Abstract
The inflammasome components NLRP3 and ASC are cytosolic proteins, which upon sensing endotoxins or danger cues, form multimeric complexes to process interleukin (IL)-1β for secretion. Here we found that antigen (Ag)-triggered degranulation of IgE-sensitized mast cells (MCs) was mediated by NLRP3 and ASC. IgE–Ag stimulated NEK7 and Pyk2 kinases in MCs to induce the deposition of NLRP3 and ASC on granules and form a distinct protein complex (granulosome) that chaperoned the granules to the cell surface. MCs deficient in NLRP3 or ASC did not form granulosomes, degranulated poorly in vitro and did not evoke systemic anaphylaxis in mice. IgE–Ag-triggered anaphylaxis was prevented by an NLRP3 inhibitor. In endotoxin-primed MCs, pro-IL-1β was rapidly packaged into granules after IgE–Ag stimulation and processed within granule remnants by proteases after degranulation, causing lethal anaphylaxis in mice. During IgE–Ag-mediated degranulation of endotoxin-primed MCs, granulosomes promoted degranulation, combined with exteriorization and processing of IL-1β, resulting in severe inflammation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Source data are available on figshare at https://doi.org/10.6084/m9.figshare.21856569.
References
Schroder, K. & Tschopp, J. The inflammasomes. Cell 140, 821–832 (2010).
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol. Cell. 10, 417–426 (2002).
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Swanson, K. V., Deng, M. & Ting, J. P.-Y. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 19, 477–489 (2019).
He, Y., Zeng, M. Y., Yang, D., Motro, B. & Nunez, G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature 530, 354–357 (2016).
Chung, I.-C. et al. Pyk2 activates the NLRP3 inflammasome by directly phosphorylating ASC and contributes to inflammasome-dependent peritonitis. Sci. Rep. 6, 36214 (2016).
Guo, H., Callaway, J. B. & Ting, J. P.-Y. Inflammasomes: mechanism of action, role in disease, and therapeutics. Nat. Med. 21, 677–687 (2015).
Wen, H., Ting, J. P.-Y. & O’Neill, L. A. A role for the NLRP3 inflammasome in metabolic diseases–did Warburg miss inflammation? Nat. Immunol. 13, 352–357 (2012).
Freeman, L. C. & Ting, J. P.-Y The pathogenic role of the inflammasome in neurodegenerative diseases. J. Neurochem. 136, 29–38 (2016).
Prochnicki, T. & Latz, E. Inflammasomes on the crossroads of innate immune recognition and metabolic control. Cell Metab. 26, 71–93 (2017).
Hughes, M. M. & O’Neill, L. A. J. Metabolic regulation of NLRP3. Immunol. Rev. 281, 88–98 (2018).
Akira, S., Misawa, T., Satoh, T. & Saitoh, T. Macrophages control innate inflammation. Diabetes Obes. Metab. 15, 10–18 (2013).
Abraham, S. N. & St John, A. L. Mast cell-orchestrated immunity to pathogens. Nat. Rev. Immunol. 10, 440–452 (2010).
Gilfillan, A. M. & Tkaczyk, C. Integrated signalling pathways for mast-cell activation. Nat. Rev. Immunol. 6, 218–230 (2006).
Nakamura, Y. et al. Critical role for mast cells in interleukin-1beta-driven skin inflammation associated with an activating mutation in the nlrp3 protein. Immunity 37, 85–95 (2012).
Jin, C. et al. Particulate allergens potentiate allergic asthma in mice through sustained IgE-mediated mast cell activation. J. Clin. Invest. 127, 3913 (2017).
Hogan, P. G., Lewis, R. S. & Rao, A. Molecular basis of calcium signaling in lymphocytes: STIM and ORAI. Annu. Rev. Immunol. 28, 491–533 (2010).
Lorentz, A., Baumann, A., Vitte, J. & Blank, U. The SNARE machinery in mast cell secretion. Front. Immunol. 3, 143 (2012).
Dwyer, D. F., Barrett, N. A., Austen, K. F. & Immunological Genome Project Consortium Expression profiling of constitutive mast cells reveals a unique identity within the immune system. Nat. Immunol. 17, 878–887 (2016).
Nocka, K. et al. Molecular bases of dominant negative and loss of function mutations at the murine c-kit/white spotting locus: W37, Wv, W41 and W. EMBO J. 9, 1805–1813 (1990).
Falcone, F. H., Wan, D., Barwary, N. & Sagi-Eisenberg, R. RBL cells as models for in vitro studies of mast cells and basophils. Immunol. Rev. 282, 47–57 (2018).
Galli, S. J. & Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 18, 693–704 (2012).
Huber, M., Hughes, M. R. & Krystal, G. Thapsigargin-induced degranulation of mast cells is dependent on transient activation of phosphatidylinositol-3 kinase. J. Immunol. 165, 124–133 (2000).
Stehlik, C. et al. Apoptosis-associated speck-like protein containing a caspase recruitment domain is a regulator of procaspase-1 activation. J. Immunol. 171, 6154–6163 (2003).
Byrne, M. J. et al. Nek7 conformational flexibility and inhibitor binding probed through protein engineering of the R-spine. Biochem. J. 477, 1525–1539 (2020).
He, H. et al. Oridonin is a covalent NLRP3 inhibitor with strong anti-inflammasome activity. Nat. Commun. 9, 2550 (2018).
Hara, H. et al. Phosphorylation of the adaptor ASC acts as a molecular switch that controls the formation of speck-like aggregates and inflammasome activity. Nat. Immunol. 14, 1247–1255 (2013).
Okazaki, H., Zhang, J., Hamawy, M. M. & Siraganian, R. P. Activation of protein-tyrosine kinase Pyk2 is downstream of Syk in FcεRI signaling. J. Biol. Chem. 272, 32443–32447 (1997).
Cheung, S. M. & Ostergaard, H. L. Pyk2 controls integrin-dependent CTL migration through regulation of de-adhesion. J. Immunol. 197, 1945–1956 (2016).
Nishida, K. et al. FcεRI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J. Cell Biol. 170, 115–126 (2005).
Draber, P., Sulimenko, V. & Draberova, E. Cytoskeleton in mast cell signaling. Front. Immunol. 3, 130 (2012).
Liu, Y., Zhu, M., Nishida, K., Hirano, T. & Zhang, W. An essential role for RasGRP1 in mast cell function and IgE-mediated allergic response. J. Exp. Med. 204, 93–103 (2007).
Reck-Peterson, S. L., Redwine, W. B., Vale, R. D. & Carter, A. P. The cytoplasmic dynein transport machinery and its many cargoes. Nat. Rev. Mol. Cell Biol. 19, 382–398 (2018).
Jiang, H. et al. Identification of a selective and direct NLRP3 inhibitor to treat inflammatory disorders. J. Exp. Med. 214, 3219–3238 (2017).
Suurmond, J., Habets, K. L. L., Dorjee, A. L., Huizinga, T. W. & Toes, R. E. M. Expansion of Th17 cells by human mast cells is driven by inflammasome-independent IL-1beta. J. Immunol. 197, 4473–4481 (2016).
Kunder, C. A. et al. Mast cell-derived particles deliver peripheral signals to remote lymph nodes. J. Exp. Med. 206, 2455–2467 (2009).
Ronnberg, E., Melo, F. R. & Pejler, G. Mast cell proteoglycans. J. Histochem. Cytochem. 60, 950–962 (2012).
Thorne, P. S. et al. Endotoxin exposure is a risk factor for asthma: the national survey of endotoxin in United States housing. Am. J. Respir. Crit. Care Med. 172, 1371–1377 (2005).
Vercelli, D. Discovering susceptibility genes for asthma and allergy. Nat. Rev. Immunol. 8, 169–182 (2008).
Martin, R. K. et al. B1 Cell IgE impedes mast cell-mediated enhancement of parasite expulsion through B2 IgE blockade. Cell Rep. 22, 1824–1834 (2018).
Knight, P. A., Wright, S. H., Lawrence, C. E., Paterson, Y. Y. & Miller, H. R. Delayed expulsion of the nematode Trichinella spiralis in mice lacking the mucosal mast cell-specific granule chymase, mouse mast cell protease-1. J. Exp. Med. 192, 1849–1856 (2000).
Monaco, G. et al. RNA-seq signatures normalized by mRNA abundance allow absolute deconvolution of human immune cell types. Cell Rep. 26, 1627–1640 (2019).
Encalada, S. E., Szpankowski, L., Xia, C. H. & Goldstein, L. S. Stable kinesin and dynein assemblies drive the axonal transport of mammalian prion protein vesicles. Cell 144, 551–565 (2011).
Efergan, A. et al. Rab12 regulates retrograde transport of mast cell secretory granules by interacting with the RILP–dynein complex. J. Immunol. 196, 1091–1101 (2016).
Wang, Z. et al. NLRP3 inflammasome and inflammatory diseases. Oxid. Med. Cell. Longev. 2020, 4063562 (2020).
Sakamaki, K. & Satou, Y. Caspases: evolutionary aspects of their functions in vertebrates. J. Fish. Biol. 74, 727–753 (2009).
Acknowledgements
We thank Duke-NUS Medical School, Singapore, for their help with light microscopy imaging. We thank the SIgN Mutant Mouse Collection Core Facility for providing the mice. Funding for this study was provided by a block grant from Duke-NUS and NIGMS R01-GM144606.
Author information
Authors and Affiliations
Contributions
A. Mencarelli, P.B. and S.N.A. conceived the study. A. Mencarelli, P.B. and H.W.C. performed experiments with advice from S.N.A. A. Mencarelli, P.B., H.J.K. and S.N.A. analyzed the data. A. Mortellaro supervised the in vivo experiments with mutant mice and contributed to data analysis. A. Mencarelli, P.B. and S.N.A. wrote the paper. All authors contributed to discussions and paper review. This work was supported by a block grant (to S.N.A.) from Duke-NUS Medical School, Singapore, and a SIgN core grant (A. Mortellaro) from A*STAR, Singapore.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Ioana Staicu, in collaboration with the Nature Immunology team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Nlrp3−/− and Asc−/− BMMCs exhibit similar early signaling events as WT.
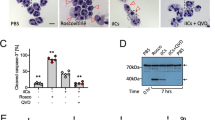
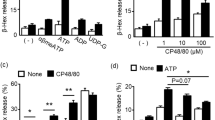
a, The impaired FcεRI-mediated degranulation observed in MCs ablated of Asc and Nlrp3 was also confirmed by flow cytometric analysis of the externalization of the granule membrane marker CD63 after IgE-Ag stimulation. b β-hexosaminidase release from IgE-Ag stimulated WT BMMCs silenced for siRNA targeting Control or Caspase-1. Western blot analysis of Caspase-1 protein levels. c, The BMMCs were subjected to toluidine blue staining which stains mature MC granule. Middle, flow cytometry analysis of BMMCs obtained from WT, Asc−/− Nlrp3−/− or Caspase1/11−/− mice showing that all BMMCs were similarly matured. The maturation markers employed were FcεRI, CD117, and avidin, respectively. d, Compared to WT BMMCs, Asc−/−, Caspase-1/11−/− and Nlrp3−/− BMMCs exhibit significantly reduced release of granule associated mediator’s TNF-α and histamine following IgE-Ag-mediated stimulation. Compared to WT BMMCs, Asc−/− and Nlrp3−/− BMMCs exhibited similar levels of secretion of de novo synthesized mediators PGD2 and IL-6. e, Western blots showing IgE-sensitized WT, Nlrp3−/− or Asc−/− BMMCs stimulated with Ag at different time points. The data are shown in a as mean ± s.d of two independent experiments, performed in triplicate, **P < 0.01, ***P < 0.005 (Ordinary one-way ANOVA multiple comparisons procedure), WT vs. Asc−/− and Nlrp3−/− BMMCs, for each time point indicated. Data are shown in b are mean ± s.e.m of one representative experiment, of three independent experiments with similar results, performed in triplicate; The images shown in c, are the representatives of four independent experiments, with similar results. The data are shown in c left, as mean ± s.d. of four independent experiments. The data are shown in d as mean ± s.d of two independent experiments, performed in duplicate or triplicate, **P < 0.01 (Ordinary one-way ANOVA multiple comparisons procedure), WT vs. Asc−/− and Nlrp3−/− BMMCs as indicated. Data shown in e are representative of three independent experiments.
Extended Data Fig. 2 The specificity of NLRP3 and ASC for MC granules CD63.
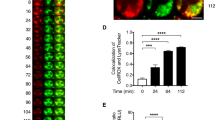
a, Immunofluorescence images of Asc−/− and Nlrp3−/− BMMCs co-stained with CD63 (green) and NLRP3 (red) or ASC (orange), respectively. Scale bar: 20 μm. b, Endogenous CD63 was immunoprecipitated from untreated or LPS/nigericin stimulated WT BMMCs. The association between CD63 and NLRP3, ASC, and Caspase-1 in different conditions was examined by Western blotting of the IP fractions. The proteins of interests in the total cell lysate (TCL) were also depicted to indicate that a similar level of CD63, NLRP3, ASC, and Caspase-1 protein present in each fraction. c, WT BMMCs were stimulated either with IgE-Ag or LPS/nigericin for the indicated time intervals, and the lysates were subjected to immunoprecipitation using NLRP3 antibody followed by immunoblotting with ASC, and NLRP3 antibodies. TCL was loaded as an input control. d, Myc-tagged ASC and Flag-tagged Caspase-1 with varying amounts of GFP-CD63 was overexpressed in HEK293T cells, and immunoprecipitated with Flag antibody revealing the binding of Caspase-1 with ASC by Western blotting of the IP fraction. The proteins of interests in the total cell lysate (TCL) was also depicted to indicate a similar level of CD63, ASC, and Caspase-1 protein present in each fraction. e, A diagrammatic representation of the spatial orientation of CD63 bound to either NLRP3 or ASC on granule membrane. Bottom, full length and its truncated forms of GFP-CD63 (1–6) used for immunoprecipitation assay with either Myc-tagged ASC or Flag-tagged NLRP3 in HEK293T cells. f, Western blots showing immunoprecipitation of GFP-tagged CD63 TM3 domain with varying amounts of ASC or NLRP3 in HEK293T cells. Two dark arrows indicate the varying concentration of NLRP3 or ASC expression plasmids. Immunofluorescence image data shown in a is representative of three independent experiments. The data shown in b is representative of two independent experiments. The data shown in c to e are representative of more than three independent experiments.
Extended Data Fig. 3 Signaling events downstream of Ca2+ axis is important for protein complex formation.
a, Western blots of immunoprecipitated CD63 with NLRP3, ASC, and tubulin from the lysates, sensitized with IgE and pretreated with or without BAPTA-AM, a chelator of intracellular calcium, following Ag stimulation in WT BMMCs. b, Western blots showing endogenous immunoprecipitation of NEK7 from WT and Asc−/− BMMCs in response to IgE-Ag stimulation as indicated. The proteins of interests in the total cell lysate (TCL) was also depicted to indicate that similar level of NLRP3, NEK7, and ASC protein present in each fraction. c, Western blots showing the status of Pyk2 and ASC activation in IgE-Ag-stimulated WT and Nlrp3−/− BMMCs. c, ASC is located downstream of Pyk2. Western blots showing the status of Pyk2 and ASC activation in IgE-Ag-stimulated WT and Nlrp3−/− BMMCs. Data presented in a to c are representative of three or more independent experiments.
Extended Data Fig. 4 Distribution of tubulin and dynein on MC granules.
a, Quantitative analysis of α-tubulin in WT, Nlrp3−/− and Asc−/− BMMCs following IgE-Ag stimulation. The α-tubulin signals were measured in each cell with a radius of 12-16 μm using ZEN Imaging Software (Zeiss). Background fluorescence using a circle of the corresponding size was subtracted from each measurement. b, Densitometric analysis of Fig. 4b tubulin protein intensity from WT, Nlrp3−/−, Asc−/−, and Caspase-1/11−/− BMMCs following IgE-Ag stimulation. c, β-hexosaminidase release from untreated or IgE-Ag stimulation in WT, Nlrp3−/−, and Asc−/−, BMMCs pre-treated with varying concentrations of Rhosin, a drug that targets RhoA GTPase. d, Western blots showing immunoprecipitation of NLRP3 with dynein from WT BMMCs primed with LPS and subsequently challenged with varying amounts of nigericin. e, Representative confocal microscopy images co-stained for CD63 (green) and tubulin (red). f, CD63 (green) and dynein (light yellow) in WT BMMCs following IgE-Ag stimulation at different time points. Scale bar: 20 μm. f, Densitometry analysis of Fig. 4f protein (GFP-CD63) intensity from siRNAs (control, Nlrp3, and Asc) of the plasma membrane. Data shown in a are mean ± s.d. of one representative experiments, n = 6 cells/group. ***P < 0.001 (Ordinary one-way ANOVA multiple comparisons procedure). Data shown in b are densitometry analysis of Fig. 4b and representatives of three independent experiments. Data shown in c are mean ± s.d of four independent experiments, performed in triplicate, ***P < 0.005 (Ordinary one-way ANOVA multiple comparisons procedure), vs. WT cells stimulated with IgE/Ag with no Rhosin. Data shown in d is representative of two independent experiments. Immunofluorescence images shown in e and f are representative of two independent experiments with similar results.
Extended Data Fig. 5 Schematic model showing a cascade of signaling events involved in the formation of granulosome complex during anaphylactic degranulation in MCs.
(1) Aggregation of FcεRI by antigen triggers a cascade of mast cell signaling events including the activation of phospholipase C (PLCγ) which in turn increases the intracellular Ca2+ levels facilitating the recruitment of two critical kinases, NEK7 and Pyk2. NEK7 kinase binds to NLRP3 and promotes its dimerization, whereas Pyk2 kinase mediates ASC phosphorylation and its dimerization. In many cases, following IgE-Ag activation, granules fuse with each other to form larger compound granules within the mast cell. Thereafter, dimerized NLRP3 and ASC deposit on compound granules enabling a conformational change that allows the oligomerization of CD63 to facilitate the formation of a multiprotein complex ‘granulosome’ comprising NLRP3, ASC, and CD63 (boxed figure) (2). Subsequent to the formation of a microtubule-organizing center (MTOC), a motor protein, dynein is recruited to the granulosome by specifically binding to NLRP3, which then moves along with MTs (microtubules) to the cell periphery where cytosolic compound granules are exteriorized (2). If the MCs are pre-exposed to endotoxin such as LPS, which induces NF-κB-mediated expression of pro-IL-1β(1), thereafter, following IgE-Ag stimulation, pro-IL-1β is rapidly incorporated into the compound granules and when granules are exteriorized, they contain bulk amounts of pro-IL-1β and chymase. In the external medium, pro-IL-1β is processed into mature IL-1β by chymase within the granule remnants (3) before both IL-1β and chymase are released into the external medium. Created with BioRender.com.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mencarelli, A., Bist, P., Choi, H.W. et al. Anaphylactic degranulation by mast cells requires the mobilization of inflammasome components. Nat Immunol 25, 693–702 (2024). https://doi.org/10.1038/s41590-024-01788-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-024-01788-y
This article is cited by
-
Pas de deux of NLRP3 and ASC with CD63 on mast cell granules
Nature Immunology (2024)