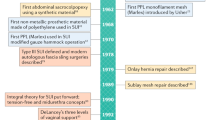
Abstract
Polypropylene (PPL) mesh is widely used in pelvic floor reconstructive surgery for prolapse and stress urinary incontinence. However, some women, particularly those treated using transvaginal PPL mesh placement for prolapse, experience intractable pain and mesh exposure or extrusion. Explanted tissue from patients with complications following transvaginal implantation of mesh is typified by a dense fibrous capsule with an immune cell-rich infiltrate, suggesting that the host immune response has a role in transvaginal PPL mesh complications through the separate contributions of the host (patient), the biological niche within which the material is implanted and biomaterial properties of the mesh. This immune response might be strongly influenced by both the baseline inflammatory status of the patient, surgical technique and experience, and the unique hormonal, immune and microbial tissue niche of the vagina. Mesh porosity, surface area and stiffness also might have an effect on the immune and tissue response to transvaginal mesh placement. Thus, a regulatory pathway is needed for mesh development that recognizes the roles of host and biological factors in driving the immune response to mesh, as well as mandatory mesh registries and the longitudinal surveillance of patients.
Key points
-
Current evidence suggests that risk factors for developing complications following transvaginal mesh placement include pro-inflammatory conditions such as patient age, smoking and diabetes, as well as surgical technique and experience.
-
Explanted mesh following transvaginal pelvic organ prolapse repair from women with complications shows poorly regulated inflammation with a fibrous capsule and a dense cellular infiltrate of predominantly macrophages.
-
Differences in the abdominal and vaginal biological niche probably contribute to dissimilar immune responses to polypropylene mesh and consequently the extent to which mesh can integrate.
-
Mesh with large pore diameters, low surface area and low local stiffness might be less likely to induce an adverse foreign body response, but this supposition has not been evaluated longitudinally.
-
A lack of transparency in pelvic floor preclinical testing and post-marketing surveillance are regulatory failings that have probably contributed to the inappropriate repurposing of meshes for some patient groups.
-
The ability to assess factors driving negative outcomes will rely on longitudinal studies to ascertain baseline drivers of tissue response in women who develop complications and those who do not.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wu, J. M., Matthews, C. A., Conover, M. M., Pate, V. & Jonsson Funk, M. Lifetime risk of stress urinary incontinence or pelvic organ prolapse surgery. Obstet. Gynecol. 123, 1201–1206 (2014).
Smith, F. J., Holman, C. D. J., Moorin, R. E. & Tsokos, N. Lifetime risk of undergoing surgery for pelvic organ prolapse. Obstet. Gynecol. 116, 1096–1100 (2010).
Haylen, B. T. et al. An international urogynecological association (IUGA)/international continence society (ICS) joint report on the terminology for female pelvic floor dysfunction. Neurourol. Urodyn. 29, 4–20 (2010).
Jelovsek, J. E. et al. Effect of uterosacral ligament suspension vs sacrospinous ligament fixation with or without perioperative behavioral therapy for pelvic organ vaginal prolapse on surgical outcomes and prolapse symptoms at 5 years in the OPTIMAL Randomized Clinical Trial. JAMA 319, 1554 (2018).
Lapitan, M. C. M., Cody, J. D. & Mashayekhi, A. Open retropubic colposuspension for urinary incontinence in women. Cochrane Database Syst. Rev. 2, CD002912 (2017).
Joint Writing Group of the American Urogynecologic Society & the International Urogynecological Association. Joint report on terminology for surgical procedures to treat pelvic organ prolapse. Female Pelvic Med. Reconstr. Surg. 26, 173–201 (2020).
Chapple, C. R. et al. Consensus statement of the European Urology Association and the European Urogynaecological Association on the use of implanted materials for treating pelvic organ prolapse and stress urinary incontinence. Eur. Urol. 72, 424–431 (2017).
Barone, W. R., Abramowitch, S. D. & Moalli, P. A. in Host Response to Biomaterials Ch. 13 (ed. Badylak, S. F.) 375–423 (Academic, 2015).
Chu, C. C. in Biotextiles Medical Implants (ed. King, M. W.) 275–334 (Woodhead, 2013).
Brown, C. & Finch, J. Which mesh for hernia repair? Ann. R. Coll. Surg. Engl. 92, 272–278 (2010).
Mangir, N., Roman, S., Chapple, C. R. & MacNeil, S. Complications related to use of mesh implants in surgical treatment of stress urinary incontinence and pelvic organ prolapse: infection or inflammation? World J. Urol. 38, 73–80 (2020).
Mangir, N., Aldemir Dikici, B., Chapple, C. R. & MacNeil, S. Landmarks in vaginal mesh development: polypropylene mesh for treatment of SUI and POP. Nat. Rev. Urol. 16, 675–689 (2019).
Balsamo, R. et al. Sacrocolpopexy with polyvinylidene fluoride mesh for pelvic organ prolapse: mid term comparative outcomes with polypropylene mesh. Eur. J. Obstet. Gynecol. Reprod. Biol. 220, 74–78 (2018).
Barski, D. et al. Transvaginal PVDF-mesh for cystocele repair: a cohort study. Int. J. Surg. 39, 249–254 (2017).
Kawaguchi, S. et al. Transvaginal polytetrafluoroethylene mesh surgery for pelvic organ prolapse: 1-year clinical outcomes. Int. J. Urol. 28, 268–272 (2021).
Ford, A. A., Rogerson, L., Cody, J. D., Aluko, P. & Ogah, J. A. Mid-urethral sling operations for stress urinary incontinence in women. Cochrane Database Syst. Rev. 1, CD006375 (2017).
Glazener, C. M. et al. Mesh, graft, or standard repair for women having primary transvaginal anterior or posterior compartment prolapse surgery: two parallel-group, multicentre, randomised, controlled trials (PROSPECT). Lancet 389, 381–392 (2017).
Welk, B., Al-Hothi, H. & Winick-Ng, J. Removal or revision of vaginal mesh used for the treatment of stress urinary incontinence. JAMA Surg. 150, 1167 (2015).
Morling, J. R. et al. Adverse events after first, single, mesh and non-mesh surgical procedures for stress urinary incontinence and pelvic organ prolapse in Scotland, 1997–2016: a population-based cohort study. Lancet 389, 629–640 (2017).
Keltie, K. et al. Complications following vaginal mesh procedures for stress urinary incontinence: an 8 year study of 92,246 women. Sci. Rep. 7, 12015 (2017).
Maher, C. et al. Surgery for women with apical vaginal prolapse. Cochrane Database Syst. Rev. 10, CD012376 (2016).
Mucowski, S. J., Jurnalov, C. & Phelps, J. Y. Use of vaginal mesh in the face of recent FDA warnings and litigation. Am. J. Obstet. Gynecol. 203, 103.e1–103.e4 (2010).
Food and Drug Administration. Serious complications associated with transvaginal placement of surgical mesh for pelvic organ prolapse: FDA safety communication (FDA, 2011).
Sedrakyan, A., Chughtai, B. & Mao, J. Regulatory warnings and use of surgical mesh in pelvic organ prolapse. JAMA Intern. Med. 176, 275 (2016).
Scottish Independent Review. Scottish Independent Review of the use, safety and efficacy of transvaginal mesh implants in the treatment of stress urinary incontinence and pelvic organ prolapse in women (2017).
Wall, L. L. & Brown, D. The perils of commercially driven surgical innovation. Am. J. Obstet. Gynecol. 202, 30.e1–4 (2010).
Danish Medicines Agency. Withdrawal of vaginal mesh. Laegemiddelstyrelsen https://laegemiddelstyrelsen.dk/en/news/2019/withdrawal-of-vaginal-mesh (2019).
Gardner, A. B., Lee, S. K. C., Woods, E. C. & Acharya, A. P. Biomaterials-based modulation of the immune system. Biomed. Res. Int. 2013, 1–7 (2013).
Nolfi, A. L. et al. Host response to synthetic mesh in women with mesh complications. Am. J. Obstet. Gynecol. 215, 206.e1–206.e8 (2016).
Kelly, M., Macdougall, K., Olabisi, O. & McGuire, N. In vivo response to polypropylene following implantation in animal models: a review of biocompatibility. Int. Urogynecol. J. 28, 171–180 (2017).
Brown, B. N. & Badylak, S. F. Expanded applications, shifting paradigms and an improved understanding of host–biomaterial interactions. Acta Biomater. 9, 4948–4955 (2013).
de Almeida, S. H. M., Rodrigues, M. A. F., Gregório, É., Crespígio, J. & Moreira, H. A. Influence of sling material on inflammation and collagen deposit in an animal model. Int. J. Urol. 14, 1040–1043 (2007).
Faulk, D. M. et al. ECM hydrogel coating mitigates the chronic inflammatory response to polypropylene mesh. Biomaterials 35, 8585–8595 (2014).
Mariani, E., Lisignoli, G., Borzì, R. M. & Pulsatelli, L. Biomaterials: foreign bodies or tuners for the immune response? Int. J. Mol. Sci. 20, 636 (2019).
Abed, H. et al. Incidence and management of graft erosion, wound granulation, and dyspareunia following vaginal prolapse repair with graft materials: a systematic review. Int. Urogynecol. J. 22, 789–798 (2011).
Olsen, A., Smith, V., Bergstrom, J., Colling, J. & Clark, A. Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet. Gynecol. 89, 501–506 (1997).
Milani, A. L., Damoiseaux, A., IntHout, J., Kluivers, K. B. & Withagen, M. I. J. Long-term outcome of vaginal mesh or native tissue in recurrent prolapse: a randomized controlled trial. Int. Urogynecol. J. 29, 847–858 (2018).
MacDonald, S., Terlecki, R., Costantini, E. & Badlani, G. Complications of transvaginal mesh for pelvic organ prolapse and stress urinary incontinence: tips for prevention, recognition, and management. Eur. Urol. Focus. 2, 260–267 (2016).
Nygaard, I. et al. Long-term outcomes following abdominal sacrocolpopexy for pelvic organ prolapse. JAMA 309, 2016–2024 (2013).
Izett-Kay, M. L. et al. Long-term mesh complications and reoperation after laparoscopic mesh sacrohysteropexy: a cross-sectional study. Int. Urogynecol. J. 31, 2595–2602 (2020).
Fall, M. et al. EAU guidelines on chronic pelvic pain. Eur. Urol. 57, 35–48 (2010).
Breivik, H., Collett, B., Ventafridda, V., Cohen, R. & Gallacher, D. Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur. J. Pain 10, 287–333 (2006).
Maher, C. et al. Transvaginal mesh or grafts compared with native tissue repair for vaginal prolapse. Cochrane Database Syst. Rev. 2, CD012079 (2016).
Caquant, F. et al. Safety of trans vaginal mesh procedure: retrospective study of 684 patients. J. Obstet. Gynaecol. Res. 34, 449–456 (2008).
Clavé, A. et al. Polypropylene as a reinforcement in pelvic surgery is not inert: comparative analysis of 100 explants. Int. Urogynecol. J. 21, 261–270 (2010).
Artsen, A. M. et al. Mesh induced fibrosis: the protective role of T regulatory cells. Acta Biomater. 96, 203–210 (2019).
Goodall, E. J., Cartwright, R., Stratta, E. C., Jackson, S. R. & Price, N. Outcomes after laparoscopic removal of retropubic midurethral slings for chronic pain. Int. Urogynecol. J. 30, 1323–1328 (2019).
Barski, D. & Deng, D. Y. Management of mesh complications after SUI and POP repair: review and analysis of the current literature. Biomed Res. Int. 2015, 831285 (2015).
Javadian, P. & O’Leary, D. Vaginally placed meshes: a review of their complications, risk factors, and management. Curr. Obstet. Gynecol. Rep. 4, 96–101 (2015).
Sirls, L. T. et al. Exploring predictors of mesh exposure after vaginal prolapse repair. Female Pelvic Med. Reconstr. Surg. 19, 206–209 (2013).
Elmér, C. et al. Risk factors for mesh complications after trocar guided transvaginal mesh kit repair of anterior vaginal wall prolapse. Neurourol. Urodyn. 31, 1165–1169 (2012).
Cundiff, G. W. et al. Risk factors for mesh/suture erosion following sacral colpopexy. Am. J. Obstet. Gynecol. 199, 688.e1–688.e5 (2008).
Deng, T., Liao, B., Luo, D., Shen, H. & Wang, K. Risk factors for mesh erosion after female pelvic floor reconstructive surgery: a systematic review and meta-analysis. BJU Int. 117, 323–343 (2016).
Kokanali, M. K. et al. Risk factors for mesh erosion after vaginal sling procedures for urinary incontinence. Eur. J. Obstet. Gynecol. Reprod. Biol. 177, 146–150 (2014).
Rao, A., Avula, M. & Grainger, D. in Host Response to Biomaterials Ch. 11 (ed. Badylak, S. F.) 269–313 (Academic, 2015).
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Wilkins, J., Ghosh, P., Vivar, J., Chakraborty, B. & Ghosh, S. Exploring the associations between systemic inflammation, obesity and healthy days: a health related quality of life (HRQOL) analysis of NHANES 2005–2008. BMC Obes. 5, 21 (2018).
Patnam, R. et al. Effect of BMI on clinical outcomes following minimally invasive sacrocolpopexy. J. Robot. Surg. 15, 63–68 (2021).
Davila, G. W., Baessler, K., Cosson, M. & Cardozo, L. Selection of patients in whom vaginal graft use may be appropriate. Int. Urogynecol. J. 23, 7–14 (2012).
Bradley, C. S., Visco, A. G., Weber LeBrun, E. E. & Barber, M. D. The pelvic floor disorders registry: purpose and development. Female Pelvic Med. Reconstr. Surg. 22, 77–82 (2016).
Silva, M. M. et al. Systemic inflammatory reaction after silicone breast implant. Aesthetic Plast. Surg. 35, 789–794 (2011).
Yao, Z., Lin, T.-H., Pajarinen, J., Sato, T. & Goodman, S. in Host Response to Biomaterials Ch. 12 (ed. Badylak, S. F.) 315–373 (Academic, 2015).
Tennyson, L. et al. Characterization of the T-cell response to polypropylene mesh in women with complications. Am. J. Obstet. Gynecol. 220, 187.e1–187.e8 (2019).
Robichaud, A. et al. Avoidance of the vaginal incision site for mesh placement in vaginal wall prolapse surgery: a prospective study. Eur. J. Obstet. Gynecol. Reprod. Biol. 217, 131–136 (2017).
Leanza, V., Zanghì, G., Vecchio, R. & Leanza, G. How to prevent mesh erosion in transobturator Tension-Free Incontinence Cystocoele Treatment (TICT): a comparative survey. G. Chir. 36, 21–25 (2015).
Achtari, C., Hiscock, R., O’Reilly, B. A., Schierlitz, L. & Dwyer, P. L. Risk factors for mesh erosion after transvaginal surgery using polypropylene (Atrium) or composite polypropylene/polyglactin 910 (Vypro II) mesh. Int. Urogynecol. J. 16, 389–394 (2005).
Chung, L., Maestas, D. Jr, Housseau, F. & Elisseeff, J. Key players in the immune response to biomaterial scaffolds for regenerative medicine. Adv. Drug Deliv. Rev. 114, 184–192 (2017).
Londono, R. & Badylak, S. F. in Host Response to Biomaterials Ch. 1 (ed. Badylak, S. F.) 1–12 (Academic, 2015).
Swartzlander, M. D. et al. Linking the foreign body response and protein adsorption to PEG-based hydrogels using proteomics. Biomaterials 41, 26–36 (2015).
Horbett, T. A. Chapter 13 Principles underlying the role of adsorbed plasma proteins in blood interactions with foreign materials. Cardiovasc. Pathol. 2, 137–148 (1993).
Al-Maawi, S., Orlowska, A., Sader, R., James Kirkpatrick, C. & Ghanaati, S. In vivo cellular reactions to different biomaterials — physiological and pathological aspects and their consequences. Semin. Immunol. 29, 49–61 (2017).
Anderson, J. M., Rodriguez, A. & Chang, D. T. Foreign body reaction to biomaterials. Semin. Immunol. 20, 86–100 (2008).
Gurevich, D. B. et al. Live imaging the Foreign Body Response reveals how dampening inflammation reduces fibrosis. J. Cell Sci. 133, jcs236075 (2019).
Chung, L. et al. Interleukin 17 and senescent cells regulate the foreign body response to synthetic material implants in mice and humans. Sci. Transl Med. 12, eaax3799 (2020).
Veiseh, O. & Vegas, A. J. Domesticating the foreign body response: recent advances and applications. Adv. Drug Deliv. Rev. 144, 148–161 (2019).
Hachim, D. et al. Effects of aging upon the host response to implants. J. Biomed. Mater. Res. A 105, 1281–1292 (2017).
Sadtler, K. et al. Design, clinical translation and immunological response of biomaterials in regenerative medicine. Nat. Rev. Mater. 1, 16040 (2016).
Duluc, D. et al. Functional diversity of human vaginal APC subsets in directing T-cell responses. Mucosal Immunol. 6, 626–638 (2013).
Heymann, F. et al. Polypropylene mesh implantation for hernia repair causes myeloid cell-driven persistent inflammation. JCI Insight 4, e123862 (2019).
Mosser, D. M. & Edwards, J. P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 8, 958–969 (2008).
Wang, A. C., Lee, L.-Y., Lin, C.-T. & Chen, J.-R. A histologic and immunohistochemical analysis of defective vaginal healing after continence taping procedures: a prospective case-controlled pilot study. Am. J. Obstet. Gynecol. 191, 1868–1874 (2004).
Wang, A., Lin, C.T., Ko, Y.S. & Lin, Y.H. Effector mechanisms of the site specific graft rejection after intravaginal mesh implantation — a long-term prospective case-controlled study [abstract 57]. in International Continence Society (2010).
Kavvadias, T., Kaemmer, D., Klinge, U., Kuschel, S. & Schuessler, B. Foreign body reaction in vaginally eroded and noneroded polypropylene suburethral slings in the female: a case series. Int. Urogynecol. J. Pelvic Floor Dysfunct. 20, 1473–1476 (2009).
Smith, T. M. et al. Pathologic evaluation of explanted vaginal mesh. Female Pelvic Med. Reconstr. Surg. 19, 238–241 (2013).
Elmer, C., Blomgren, B., Falconer, C., Zhang, A. & Altman, D. Histological inflammatory response to transvaginal polypropylene mesh for pelvic reconstructive surgery. J. Urol. 181, 1189–1195 (2009).
Shi, C. et al. Clinical analysis of pain after transvaginal mesh surgery in patients with pelvic organ prolapse. BMC Womens Health 21, 46 (2021).
Crosby, E. C. et al. Symptom resolution after operative management of complications from transvaginal mesh. Obstet. Gynecol. 123, 134–139 (2014).
Holihan, J. L. et al. Mesh location in open ventral hernia repair: a systematic review and network meta-analysis. World J. Surg. 40, 89–99 (2016).
Wise, J. Hernia mesh complications may have affected up to 170 000 patients, investigation finds. BMJ 362, k4104 (2018).
Sevinç, B., OkuŞ, A., Ay, S., Aksoy, N. & Karahan, Ö. Randomized prospective comparison of long-term results of onlay and sublay mesh repair techniques for incisional hernia. Turkish J. Surg. 34, 17–20 (2018).
de Landsheere, L. et al. Surgical intervention after transvaginal Prolift mesh repair: retrospective single-center study including 524 patients with 3 years’ median follow-up. Am. J. Obstet. Gynecol. 206, 83.e1–83.e7 (2012).
Wang, Y. et al. Vaginal type-II mucosa is an inductive site for primary CD8+ T-cell mucosal immunity. Nat. Commun. 6, 6100 (2015).
Mselle, T. F. et al. Unique characteristics of NK cells throughout the human female reproductive tract. Clin. Immunol. 124, 69–76 (2007).
Johansson, E. L., Rudin, A., Wassén, L. & Holmgren, J. Distribution of lymphocytes and adhesion molecules in human cervix and vagina. Immunology 96, 272–277 (1999).
Usala, S. J., Usala, F. O., Haciski, R., Holt, J. A. & Schumacher, G. F. IgG and IgA content of vaginal fluid during the menstrual cycle. J. Reprod. Med. 34, 292–294 (1989).
Zhou, J. Z., Way, S. S. & Chen, K. Immunology of the uterine and vaginal mucosae. Trends Immunol. 39, 302–314 (2018).
Ghosh, M., Rodriguez-Garcia, M. & Wira, C. R. The immune system in menopause: pros and cons of hormone therapy. J. Steroid Biochem. Mol. Biol. 142, 171–175 (2014).
Arruvito, L., Sanz, M., Banham, A. H. & Fainboim, L. Expansion of CD4+CD25+ and FOXP3+ regulatory T cells during the follicular phase of the menstrual cycle: implications for human reproduction. J. Immunol. 178, 2572–2578 (2007).
Veit-Rubin, N. et al. Abnormal vaginal microbiome associated with vaginal mesh complications. Neurourol. Urodyn. 38, 2255–2263 (2019).
Miller, E. A., Beasley, D. E., Dunn, R. R. & Archie, E. A. Lactobacilli dominance and vaginal pH: why is the human vaginal microbiome unique? Front. Microbiol. 7, 1936 (2016).
Diez-Itza, I., Aizpitarte, I. & Becerro, A. Risk factors for the recurrence of pelvic organ prolapse after vaginal surgery: a review at 5 years after surgery. Int. Urogynecol. J. Pelvic Floor Dysfunct. 18, 1317–1324 (2007).
Whiteside, J. L., Weber, A. M., Meyn, L. A. & Walters, M. D. Risk factors for prolapse recurrence after vaginal repair. Am. J. Obstet. Gynecol. 191, 1533–1538 (2004).
Alperin, M., Cook, M., Tuttle, L. J., Esparza, M. C. & Lieber, R. L. Impact of vaginal parity and aging on the architectural design of pelvic floor muscles. Am. J. Obstet. Gynecol. 215, 312.e1–9 (2016).
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Inflammaging: a new immune–metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Ferrucci, L. & Fabbri, E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 15, 505–522 (2018).
Bharath, L. P. et al. Metformin enhances autophagy and normalizes mitochondrial function to alleviate aging-associated inflammation. Cell Metab. 32, 44–55.e6 (2020).
Geller, E. J., Babb, E., Nackley, A. G. & Zolnoun, D. Incidence and risk factors for pelvic pain after mesh implant surgery for the treatment of pelvic floor disorders. J. Minim. Invasive Gynecol. 24, 67–73 (2017).
Kasyan, G., Abramyan, K., Popov, A. A., Gvozdev, M. & Pushkar, D. Mesh-related and intraoperative complications of pelvic organ prolapse repair. Cent. Eur. J. Urol. 67, 296–301 (2014).
Sendama, W. The effect of ageing on the resolution of inflammation. Ageing Res. Rev. 57, 101000 (2020).
Sebastian-Valverde, M. & Pasinetti, G. M. The NLRP3 inflammasome as a critical actor in the inflammaging process. Cells 9, 1552 (2020).
Khadzhieva, M. B., Kolobkov, D. S., Kamoeva, S. V. & Salnikova, L. E. Expression changes in pelvic organ prolapse: a systematic review and in silico study. Sci. Rep. 7, 7668 (2017).
Zhao, Y., Xia, Z., Lin, T. & Yin, Y. Significance of hub genes and immune cell infiltration identified by bioinformatics analysis in pelvic organ prolapse. PeerJ 8, e9773 (2020).
Mouthuy, P.-A. et al. Biocompatibility of implantable materials: an oxidative stress viewpoint. Biomaterials 109, 55–68 (2016).
Wattamwar, P. P. & Dziubla, T. D. in Engineering Biomaterials for Regenerative Medicine (ed. Bhatia, S.) 161–192 (Springer, 2012).
Iakovlev, V. V., Guelcher, S. A. & Bendavid, R. Degradation of polypropylene in vivo: a microscopic analysis of meshes explanted from patients. J. Biomed. Mater. Res. B Appl. Biomater. 105, 237–248 (2017).
Cochran, D. & Dziubla, T. D. in Antioxidant Polymers Ch. 15 (eds Cirilo, G. & Iemma, F.) 459–484 (Wiley, 2012).
Davison, N. L., Barrère-de Groot, F. & Grijpma, D. W. in Tissue Engineering Ch. 6 (eds Van Blitterswijk, C. A. & De Boer, J.) 177–215 (Academic, 2014).
Talley, A. D., Rogers, B. R., Iakovlev, V., Dunn, R. F. & Guelcher, S. A. Oxidation and degradation of polypropylene transvaginal mesh. J. Biomater. Sci. Polym. Ed. 28, 444–458 (2017).
Costello, C. R., Bachman, S. L., Ramshaw, B. J. & Grant, S. A. Materials characterization of explanted polypropylene hernia meshes. J. Biomed. Mater. Res. B 83, 44–49 (2007).
Göpferich, A. Mechanisms of polymer degradation and erosion. Biomaterials 17, 103–114 (1996).
Thames, S. F., White, J. B. & Ong, K. L. The myth: in vivo degradation of polypropylene-based meshes. Int. Urogynecol. J. 28, 285–297 (2017).
Milleret, V. et al. Protein adsorption steers blood contact activation on engineered cobalt chromium alloy oxide layers. Acta Biomater. 24, 343–351 (2015).
Serpooshan, V. et al. Protein corona influences cell-biomaterial interactions in nanostructured tissue engineering Scaffolds. Adv. Funct. Mater. 25, 4379–4389 (2015).
Meder, F., Brandes, C., Treccani, L. & Rezwan, K. Controlling protein–particle adsorption by surface tailoring colloidal alumina particles with sulfonate groups. Acta Biomater. 9, 5780–5787 (2013).
Feola, A. et al. Deterioration in biomechanical properties of the vagina following implantation of a high-stiffness prolapse mesh. BJOG 120, 224–232 (2013).
Liang, R. et al. Vaginal degeneration following implantation of synthetic mesh with increased stiffness. BJOG 120, 233–243 (2013).
Sumner, D. R. & Galante, J. O. Determinants of stress shielding. Clin. Orthop. Relat. Res. 274, 202–212 (1992).
Sridharan, R., Cavanagh, B., Cameron, A. R., Kelly, D. J. & O’Brien, F. J. Material stiffness influences the polarization state, function and migration mode of macrophages. Acta Biomater. 89, 47–59 (2019).
Ji, Y. et al. Substrate stiffness affects the immunosuppressive and trophic function of hMSCs via modulating cytoskeletal polymerization and tension. Biomater. Sci. 7, 5292–5300 (2019).
Blakney, A. K., Swartzlander, M. D. & Bryant, S. J. The effects of substrate stiffness on the in vitro activation of macrophages and in vivo host response to poly(ethylene glycol)-based hydrogels. J. Biomed. Mater. Res. A 100, 1375–1386 (2012).
Roman, S. et al. Use of a simple in vitro fatigue test to assess materials used in the surgical treatment of stress urinary incontinence and pelvic organ prolapse. Neurourol. Urodyn. 38, 107–115 (2019).
Kruger, J. A., Yan, X., Li, X., Nielsen, P. M. F. & Nash, M. P. in Biomechanics of the Female Pelvic Floor Ch. 18 (eds Hoyte, L. & Damaser, M.) 367–382 (Academic, 2016).
Kruger, J., Hayward, L., Nielsen, P., Loiselle, D. & Kirton, R. Design and development of a novel intra-vaginal pressure sensor. Int. Urogynecol. J. 24, 1715–1721 (2013).
Velayudhan, S., Martin, D. & Cooper-White, J. Evaluation of dynamic creep properties of surgical mesh prostheses — uniaxial fatigue. J. Biomed. Mater. Res. B 91, 287–296 (2009).
Liang, R., Knight, K., Abramowitch, S. & Moalli, P. A. Exploring the basic science of prolapse meshes. Curr. Opin. Obstet. Gynecol. 28, 413–419 (2016).
Durst, P. J. & Heit, M. H. Polypropylene mesh predicts mesh/suture exposure after sacrocolpopexy independent of known risk factors: a retrospective case-control study. Female Pelvic Med. Reconstr. Surg. 24, 360–366 (2018).
Goldstein, H. S. Selecting the right mesh. Hernia 3, 23–26 (1999).
Barone, W. R., Moalli, P. A. & Abramowitch, S. D. Textile properties of synthetic prolapse mesh in response to uniaxial loading. Am. J. Obstet. Gynecol. 215, 326.e1–326.e9 (2016).
Amid, P. K. Classification of biomaterials and their related complications in abdominal wall hernia surgery. Hernia 1, 15–21 (1997).
Klinge, U. & Klosterhalfen, B. Modified classification of surgical meshes for hernia repair based on the analyses of 1,000 explanted meshes. Hernia 16, 251–258 (2012).
Otto, J., Kaldenhoff, E., Kirschner-Hermanns, R., Mühl, T. & Klinge, U. Elongation of textile pelvic floor implants under load is related to complete loss of effective porosity, thereby favoring incorporation in scar plates. J. Biomed. Mater. Res. A 102, 1079–1084 (2014).
Junge, K. et al. Mesh biocompatibility: effects of cellular inflammation and tissue remodelling. Langenbecks Arch. Surg. 397, 255–270 (2012).
Manodoro, S. et al. Graft-related complications and biaxial tensiometry following experimental vaginal implantation of flat mesh of variable dimensions. BJOG 120, 244–250 (2013).
Klinge, U. et al. Impact of polymer pore size on the interface scar formation in a rat model. J. Surg. Res. 103, 208–214 (2002).
US Food and Drug Administration. Search the releasable 510(k) database (FDA, 2018).
Institute of Medicine. Medical Devices and the Public’s Health (National Academies, 2011).
International Consortium of Investigative Journalists. Medical devices harm patients worldwide as governments fail on safety. ICIJ https://www.icij.org/investigations/implant-files/medical-devices-harm-patients-worldwide-as-governments-fail-on-safety/ (2018).
Heneghan, C. et al. Transvaginal mesh failure: lessons for regulation of implantable devices. BMJ 359, j5515 (2017).
Zargar, N. & Carr, A. The regulatory ancestral network of surgical meshes. PLoS ONE 13, e0197883 (2018).
Ostergard, D. R. Vaginal mesh grafts and the Food and Drug Administration. Int. Urogynecol. J. 21, 1181–1183 (2010).
Heneghan, C. et al. Trials of transvaginal mesh devices for pelvic organ prolapse: a systematic database review of the US FDA approval process. BMJ Open 7, e017125 (2017).
Barber, S. Surgical mesh implants (House of Commons Library, 2018).
Obstetrical and Gynecological Devices. Reclassification of Surgical mesh for transvaginal pelvic organ prolapse repair; final order. Fed. Regist. 81, 353–361 (2016).
US Food and Drug Administration. Code of Federal Regulations — Title 21 (FDA, 2019).
Hartung, T. & Daston, G. Are in vitro tests suitable for regulatory use? Toxicol. Sci. 111, 233–237 (2009).
International Organization for Standardization. ISO 10993-6:2016: biological evaluation of medical devices — Part 6: Tests for local effects after implantation (ISO, 2020).
International Organization for Standardization. ISO 10993-11:2006: biological evaluation of medical devices — Part 11: Tests for systemic toxicity (ISO, 2020).
Hast, M. W., Zuskov, A. & Soslowsky, L. J. The role of animal models in tendon research. Bone Jt. Res. 3, 193–202 (2014).
Pierce, L. M. et al. Long-term histologic response to synthetic and biologic graft materials implanted in the vagina and abdomen of a rabbit model. Am. J. Obstet. Gynecol. 200, 546.e1–546.e8 (2009).
Ostergard, D. R. Degradation, infection and heat effects on polypropylene mesh for pelvic implantation: what was known and when it was known. Int. Urogynecol. J. 22, 771–774 (2011).
Medicines and Healthcare Products Regulatory Agency. Yellow card scheme. Yellow Card https://yellowcard.mhra.gov.uk/ (2021).
US Food and Drug Administration. MAUDE — manufacturer and user facility device experience (FDA, 2021).
Rimmer, A. Vaginal mesh procedures need compulsory register, says Royal College. BMJ 360, k586 (2018).
National Joint Registry. Welcome to the national joint registry (NJR). NJR http://www.njrcentre.org.uk/njrcentre/ (2020).
NHS Digital. Breast and cosmetic implant registry (NHS Digital, 2021).
Hansen, U. D., Gradel, K. O. & Larsen, M. D. Danish urogynaecological database. Clin. Epidemiol. 8, 709–712 (2016).
Daly, J. O., Ahern, S., Herkes, R. & O’Connell, H. E. The Australasian pelvic floor procedure registry: not before time. Aust. N. Zeal. J. Obstet. Gynaecol. 59, 473–476 (2019).
New Zealand Ministry of Health. Surgical mesh registry: cost benefit analysis (Deloitte, 2018).
Bako, A. & Dhar, R. Review of synthetic mesh-related complications in pelvic floor reconstructive surgery. Int. Urogynecol. J. 20, 103–111 (2009).
International Association for the Study of Pain. Definition of pain. IASP https://www.iasp-pain.org/Education/Content.aspx?ItemNumber=1698#Pain (2020).
Haylen, B. et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint terminology and classification of the complications related directly to the insertion of prostheses (meshes, implants, tapes) and grafts in female pelvic floor surgery. Neurourol. Urodyn. 30, 2–12 (2011).
Author information
Authors and Affiliations
Contributions
R.E.A., H.L.M. and S.J.B.S. researched data for the article, R.E.A., M.L.I.-K., R.C. and S.J.B.S. made substantial contributions to the discussion of the content of the article, R.E.A., M.I., H.L.M. and S.S. wrote the article, R.E.A., M.I., H.L.M., R.C. and S.S. reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Urology thanks S. Takahashi, P. Moalli and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Biocompatible
-
Biocompatibility is the ability of a material to perform its intended function with an appropriate host immune response for a specific application.
- Microporous meshes
-
According to the widely used Amid classification, microporous mesh refers to mesh that contains pores that are <10 microns in size.
- M1 macrophages
-
Classically activated macrophages typically associated with pro-inflammatory responses in in vitro studies.
- M2 macrophages
-
Alternatively activated macrophages that typically induce pro-resolving and anti-inflammatory responses in in vitro studies.
- TH1 cytokines
-
For example, IFNγ and TNF are typically associated with pro-inflammatory responses driving immune cell activation and maturation.
- Vroman effect
-
The sequence in which serum proteins adsorb to a surface; proteins that adsorb earlier are competitively displaced by other proteins with stronger binding affinities.
- Mesh knot
-
Refers to a dense area of mesh fibres immediately adjacent to each other.
- 510k route
-
Refers to a premarket submission process made to the FDA, demonstrating that the device that is to be marketed is as safe and effective (that is, substantially equivalent) to a legally marketed device.
- Predicate device
-
A medical device that is used as a point of comparison for new medical devices seeking approval through the FDA’s 510k premarket clearance pathway.
Rights and permissions
About this article
Cite this article
Abhari, R.E., Izett-Kay, M.L., Morris, H.L. et al. Host–biomaterial interactions in mesh complications after pelvic floor reconstructive surgery. Nat Rev Urol 18, 725–738 (2021). https://doi.org/10.1038/s41585-021-00511-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-021-00511-y
This article is cited by
-
Strengthening preclinical testing to increase safety in surgical mesh
Nature Reviews Urology (2024)
-
Elaborating Polyurethane Pillowy Soft Mat on Polypropylene Monofilament Surface with Stepwise Surface Treatments
Chinese Journal of Polymer Science (2022)