method. Can. Respir. J. 2016, 5297329 (2016).
Spindler, K. L. G., Pallisgaard, N., Vogelius, I. & Jakobsen, A. Quantitative cell-free DNA, KRAS, and BRAF mutations in plasma from patients with metastatic colorectal cancer during treatment with cetuximab and irinotecan. Clin. Cancer Res. 18, 1177–1185 (2012).
Milbury, C. A. et al. Multiplex amplification coupled with COLD-PCR and high resolution melting enables identification of low-abundance mutations in cancer samples with low DNA content. J. Mol. Diagn. 13, 220–232 (2011).
Diehl, F. et al. BEAMing: single-molecule PCR on microparticles in water-in-oil emulsions. Nat. Methods 3, 551–559 (2006).
Sanmamed, M. F. et al. Quantitative cell-free circulating BRAF(V600E) mutation analysis by use of droplet digital PCR in the follow-up of patients with melanoma being treated with BRAF inhibitors. Clin. Chem. 61, 297–304 (2015).
Hindson, B. J. et al. High-throughput droplet digital PCR system for absolute quantitation of DNA copy number. Anal. Chem. 83, 8604–8610 (2011).
Han, X., Wang, J. Y. & Sun, Y. L. Circulating tumor DNA as biomarkers for cancer detection. Genom. Proteom. Bioinf. 15, 59–72 (2017).
Ehlert, T. et al. Establishing PNB-qPCR for quantifying minimal ctDNA concentrations during tumour resection. Sci. Rep. 7, 8876 (2017).
Oxnard, G. R., Paweletz, C. P. & Sholl, L. M. Genomic analysis of plasma cell-free DNA in patients with cancer. JAMA Oncol. 3, 740–741 (2017).
Forshew, T. et al. Noninvasive identification and monitoring of cancer mutations by targeted deep sequencing of plasma DNA. Sci. Transl Med. 4, 136ra68 (2012).
Kinde, I., Wu, J., Papadopoulos, N., Kinzler, K. W. & Vogelstein, B. Detection and quantification of rare mutations with massively parallel sequencing. Proc. Natl Acad. Sci. USA 108, 9530–9535 (2011).
Newman, A. M. et al. An ultrasensitive method for quantitating circulating tumor DNA with broad patient coverage. Nat. Med. 20, 552–558 (2014).
Newman, A. M. et al. Integrated digital error suppression for improved detection of circulating tumor DNA. Nat. Biotechnol. 34, 547–555 (2016).
Leary, R. J. et al. Detection of chromosomal alterations in the circulation of cancer patients with whole-genome sequencing. Sci. Transl Med. 4, 162ra154 (2012).
Diaz, L. A., Sausen, M., Fisher, G. A. & Velculescu, V. E. Insights into therapeutic resistance from whole-genome analyses of circulating tumor DNA. Oncotarget 4, 1856–1857 (2013).
Chan, K. C. A. et al. Cancer genome scanning in plasma: detection of tumor-associated copy number aberrations, single-nucleotide variants, and tumoral heterogeneity by massively parallel sequencing. Clin. Chem. 59, 211–224 (2013).
Leary, R. J. et al. Development of personalized tumor biomarkers using massively parallel sequencing. Sci. Transl Med. 2, 20ra14 (2010).
Schram, A. M., Berger, M. F. & Hyman, D. M. Precision oncology: charting a path forward to broader deployment of genomic profiling. PLoS Med. 14, e1002242 (2017).
Heitzer, E., Ulz, P. & Geigl, J. B. Circulating tumor DNA as a liquid biopsy for cancer. Clin. Chem. 61, 112–123 (2015).
Drago, A., De Ronchi, D. & Serretti, A. Incomplete coverage of candidate genes: a poorly considered bias. Curr. Genomics 8, 476–483 (2007).
van Amerongen, R. A. et al. Next-generation sequencing in NSCLC and melanoma patients: a cost and budget impact analysis. Ecancermedicalscience 10, 684 (2016).
Ahmadzada, T. et al. An update on predictive biomarkers for treatment selection in non-small cell lung cancer. J. Clin. Med. 7, 153 (2018).
Pennell, N. A. et al. Economic impact of next generation sequencing vs sequential single-gene testing modalities to detect genomic alterations in metastatic non-small cell lung cancer using a decision analytic model [abstract]. J. Clin. Oncol. 36 (Suppl. 15), 9031 (2018).
Stenzinger, A. et al. Tumor mutational burden standardization initiatives: recommendations for consistent tumor mutational burden assessment in clinical samples to guide immunotherapy treatment decisions. Genes. Chromosomes Cancer 58, 578–588 (2019).
Rizvi, N. A. et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348, 124–128 (2015).
Hellmann, M. D. et al. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N. Engl. J. Med. 378, 2093–2104 (2018).
Wyatt, A. W. et al. Concordance of circulating tumor DNA and matched metastatic tissue biopsy in prostate cancer. J. Natl Cancer Inst. 109, djx118 (2017).
Lorente, D. et al. Castration-resistant prostate cancer tissue acquisition from bone metastases for molecular analyses. Clin. Genitourin. Cancer 14, 485–493 (2016).
McKay, R. R. et al. Imaging, procedural and clinical variables associated with tumor yield on bone biopsy in metastatic castration-resistant prostate cancer. Prostate Cancer Prostatic Dis. 17, 325–331 (2014).
Vandekerkhove, G. et al. Circulating tumor DNA abundance and potential utility in de novo metastatic prostate cancer. Eur. Urol. 75, 667–675 (2019).
De Laere, B. et al. TP53 outperforms other androgen receptor biomarkers to predict abiraterone or enzalutamide outcome in metastatic castration-resistant prostate cancer. Clin. Cancer Res. 25, 1766–1773 (2019).
Boysen, G. et al. SPOP-mutated/CHD1-deleted lethal prostate cancer and abiraterone sensitivity. Clin. Cancer Res. 24, 5585–5593 (2018).
Nava Rodrigues, D. et al. Immunogenomic analyses associate immunological alterations with mismatch repair defects in prostate cancer. J. Clin. Invest. 128, 4441–4453 (2018).
Mateo, J. et al. DNA-repair defects and olaparib in metastatic prostate cancer. N. Engl. J. Med. 373, 1697–1708 (2015).
Hahn, A. W. et al. Correlation of genomic alterations assessed by next-generation sequencing (NGS) of tumor tissue DNA and circulating tumor DNA (ctDNA) in metastatic renal cell carcinoma (mRCC): potential clinical implications. Oncotarget 8, 33614–33620 (2017).
Barata, P. C. et al. Next-generation sequencing (NGS) of cell-free circulating tumor DNA and tumor tissue in patients with advanced urothelial cancer: a pilot assessment of concordance. Ann. Oncol. 28, 2458–2463 (2017).
Schwaederle, M. C. et al. Utility of genomic assessment of blood-derived circulating tumor DNA (ctDNA) in patients with advanced lung adenocarcinoma. Clin. Cancer Res. 23, 5101–5111 (2017).
Kim, S. T. et al. Prospective blinded study of somatic mutation detection in cell-free DNA utilizing a targeted 54-gene next generation sequencing panel in metastatic solid tumor patients. Oncotarget 6, 40360–40369 (2015).
Mok, T. et al. Detection and dynamic changes of EGFR mutations from circulating tumor DNA as a predictor of survival outcomes in NSCLC patients treated with first-line intercalated erlotinib and chemotherapy. Clin. Cancer Res. 21, 3196–3203 (2015).
Torga, G. & Pienta, K. J. Patient-paired sample congruence between 2 commercial liquid biopsy tests. JAMA Oncol. 4, 868–870 (2018).
Genovese, G. et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N. Engl. J. Med. 371, 2477–2487 (2014).
Mayrhofer, M. et al. Cell-free DNA profiling of metastatic prostate cancer reveals microsatellite instability, structural rearrangements and clonal hematopoiesis. Genome Med. 10, 85 (2018).
Hahn, A. W., Nussenzveig, R. H., Pal, S. K. & Agarwal, N. Blood- and tissue-based tumor genomics: a battle royale or match made in heaven? Ann. Oncol. 28, 2333–2335 (2017).
Maia, M. C. et al. Association of circulating tumor DNA (ctDNA) detection in metastatic renal cell carcinoma (mRCC) with tumor burden. Kidney Cancer 1, 65–70 (2017).
Phallen, J. et al. Direct detection of early-stage cancers using circulating tumor DNA. Sci. Transl Med. 9, eaan2415 (2017).
National Comprehensive Cancer Network. Kidney cancer (version 4.2018). NCCN https://www.nccn.org/professionals/physician_gls/pdf/kidney.pdf (2018).
Grimes, A. D. & Schulz, K. F. Epidemiology 2: compared to what? Finding controls for case-control studies. Lancet 365, 1429–1433 (2005).
Janzen, N. K., Kim, H. L., Figlin, R. A. & Belldegrun, A. S. Surveillance after radical or partial nephrectomy for localized renal cell carcinoma and management of recurrent disease. Urol. Clin. North Am. 30, 843–852 (2003).
Al-Qassab, U. et al. PNFBA-12 liquid biopsy for renal cell carcinoma. J. Urol. 197, e913–e914 (2017).
Hauser, S. et al. Cell-free circulating DNA: diagnostic value in patients with renal cell cancer. Anticancer Res. 30, 2785–2789 (2010).
Skrypkina, I. et al. Concentration and methylation of cell-free DNA from blood plasma as diagnostic markers of renal cancer. Dis. Markers 2016, 3693096 (2016).
Tie, J. et al. Circulating tumor DNA as an early marker of therapeutic response in patients with metastatic colorectal cancer. Ann. Oncol. 26, 1715–1722 (2015).
Beaver, J. A. et al. Detection of cancer DNA in plasma of patients with early-stage breast cancer. Clin. Cancer Res. 20, 2643–2650 (2014).
Chen, Z. et al. Analysis of cancer mutation signatures in blood by a novel ultra-sensitive assay: monitoring of therapy or recurrence in non-metastatic breast cancer. PLoS One 4, e7220 (2009).
Wan, J., Zhu, L., Jiang, Z. & Cheng, K. Monitoring of plasma cell-free DNA in predicting postoperative recurrence of clear cell renal cell carcinoma. Urol. Int. 91, 273–278 (2013).
Lu, H. et al. Diagnostic and prognostic potential of circulating cell-free genomic and mitochondrial DNA fragments in clear cell renal cell carcinoma patients. Clin. Chim. Acta 452, 109–119 (2016).
Thompson, J. C. et al. Detection of therapeutically targetable driver and resistance mutations in lung cancer patients by next-generation sequencing of cell-free circulating tumor DNA. Clin. Cancer Res. 22, 5772–5782 (2016).
Sacher, A. G. et al. Prospective validation of rapid plasma genotyping for the detection of EGFR and KRAS mutations in advanced lung cancer. JAMA Oncol. 2, 1014–1022 (2016).
Oxnard, G. R. et al. Association between plasma genotyping and outcomes of treatment with osimertinib (AZD9291) in advanced non-small-cell lung cancer. J. Clin. Oncol. 34, 3375–3382 (2016).
Thress, K. S. et al. EGFR mutation detection in ctDNA from NSCLC patient plasma: a cross-platform comparison of leading technologies to support the clinical development of AZD929. Lung Cancer 90, 509–515 (2015).
Dawson, S. J. et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N. Engl. J. Med. 368, 1199–1209 (2013).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499, 43–49 (2013).
Ferrero, A. et al. Understanding, justifying, and optimizing radiation exposure for CT imaging in nephrourology. Nat. Rev. Urol. 16, 231–244 (2019).
Lee, J. H. et al. Association between circulating tumor DNA and pseudoprogression in patients with metastatic melanoma treated with anti-programmed cell death 1 antibodies. JAMA Oncol. 4, 717–721 (2018).
Dizman, N. et al. Exceptional response to nivolumab rechallenge in metastatic renal cell carcinoma with parallel changes in genomic profile. Eur. Urol. 73, 308–310 (2018).
Maia, M. C., Dizman, N., Salgia, M. & Pal, S. K. Therapeutic sequencing in metastatic renal cell carcinoma. Kidney Cancer 1, 15–29 (2017).
Rini, B. I. et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N. Engl. J. Med. 380, 1116–1127 (2019).
Motzer, R. J. et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N. Engl. J. Med. 380, 1103–1115 (2019).
Brott, T. Treatment of acute ischemic stroke. N. Engl. J. Med. 343, 710–722 (2000).
Choueiri, T. K. et al. Cabozantinib versus sunitinib as initial targeted therapy for patients with metastatic renal cell carcinoma of poor or intermediate risk: the alliance A031203 CABOSUN trial. J. Clin. Oncol. 35, 591–597 (2017).
Motzer, R. J. et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N. Engl. J. Med. 378, 1277–1290 (2018).
Snyder, A. et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N. Engl. J. Med. 371, 2189–2199 (2014).
Gandara, D. R. et al. Blood-based tumor mutational burden as a predictor of clinical benefit in non-small-cell lung cancer patients treated with atezolizumab. Nat. Med.24, 1441–1448 (2018).
Rosenberg, J. E. et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 387, 1909–1920 (2016).
Ferrone, S. & Gameiro, S. R. Chemotherapy-induced immunogenic modulation of tumor cells enhances killing by cytotoxic T lymphocytes and is distinct from immunogenic cell death. Int. J. Cancer 133, 624–636 (2013).
Maia, M. C., Almeida, L., Bergerot, P. G., Dizman, N. & Pal, S. K. Relationship of tumor mutational burden (TMB) to immunotherapy response in metastatic renal cell carcinoma (mRCC) [abstract]. J. Clin. Oncol. 36 (Suppl. 6), 662 (2018).
Khagi, Y. et al. Hypermutated circulating tumor DNA: correlation with response to checkpoint inhibitor-based immunotherapy. Clin. Cancer Res. 23, 5729–5736 (2017).
Wang, Z. et al. Assessment of blood tumor mutational burden as a potential biomarker for immunotherapy in patients with non-small cell lung cancer with use of a next-generation sequencing cancer gene panel. JAMA Oncol. 5, 696–702 (2019).
Jordan, B. & Meeks, J. J. T1 bladder cancer: current considerations for diagnosis and management. Nat. Rev. Urol. 16, 23–34 (2019).
Chang, S. S. et al. Treatment of non-metastatic muscle-invasive bladder cancer: AUA/ASCO/ASTRO/SUO guideline. J. Urol. 198, 552–559 (2017).
Babjuk, M. et al. EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: update 2016. Eur. Urol. 71, 447–461 (2017).
Birkenkamp-Demtroder, K. et al. Genomic alterations in liquid biopsies from patients with bladder cancer. Eur. Urol. 70, 75–82 (2016).
Christensen, E. et al. Liquid biopsy analysis of FGFR3 and PIK3CA hotspot mutations for disease surveillance in bladder cancer. Eur. Urol. 71, 961–969 (2017).
Birkenkamp-Demtröder, K. et al. Monitoring treatment response and metastatic relapse in advanced bladder cancer by liquid biopsy analysis. Eur. Urol. 73, 535–540 (2017).
Todenhöfer, T. et al. Using liquid biopsy to assess the genomic landscape of metastatic urothelial carcinoma [abstract 385]. Eur. Urol. Suppl. 16, e677–e678 (2017).
Bellmunt, J., Powles, T. & Vogelzang, N. J. A review on the evolution of PD-1/PD-L1 immunotherapy for bladder cancer: the future is now. Cancer Treat. Rev. 54, 58–67 (2017).
Rosenberg, J. E. et al. Pivotal trial of enfortumab vedotin in urothelial carcinoma after platinum and anti-programmed death 1/programmed death ligand 1 therapy. J. Clin. Oncol. 37, 2592–2600 (2019).
Loriot, Y. et al. Erdafitinib (ERDA; JNJ-42756493), a pan-fibroblast growth factor receptor (FGFR) inhibitor, in patients (pts) with metastatic or unresectable urothelial carcinoma (mUC) and FGFR alterations (FGFRa): phase 2 continuous versus intermittent dosing [abstract]. J. Clin. Oncol. 36 (Suppl. 6), 411 (2018).
Challita-Eid, P. M. et al. Enfortumab vedotin antibody-drug conjugate targeting nectin-4 is a highly potent therapeutic agent in multiple preclinical cancer models. Cancer Res. 76, 3003–3013 (2016).
Iyer, G. et al. Genome sequencing identifies a basis for everolimus sensitivity. Science 338, 221 (2012).
Choudhury, N. J. et al. Afatinib activity in platinum-refractory metastatic urothelial carcinoma in patients with ERBB alterations. J. Clin. Oncol. 34, 2165–2171 (2016).
Plimack, E. R. et al. Defects in DNA repair genes predict response to neoadjuvant cisplatin-based chemotherapy in muscle-invasive bladder cancer. Eur. Urol. 68, 959–967 (2015).
Van Allen, E. M. et al. Somatic ERCC2 mutations correlate with cisplatin sensitivity in muscle-invasive urothelial carcinoma. Cancer Discov. 4, 1140–1153 (2014).
Necchi, A. et al. Pembrolizumab as neoadjuvant therapy before radical cystectomy in patients with muscle-invasive urothelial bladder carcinoma (PURE-01): an open-label, single-arm, phase II study. J. Clin. Oncol. 36, 3353–3360 (2018).
Agarwal, N. et al. Characterization of metastatic urothelial carcinoma via comprehensive genomic profiling of circulating tumor DNA. Cancer 124, 2115–2124 (2018).
Grivas, P. et al. Circulating tumor (ct)-DNA alterations in advanced urothelial carcinoma: association with outcomes and evolution with therapy [abstract]. J. Clin. Oncol. 35, 334 (2017).
Hussain, M. et al. PROfound: phase III study of olaparib versus enzalutamide or abiraterone for metastatic castration-resistant prostate cancer (mCRPC) with homologous recombination repair (HRR) gene alterations [abstract LBA12_PR]. Ann. Oncol. 30 (Suppl. 5), v881–v882 (2019).
Golan, T. et al. Maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. N. Engl. J. Med. 381, 317–327 (2019).
Anguera, G. & Majem, M. BRAF inhibitors in metastatic non-small cell lung cancer. J. Thorac. Dis. 10, 589–592 (2018).
Planchard, D. et al. Dabrafenib plus trametinib in patients with previously treated BRAF(V600E)-mutant metastatic non-small cell lung cancer: an open-label, multicentre phase 2 trial. Lancet Oncol. 17, 984–993 (2016).
Flaherty, K. T. et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N. Engl. J. Med. 367, 107–114 (2012).
Alexandrov, L. B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Jiang, T. et al. Accurate measurement of tumor mutation burden in liquid biopsy (bTMB) using a 500 gene panel [abstract 163P]. Ann. Oncol. 29 (Suppl. 8), viii51 (2018).
Quinn, K. et al. Development and analytical validation of a plasma-based tumor mutational burden (TMB) score from next-generation sequencing panels [abstract 131P]. Ann. Oncol. 29 (Suppl. 8), viii41 (2018).
Raja, R. et al. Early reduction in ctDNA predicts survival in patients with lung and bladder cancer treated with durvalumab. Clin. Cancer Res. 24, 6212–6222 (2018).
Global Burden of Disease Collaboration Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the Global Burden of Disease Study. JAMA Oncol. 3, 524–548 (2017).
Abdollah, F. et al. Comparison of mortality outcomes after radical prostatectomy versus radiotherapy in patients with localized prostate cancer: a population-based analysis. Int. J. Urol. 19, 836–844 (2012).
Fleshner, K., Carlsson, S. V. & Roobol, M. J. The effect of the USPSTF PSA screening recommendation on prostate cancer incidence patterns in the USA. Nat. Rev. Urol. 14, 26–37 (2017).
Pound, C. R. et al. Natural history of progression after PSA elevation following radical prostatectomy. JAMA 281, 1591–1597 (1999).
Schwarzenbach, H. et al. Cell-free tumor DNA in blood plasma as a marker for circulating tumor cells in prostate cancer. Clin. Cancer Res. 15, 1032–1038 (2009).
Chun, F. K. et al. Circulating tumour-associated plasma DNA represents an independent and informative predictor of prostate cancer. BJU Int. 98, 544–548 (2006).
Altimari, A. et al. Diagnostic role of circulating free plasma DNA detection in patients with localized prostate cancer. Am. J. Clin. Pathol. 129, 756–762 (2008).
D’Amico, A. V. et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280, 969–974 (1998).
ICECaP Working Group The development of intermediate clinical endpoints in cancer of the prostate (ICECaP). J. Natl Cancer Inst. 107, djv261 (2015).
Spratt, D. E. et al. Development and validation of a novel integrated clinical-genomic risk group classification for localized prostate cancer. J. Clin. Oncol. 36, 581–590 (2018).
Gorin, M. A. et al. Circulating tumour cells as biomarkers of prostate, bladder, and kidney cancer. Nat. Rev. Urol. 14, 90–97 (2017).
Heller, G. et al. Circulating tumor cell number as a response measure of prolonged survival for metastatic castration-resistant prostate cancer: a comparison with prostate-specific antigen across five randomized phase III clinical trials. J. Clin. Oncol. 36, 572–580 (2018).
Scher, H. I. et al. Circulating tumor cell biomarker panel as an individual-level surrogate for survival in metastatic castration-resistant prostate cancer. J. Clin. Oncol. 33, 1348–1355 (2015).
Scher, H. I. et al. Clinical outcome of metastatic castration-resistant prostate cancer (mCRPC) patients (pts) with a post-treatment circulating tumor cell (CTC) of 0 vs CTC>0: post hoc analysis of COU-AA-301 [abstract]. J. Clin. Oncol. 35 (Suppl. 15), 5015 (2017).
Lorente, D. et al. Decline in circulating tumor cell count and treatment outcome in advanced prostate cancer. Eur. Urol. 70, 985–992 (2016).
Antonarakis, E. S. et al. Clinical significance of androgen receptor splice variant-7 mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J. Clin. Oncol. 35, 2149–2156 (2017).
Kohli, M. et al. Prognostic association of plasma cell-free DNA-based androgen receptor amplification and circulating tumor cells in pre-chemotherapy metastatic castration-resistant prostate cancer patients. Prostate Cancer Prostatic Dis. 21, 411–418 (2018).
Annala, M. et al. Circulating tumor DNA genomics correlate with resistance to abiraterone and enzalutamide in prostate cancer. Cancer Discov. 8, 444–457 (2018).
Choudhury, A. D. et al. Tumor fraction in cell-free DNA as a biomarker in prostate cancer. JCI Insight 3, e122109 (2018).
Costelloe, C. M., Chuang, H. H., Madewell, J. E. & Ueno, N. T. Cancer response criteria and bone metastases: RECIST 1.1, MDA and PERCIST. J. Cancer 1, 80–92 (2010).
Wyatt, A. W. et al. Genomic alterations in circulating tumor DNA (ctDNA) are associated with clinical outcomes in treatment-naive metastatic castration-resistant prostate cancer (mCRPC) patients commencing androgen receptor (AR)-targeted therapy [abstract 56P]. Ann. Oncol. 27 (Suppl. 6), vi17 (2016).
Azad, A. A. et al. Androgen receptor gene aberrations in circulating cell-free DNA: biomarkers of therapeutic resistance in castration-resistant prostate cancer. Clin. Cancer Res. 21, 2315–2324 (2015).
Antonarakis, E. S. et al. Germline DNA-repair gene mutations and outcomes in men with metastatic castration-resistant prostate cancer receiving first-line abiraterone and enzalutamide. Eur. Urol. 74, 218–225 (2018).
Hussain, M. et al. Targeting androgen receptor and DNA repair in metastatic castration-resistant prostate cancer: results from NCI 9012. J. Clin. Oncol. 36, 991–999 (2018).
Cheng, H. H., Pritchard, C. C., Boyd, T., Nelson, P. S. & Montgomery, B. Biallelic inactivation of BRCA2 in platinum-sensitive metastatic castration-resistant prostate cancer. Eur. Urol. 69, 992–995 (2016).
Quigley, D. et al. Analysis of circulating cell-free DNA identifies multiclonal heterogeneity of BRCA2 reversion mutations associated with resistance to PARP inhibitors. Cancer Discov. 7, 999–1005 (2017).
Hyman, D. M. et al. Vemurafenib in multiple nonmelanoma cancers with BRAF V600 mutations. N. Engl. J. Med. 373, 726–736 (2015).
Velho, P. I. et al. Efficacy of radium-223 in bone-metastatic castration-resistant prostate cancer with and without homologous repair gene defects. Eur. Urol. 76, 170–176 (2019).
Tannock, I. F. & Hickman, J. A. Limits to precision cancer medicine. N. Engl. J. Med. 376, 96–97 (2017).
Meric-Bernstam, F. et al. Feasibility of large-scale genomic testing to facilitate enrollment onto genomically matched clinical trials. J. Clin. Oncol. 33, 2753–2762 (2015).
Stockley, T. L. et al. Molecular profiling of advanced solid tumors and patient outcomes with genotype-matched clinical trials: the Princess Margaret IMPACT/COMPACT trial. Genome Med. 8, 109 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02535351 (2018).
Soares, A., Maia, M. C., Vidigal, F. & Marques Monteiro, F. S. Cytoreductive nephrectomy for metastatic renal cell carcinoma: how to apply new evidence in clinical practice. Oncology 98, 1–9 (2020).
Mejean, A. et al. Sunitinib alone or after nephrectomy in metastatic renal-cell carcinoma. N. Engl. J. Med. 379, 417–427 (2018).
Bex, A. et al. Comparison of immediate vs deferred cytoreductive nephrectomy in patients with synchronous metastatic renal cell carcinoma receiving sunitinib: the SURTIME Randomized Clinical Trial. JAMA Oncol. 5, 164–170 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03469713 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03414827 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03091192 (2019).
Nandagopal, L., Sonpavde, G. P. & Agarwal, N. Investigational MET inhibitors to treat renal cell carcinoma. Expert. Opin. Investig. Drugs 28, 851–860 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02546661 (2020).
Loriot, Y. et al. Erdafitinib in locally advanced or metastatic urothelial carcinoma. N. Engl. J. Med. 381, 338–348 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03263039 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03385655 (2020).
Mateo, J. et al. Olaparib in patients with metastatic castration-resistant prostate cancer with DNA repair gene aberrations (TOPARP-B): a multicentre, open-label, randomised, phase 2 trial. Lancet Oncol. 21, 162–174 (2020).
de Bono, J. S. et al. Randomized phase II study evaluating akt blockade with ipatasertib, in combination with abiraterone, in patients with metastatic prostate cancer with and without PTEN loss. Clin. Cancer Res. 25, 928–936 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02952534 (2020).
Torgovnick, A. & Schumacher, B. DNA repair mechanisms in cancer development and therapy. Front. Genet. 6, 157 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02975934 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03228810 (2019).
James, N. D. et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 6736, 1–15 (2015).
Decaestecker, K. et al. Surveillance or metastasis-directed Therapy for OligoMetastatic Prostate cancer recurrence (STOMP): study protocol for a randomized phase II trial. BMC Cancer 14, 671 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02826772 (2019).
Dellis, A. E. & Papatsoris, A. G. Perspectives on the current and emerging chemical androgen receptor antagonists for the treatment of prostate cancer. Expert. Opin. Pharmacother. 20, 163–172 (2019).
Conteduca, V. et al. Androgen receptor gene status in plasma DNA associates with worse outcome on enzalutamide or abiraterone for castration-resistant prostate cancer: a multi-institution correlative biomarker study. Ann. Oncol. 28, 1508–1516 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03677414 (2018).
Chen, Z., Wang, L., Wang, Q. & Li, W. Histone modifications and chromatin organization in prostate cancer. Epigenomics 2, 551–560 (2010).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03522064 (2019).
Chuu, C. P., Hiipakka, R. A., Fukuchi, J., Kokontis, J. M. & Liao, S. Androgen causes growth suppression and reversion of androgen-independent prostate cancer xenografts to an androgen-stimulated phenotype in athymic mice. Cancer Res. 65, 2082–2084 (2005).
Teply, B. A. et al. Bipolar androgen therapy in men with metastatic castration-resistant prostate cancer after progression on enzalutamide: an open-label, phase 2, multicohort study. Lancet Oncol. 19, 76–86 (2018).
Chen, J., Joshua, A. M., Denmeade, S. R., Antonarakis, E. S. & Crumbaker, M. High dose testosterone in men with metastatic castrate-resistant prostate cancer (mCRPC) and homologous recombination deficiency (HRD) [abstract]. J. Clin. Oncol. 37 (Suppl. 15), TPS5095 (2019).
Haffner, M. C. et al. Androgen-induced TOP2B-mediated double-strand breaks and prostate cancer gene rearrangements. Nat. Genet. 42, 668–675 (2010).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03644303 (2019).
Palma, D. A. et al. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet 393, 2051–2058 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02735252 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02889978 (2019).
Liu, M. C. et al. Plasma cell-free DNA (cfDNA) assays for early multi-cancer detection: the circulating cell-free genome atlas (CCGA) study [abstract 50O]. Ann. Oncol. 29 (Suppl. 8), viii14 (2018).
Author information
Authors and Affiliations
Contributions
M.C.M. and M.S. researched data for and wrote the article. M.C.M. and S.K.P. made substantial contributions to discussion of content and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.C.M. has received speaker’s fees from Pfizer, Astellas, AstraZeneca and BMS, educational meeting sponsorship from Astellas, Jansen, BMS, Libbs, Roche, Ipsen, MSD, Novartis and Bayer, and consulting fees from AstraZeneca and MSD. S.K.P. has received consulting fees from Genentech, Aveo, Eisai, Roche, Pfizer, Novartis, Exelixis, Ipsen, BMS and Astellas. M.S. declares no competing interests.
Additional information
Peer review information
Nature Reviews Urology thanks M. Annala, E. Christensen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Whole-genome sequencing
-
A laboratory process that is used to determine nearly all nucleotides of an individual’s complete DNA sequence, including coding and non-coding sequences.
- Predictive biomarker
-
A biological variable that is associated with a response or lack of response to a particular agent or therapy.
- Sanger sequencing
-
A low-throughput method used to determine a portion of the nucleotide sequence of an individual’s genome. This technique uses PCR amplification of genetic regions of interest followed by sequencing of PCR products.
- Deep sequencing
-
It refers to sequencing a particular genomic region multiple times, sometimes hundreds or even thousands of times.
- Next-generation sequencing
-
(NGS). A high-throughput method used to determine a portion of the nucleotide sequence of an individual’s genome. This technique utilizes DNA sequencing technologies that are capable of processing multiple DNA sequences in parallel.
- Whole-exome sequencing
-
(WES). A laboratory process that is used to determine the nucleotide sequence primarily of the exonic (or protein-coding) regions of an individual’s genome and related sequences, which represents approximately 1% of the complete DNA sequence.
- Tumour mutational burden
-
(TMB). The total number of mutations found in tumour DNA of a particular patient. It has been shown to correlate with response to immunotherapy (checkpoint inhibitors). In general, most authors consider a high TMB as ≥10 mutations per megabase (mut/Mb) and low TMB anything below this.
- Biomarker
-
Any biological variable that can be measured and whose presence indicates a disease state and/or disease severity cut-off, although there is no standard definition.
- Intrapatient spatial heterogeneity
-
The expected differences in DNA sequences across different metastatic sites in a given patient.
- Variant allelic fractions
-
(VAFs). The incidence of a gene variant (allele) in a person. Alleles are variant forms of a gene that are located at the same position (genetic locus). It is calculated by dividing the number of times the allele is observed by the total number of copies of all the alleles at that particular genetic locus and can be expressed as a percentage or fraction.
- Pseudoprogression
-
Radiologically, it is defined as the development of a new or enlarging area (or areas) in the absence of true tumour growth, which usually subsides or stabilizes without a change in therapy. It can occur weeks to months after finishing radiation therapy (most commonly to a brain tumour, for example) but has been shown to occur after initiation of immunotherapies (such as checkpoint inhibitors). The potential mechanisms behind pseudoprogression are related to tumour necrosis with associated oedema (especially after radiation exposure) or immune cell infiltration of the tumour (in the case of treatment with immunotherapies).
- Prognostic biomarker
-
A biological variable associated with clinical outcomes regardless of therapy received.
- Circulating Cell-Free Genome Atlas
-
(CCGA). It is an ongoing international prospective, observational study developed to characterize the landscape of genomic cancer alterations in the blood of people with and without cancer. The study includes both people with a diagnosis of cancer and people without a known diagnosis of cancer. The objectives of the study are to discover, develop and validate a blood test for early cancer detection.
Rights and permissions
About this article
Cite this article
Maia, M.C., Salgia, M. & Pal, S.K. Harnessing cell-free DNA: plasma circulating tumour DNA for liquid biopsy in genitourinary cancers. Nat Rev Urol 17, 271–291 (2020). https://doi.org/10.1038/s41585-020-0297-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-020-0297-9
This article is cited by
-
Assessment of a cancer genomic profile test for patients with metastatic breast cancer
Scientific Reports (2022)
-
Genomic profiling of Chinese patients with urothelial carcinoma
BMC Cancer (2021)
-
Building confidence in circulating tumour DNA assays for metastatic castration-resistant prostate cancer
Nature Reviews Urology (2021)
-
Clinical implications of genomic alterations in metastatic prostate cancer
Prostate Cancer and Prostatic Diseases (2021)