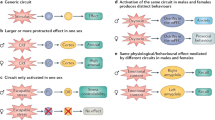
Abstract
We show that the sex of human experimenters affects mouse behaviors and responses following administration of the rapid-acting antidepressant ketamine and its bioactive metabolite (2R,6R)-hydroxynorketamine. Mice showed aversion to the scent of male experimenters, preference for the scent of female experimenters and increased stress susceptibility when handled by male experimenters. This human-male-scent-induced aversion and stress susceptibility was mediated by the activation of corticotropin-releasing factor (CRF) neurons in the entorhinal cortex that project to hippocampal area CA1. Exposure to the scent of male experimenters before ketamine administration activated CA1-projecting entorhinal cortex CRF neurons, and activation of this CRF pathway modulated in vivo and in vitro antidepressant-like effects of ketamine. A better understanding of the specific and quantitative contributions of the sex of human experimenters to study outcomes in rodents may improve replicability between studies and, as we have shown, reveal biological and pharmacological mechanisms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within the paper and its Supplementary Information files. Source data are provided with this paper.
Code availability
Bonsai script was from the Neurophotometrics system manual at https://static1.squarespace.com/static/60ff345fca665d50e1adc805/t/616103731b92d50b0f4d5833/1633747831997/Full+Length+Manual-2021.pdf
References
Mogil, J. S. Laboratory environmental factors and pain behavior: the relevance of unknown unknowns to reproducibility and translation. Lab Anim. 46, 136–141 (2017).
Sorge, R. E. et al. Olfactory exposure to males, including men, causes stress and related analgesia in rodents. Nat. Methods 11, 629–632 (2014).
Davis, H., Taylor, A. A. & Norris, C. Preference for familiar humans by rats. Psychon. Bull. Rev. 4, 118–120 (1997).
van Driel, K. S. & Talling, J. C. Familiarity increases consistency in animal tests. Behav. Brain Res. 159, 243–245 (2005).
Can, A. et al. The mouse forced swim test. J. Vis. Exp. (2012) https://doi.org/10.3791/3638
Planchez, B., Surget, A. & Belzung, C. Animal models of major depression: drawbacks and challenges. J. Neural Transm. 126, 1383–1408 (2019).
Samuels, B. A. & Hen, R. in Mood and Anxiety Related Phenotypes in Mice: Characterization Using Behavioral Tests, Vol. 2 (ed. Gould, T. D.) Ch. XXX (Humana Press, 2011)..
Zanos, P. et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 533, 481–486 (2016).
Li, N. et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 329, 959–964 (2010).
Autry, A. E. et al. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 475, 91–95 (2011).
Maeng, S. et al. Cellular mechanisms underlying the antidepressant effects of ketamine: role of α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors. Biol. Psychiatry 63, 349–352 (2008).
Highland, J. N., Zanos, P., Georgiou, P. & Gould, T. D. Group II metabotropic glutamate receptor blockade promotes stress resilience in mice. Neuropsychopharmacology 44, 1788–1796 (2019).
Zanos, P. et al. (2R,6R)-Hydroxynorketamine exerts mGlu 2 receptor-dependent antidepressant actions. Proc. Natl Acad. Sci. USA 116, 6441–6450 (2019).
Zanos, P. et al. (R)-Ketamine exerts antidepressant actions partly via conversion to (2R,6R)-hydroxynorketamine, while causing adverse effects at sub-anaesthetic doses. Br. J. Pharmacol. 176, 2573–2592 (2019).
Lumsden, E. W. et al. Antidepressant-relevant concentrations of the ketamine metabolite (2R,6R)-hydroxynorketamine do not block NMDA receptor function. Proc. Natl Acad. Sci. USA 116, 5160–5169 (2019).
Kim, J.-W. et al. Sustained effects of rapidly acting antidepressants require BDNF-dependent MeCP2 phosphorylation. Nat. Neurosci. 24, 1100–1109 (2021).
Wulf, H. A., Browne, C. A., Zarate, C. A. & Lucki, I. Mediation of the behavioral effects of ketamine and (2R,6R)-hydroxynorketamine in mice by kappa opioid receptors. Psychopharmacology (Berl.) (2022) https://doi.org/10.1007/s00213-022-06118-4
Pham, T. H. et al. Common neurotransmission recruited in (R,S)-ketamine and (2R,6R)-hydroxynorketamine-induced sustained antidepressant-like effects. Biol. Psychiatry 84, e3–e6 (2018).
Chen, B. K. et al. Sex-specific neurobiological actions of prophylactic (R,S)-ketamine, (2R,6R)-hydroxynorketamine, and (2S,6S)-hydroxynorketamine. Neuropsychopharmacology 45, 1545–1556 (2020).
Zanos, P. et al. Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacol. Rev. 70, 621 LP–621660 (2018).
Yao, N., Skiteva, O., Zhang, X., Svenningsson, P. & Chergui, K. Ketamine and its metabolite (2R,6R)-hydroxynorketamine induce lasting alterations in glutamatergic synaptic plasticity in the mesolimbic circuit. Mol. Psychiatry 23, 2066–2077 (2018).
Aguilar-Valles, A. et al. Antidepressant actions of ketamine engage cell-specific translation via eIF4E. Nature 590, 315–319 (2021).
Highland, J. N. et al. Hydroxynorketamines: pharmacology and potential therapeutic applications. Pharmacol. Rev. 73, 763 LP–763791 (2021).
Fukumoto, K. et al. Activity-dependent brain-derived neurotrophic factor signaling is required for the antidepressant actions of (2R,6R)-hydroxynorketamine. Proc. Natl Acad. Sci. USA 116, 297–302 (2019).
Carreno, F. R. et al. Activation of a ventral hippocampus-medial prefrontal cortex pathway is both necessary and sufficient for an antidepressant response to ketamine. Mol. Psychiatry 21, 1298–1308 (2016).
Hong, L. E. et al. Gamma and delta neural oscillations and association with clinical symptoms under subanesthetic ketamine. Neuropsychopharmacology 35, 632–640 (2010).
Smith, S. M. & Vale, W. W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 8, 383–395 (2006).
Anisman, H., Lacosta, S., Kent, P., McIntyre, D. C. & Merali, Z. Stressor-induced corticotropin-releasing hormone, bombesin, ACTH and corticosterone variations in strains of mice differentially responsive to stressors. Stress 2, 209–220 (1998).
Conti, L. H., Costello, D. G., Martin, L. A., White, M. F. & Abreu, M. E. Mouse strain differences in the behavioral effects of corticotropin-releasing factor (CRF) and the CRF antagonist α-helical CRF9-41. Pharmacol. Biochem. Behav. 48, 497–503 (1994).
Riggs, L. M. et al. (2R,6R)-Hydroxynorketamine rapidly potentiates hippocampal glutamatergic transmission through a synapse-specific presynaptic mechanism.Neuropsychopharmacology 45, 426–436 (2020).
Riggs, L. M., Thompson, S. M. & Gould, T. D. (2R,6R)-Hydroxynorketamine rapidly potentiates optically-evoked Schaffer collateral synaptic activity. Neuropharmacology 214, 109153 (2022).
Blank, T., Nijholt, I., Eckart, K. & Spiess, J. Priming of long-term potentiation in mouse hippocampus by corticotropin-releasing factor and acute stress: implications for hippocampus-dependent learning. J. Neurosci. 22, 3788–3794 (2002).
Aldenhoff, J. B., Gruol, D. L., Rivier, J., Vale, W. & Siggins, G. R. Corticotropin releasing factor decreases postburst hyperpolarizations and excites hippocampal neurons. Science 221, 875–877 (1983).
Wilson, D. A. et al. Cortical odor processing in health and disease. Prog. Brain Res. 208, 275–305 (2014).
Bitzenhofer, S. H., Westeinde, E. A., Zhang, H.-X. B. & Isaacson, J. S. Rapid odor processing by layer 2 subcircuits in lateral entorhinal cortex. eLife 11, e75065 (2022).
Ferry, B., Herbeaux, K., Javelot, H. & Majchrzak, M. The entorhinal cortex is involved in conditioned odor and context aversions. Front. Neurosci. 9, 342 (2015).
Xu, W. & Wilson, D. A. Odor-evoked activity in the mouse lateral entorhinal cortex. Neuroscience 223, 12–20 (2012).
Persson, B. M. et al. Lateral entorhinal cortex lesions impair odor-context associative memory in male rats. J. Neurosci. Res. 100, 1030–1046 (2022).
Butler-Struben, H. M., Kentner, A. C. & Trainor, B. C. What’s wrong with my experiment?: The impact of hidden variables on neuropsychopharmacology research. Neuropsychopharmacology (2022) https://doi.org/10.1038/s41386-022-01309-1
McBride, K., Slotnick, B. & Margolis, F. L. Does intranasal application of zinc sulfate produce anosmia in the mouse? An olfactometric and anatomical study. Chem. Senses 28, 659–670 (2003).
Bruno, C. A. et al. pMAT: an open-source software suite for the analysis of fiber photometry data. Pharmacol. Biochem. Behav. 201, 173093 (2021).
Gagnon, R. C. & Peterson, J. J. Estimation of confidence intervals for area under the curve from destructively obtained pharmacokinetic data. J. Pharmacokinet. Biopharm. 26, 87–102 (1998).
Acknowledgements
We thank D. Sparta for providing the CRF-ires-cre founder mice. We thank the many volunteers participating in these experiments. Brain section schematics were created with BioRender.com. This work was supported by NIH grant no. MH107615 and VA Merit grant no. 1I01BX004062 (T.D.G.), NIH grant no. MH086828 (S.M.T.) and NIH grant no. MH093897 (R.S.D.). R.M. and C.A.Z. laboratories are supported by the NIH Intramural Research Program. The contents do not represent the views of the US Department of Veterans Affairs or the US government.
Author information
Authors and Affiliations
Contributions
P.G. and T.D.G. were responsible for the overall experimental design. T.M.M. and K.J.P. performed the RNAscope experiments. T.M.M. performed automated RNAscope analysis with supervision by L.H.T. and S.M.C. D.M.G. and R.S.D. performed the independent FST replication at Yale University. C.F.P. and X.A. perfused mice and processed the brains for expression and cannula implantation confirmation. D.I.D. performed the whole-cell electrophysiology experiment with supervision from E.F.R.P. L.E.P. and P.G. performed analysis for the fiber photometry experiments. C.E.J. and B.W.S. performed qEEG surgeries. B.W.S. and P.G. performed qEEG data analysis. C.E.J. performed the FST. P.Z., J.N.H., P.G., B.W.S. and S.M.C. performed the PK studies (injections, euthanasia and tissue collection) and experiments utilizing single experimenters. R.M. and J.L. conducted bioanalytical quantitation of ketamine and metabolites. P.Y. and C.A.Z. performed the western blot experiments. S.M.T. helped design and analyze the electrophysiological experiments. P.G. conducted the experiments and their analysis unless otherwise noted. P.G. and T.D.G. outlined and wrote the paper, which was reviewed by all authors.
Corresponding author
Ethics declarations
Competing interests
C.A.Z. is a co-inventor on a patent for the use of ketamine in major depression and suicidal ideation. P.Z., J.N.H., R.M., C.A.Z. and T.D.G. are co-inventors in patents or patent applications related to the pharmacology and use of (2R,6R)-HNK in the treatment of depression, anxiety, anhedonia, suicidal ideation and post-traumatic stress disorders. R.M. and C.A.Z. have assigned their patent rights to the US government but will share a percentage of any royalties that may be received by the government. P.Z., J.N.H. and T.D.G. have assigned their patent rights to the University of Maryland Baltimore but will share a percentage of any royalties that may be received by the University of Maryland Baltimore. T.D.G. has received research funding from Allergan and Roche Pharmaceuticals and has served as a consultant for FSV7 LLC, during the preceding 3 yr. All other authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Sex of human experimenter effects on stress-related behaviours.
(a) Immobility time measured in the forced-swim test (FST) following saline injections by male and female experimenters in CD1 mice combined from all the experiments performed for the present manuscript where the mean immobility time of each experiment was used (n = 13 experiments; two-sided unpaired t-test, p = 0.007). (b,c) Escape failures following inescapable shock training in the learned helplessness paradigm in Swiss-Webster (CFW) mice handled by a male or a female experimenter (n = 50 mice; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli for B; 11–15 trials q = 0.0099, 16–20 trials q = 0.021, 21–25 trials q = 0.004, 26-30 trials q = 0.0125 and two-sided chi-square test for C, p = 0.046). (d) Average sucrose preference over 48 hours following 10-days of chronic social defeat performed by a male and female experimenter (C57BL/6 J mice; n = 15, 16 mice; two-sided unpaired t-test; p = 0.016) and (e) time spent in light compartment in the light/dark box performed by male and female experimenters (CD1 mice; n = 10 experimenters; n = 20 mice/sex; two-sided unpaired t-test; p = 0.221). Data shown are mean ± S.E.M. * p < 0.05; ** p < 0.01. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 2 Effects of the sex of human experimenter on the antidepressant responses to ketamine.
(a) Immobility time in the forced-swim test (FST) 24 hours post- saline (SAL; 7.5 ml/kg) and ketamine (KET; 10 mg/kg) injections by a male and female experimenter in male CD1 mice (n = 9,8,9,8 mice; two-sided two-way ANOVA followed by |Holm-Sidak test; p = 0.023). (b) Immobility time in the FST 1-hour post-SAL or KET (10 mg/kg) injection by a male or female experimenter in female CD1 mice (n = 10 mice/experimenter/treatment group; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli; q = 0.0096). (c) Immobility time 1-hour post-SAL (7.5 ml/kg) or KET (10 mg/kg) injection by a male or female experimenter in male CD1 mice pre-exposed to a 15-min swim session 24 hours prior to the FST (n = 10,9,10,9 mice; two-sided two-way ANOVA followed by |Holm-Sidak test; p = 0.024). (d) Total escape failures in the learned helplessness paradigm following SAL (7.5 ml/kg) or KET (10 mg/kg) injections by male and female experimenters in male Swiss-webster (CFW) mice (n = 16,14,10,10 mice; two-sided two-way ANOVA followed by |Holm-Sidak test; p = 0.032). (e) Immobility scores in the FST 1-hour following SAL (7.5 ml/kg) or KET (10 mg/kg) injections for each individual experimenter in male CD1 mice, performed at a different institution (n = 2/experimenter). (f) KET dose-response (5, 10, 20, 40 mg/kg) in the FST 1-hr post-injection by a female experimenter in male CD1 mice (n = 9 mice/dose; two-sided one-way ANOVA). Data are shown as mean ± S.E.M. * p < 0.05, **p < 0.01. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 3 Sex of human experimenter effects on NMDAR inhibition dependent behavioural effects.
Distance travelled per 5 min binned intervals in the open-field test in male CD1 mice that received no treatment (HAB; habituation) followed by injections of saline (SAL; 7.5 ml/kg) and then ketamine (KET; 10 mg/kg) administered by a male or female experimenter (n = 8 mice/experimenter/treatment group; two-sided RM two-way ANOVA with Geisser-Greenhouse correction). Immobility time in the forced-swim test 1-hour post-injection following administration of the (c) N-methyl-D-aspartate (NMDAR) receptor antagonist, MK-801 (0.03 mg/kg) (CD1 mice; n = 8,7,8,7 mice; two-sided two-way ANOVA; Treatment effect: p = 0.004), (d,e) ketamine metabolite, (2R,6R)-hydroxynorketamine (HNK; 10 and 50 mg/kg; CD1 mice; n = 2/experimenter for D and; n = 8,9,8,8,9,8 mice and two-sided two-way ANOVA followed by Holm-Sidak test for E; SAL vs 10 mg/k – p = 0.038, SAL vs 50 mg/kg – p = 0.0006), and (f) the classical antidepressant desipramine (DSP; 20 mg/kg) vs SAL (7.5 ml/kg) injections by a male or female experimenters (CD1 mice; n = 10 mice/group; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli; Male: p = 0.001, Female: p = 0.011). Data are shown as mean ± S.E.M. * p < 0.05; ** p < 0.01; *** p < 0.001. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 4 Effects of experimenter scent on the antidepressant-like responses of ketamine.
(a) Elimination of experimenter scent by administering saline (SAL; 7.5 ml/kg) or ketamine (KET; 10 mg/kg) within a biosafety cabinet and testing the mice in the forced-swim test (FST) 1-hour post-injection (CD1 mice; n = 8,7,8,7,8,7,8,7 mice; two-sided three-way ANOVA followed by Holm-Sidak test; p = 0.043). (b) Immobility time in mice tested in the FST 1-hour following injections of SAL (7.5 ml/kg) and KET (10 mg/kg) performed on a male worn t-shirt within the biosafety cabinet by a female experimenter (CD1 mice; n = 10 mice/group; two-sided two-way ANOVA followed by Holm-Sidak test; p = 0.011) Data are shown as mean ± S.E.M. * p < 0.05. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 5 Effects of experimenter scent on the quantitative electroencephalographic oscillations.
Effects of ketamine (KET; 10 mg/kg) administration by male and female experimenters on cortical quantitative electroencephalographic (qEEG) measurements in CD1 mice (n = 7 experimenters; n = 28–29 mice/sex) using the traditionally defined frequency bands (a) alpha (8–12 Hz), (b) beta (13–29 Hz), (c) delta (1–4 Hz), (d) theta (4–8 Hz), (e) gamma (30–100 Hz; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli; Male: Baseline vs 10 q = 0.0121, vs 20 q = 0.0003, vs 30 q = 0.0003, vs 40 q = 0.0005, vs 50 q = 0.0009, vs 60 q = 0.0022; Female: Baseline vs 10 q = 0.057, vs 20 q = 0.0011, vs 30 q = 0.0009, vs 40 q = 0.0023, vs 50 q = 0.0124, vs 60 q = 0.0613; Male vs Female q = 0.0426), and (f) high frequency oscillations (100–160 Hz; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli; Male: Baseline vs 10 q = 0.0315, vs 20 q = 0.0011, vs 30 q = 0.0004, vs 40 q = 0.0004, vs 50 p = 0.0004, vs 60 q = 0.0013; Female: Baseline vs 10 q = 0.0446, vs 20 q = 0.0074, vs 30 q = 0.0004, vs 40 q = 0.0011, vs 50 q = 0.0039, vs 60 p = 0.0539; Male vs Female p = 0.0342). Data are normalised to baseline, and the dashed vertical line indicates the time point of ketamine administration. Data are shown as mean ± S.E.M. +, # p < 0.05; ++, ## p < 0.01; +++, ### p < 0.001. Differences between ketamine response administered by male and female experimenters is indicated by *. Differences between baseline and ketamine is indicated by # for male experimenters and by + for female experimenters. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 6 Effects of corticosterone on the antidepressant responses to ketamine.
(a) Forced-swim test (FST) immobility measured in mice that received metyrapone (30, 50 and 70 mg/kg) prior to KET (10 mg/kg) and tested 1- and 24-hours later (CD1 mice; n = 9,8,8,8,8,8,8,8 mice; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli; VEH-SAL vs VEK-KET p = 0.028, MET 30-SAL vs MET 30-KET p = 0.028, MET 50-SAL vs MET 50-KET p = 0.009, MET 70-SAL vs MET 70-KET p = 0.019 for 1 -hour; MET 70-SAL vs MET 70-KET p = 0.0021 for 24-hours). (b) Immobility time measured in the FST following saline (SAL; 7.5 ml/kg) or ketamine (KET; 10 mg/kg) administration by male and female experimenters to male BALB/cAnNCrl mice (n = 10 mice/treatment group; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli for 1-hour; Male: SAL vs KET q = 0.0075, Female: SAL vs KET q = 0.0069 and two-sided two-way ANOVA for 24-hours; Treatment effect: p = 0.0024). Data are shown as mean ± S.E.M. * p < 0.05; ** p < 0.01; for non-parametric analysis ** q < 0.01. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 7 Effects of the CRF1 antagonist, CP-154,526, on electroencephalographic measures following ketamine administration.
Effects of the corticotropin-releasing factor 1 antagonist (CRF1), CP-154,526 (CP-526; 30 mg/kg) or vehicle (VEH; 1.5 ml/kg) prior to ketamine (KET; 10 mg/kg) administration by a male experimenter on cortical qEEG measurements in CD1 mice (n = 6 mice/treatment group) using the traditionally defined frequency bands (a) alpha (8–12 Hz), (b) beta (13–29 Hz), (c) delta (1–3 Hz), (d) theta power (4–8 Hz), (e) gamma (30–100 Hz; two-sided Kruskal-Wallis followed by correction with two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli (pre-selected comparisons); VEH-KET vs CP-526-KET at 30 min q = 0.0439 and at 40 min q = 0.0518; Baseline 30 min vs 10 min VEH q = 0.0407, vs 10 min KET q = 0.0086, vs 20 min KET q = 0.0068, vs 30 min KET q = 0.0068, vs 40 min KET q = 0.0094, vs 50 min KET q = 0.052, vs 60 min KET q = 0.1150), and (f) high frequency oscillations (100–160 Hz). Data are normalised to baseline. The first dashed vertical line indicates the time point of VEH or CP-526 administration and the second dashed vertical line indicates the time point of KET administration. Data are shown as mean ± S.E.M. *, # p < 0.05, ***, ### p < 0.001. Differences between mice pre-treated with CP-526 and VEH are indicated by *. Differences between the baseline and treatment in mice that received VEH prior to KET are indicated with #. For detailed statistics information, see Supplementary Table 1.
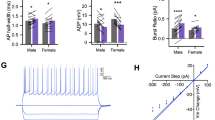
Extended Data Fig. 8 Effects of the combined CRF and (2R,6R)-hydroxynorketamine on field excitatory postsynaptic potentials in hippocampal slices in the SC-CA1 pathway.
(a) Representative traces of Schaffer collateral-hippocampal CA1 (SC-CA1) field excitatory postsynaptic potentials (fEPSPs) and (b) quantification of fEPSPs slopes following SC-CA1 pathway stimulation during wash-in with Saline (SAL; n = 11 slices), corticotropin-releasing factor (CRF; 125 nM; n = 12 slices), (2R,6R)-hydroxynorketamine (HNK; 15 μM; n = 11 slices) or combined CRF (125 nM) + (2R,6R)-HNK (15 μM) (n = 11 slices; two-sided one-way ANOVA followed by Holm-Sidak test; SAL vs HNK p = 0.0053, SAL vs CRF+ HNK p = 0.0003, CRF vs HNK p = 0.0043, CRF vs CRF + HNK p = 0.0002). Data are shown as mean ± S.E.M. ** p < 0.01. For detailed statistics information, see Supplementary Table 1.
Extended Data Fig. 9 Identification of EC to CA1 projections.
(a) Representative images of the injection site at ventral CA1 with retrograde conjugated cholera toxin and the (b,c) labelling in the anterior and posterior entorhinal cortex (EC). Representative RNAscope images from the EC revealing (d) DAPI labeling, (e) CRF transcript labelling, (f) CTb labelling and (g) the co-labelling between the tracer and CRF transcripts. (h) Quantification of RNA scope and tracer co-labelling at the anterior (aEC) and posterior EC (pEC) and the lateral (LEC) and medial EC (MEC) (n = 8-9 samples from 2 animals thus these findings were independently replicated twice).
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2 and Figs. 1 and 2.
Supplementary Video
Representative video clip assessing real-time place preference to male- and female-worn t-shirts. All t-shirts were purchased new, identical other than the size, and washed in a nonscented, hypoallergenic detergent before the experiment. T-shirts were worn for 24 h before collection. Control t-shirts were unworn. Mice show aversion to the chamber with the male worn t-shirt and preference to the chamber with the female worn t-shirt.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Rights and permissions
About this article
Cite this article
Georgiou, P., Zanos, P., Mou, TC.M. et al. Experimenters’ sex modulates mouse behaviors and neural responses to ketamine via corticotropin releasing factor. Nat Neurosci 25, 1191–1200 (2022). https://doi.org/10.1038/s41593-022-01146-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-022-01146-x
This article is cited by
-
Using mice from different breeding sites fails to improve replicability of results from single-laboratory studies
Lab Animal (2024)
-
Therapeutic mechanisms of psychedelics and entactogens
Neuropsychopharmacology (2024)
-
Sex dependence of opioid-mediated responses to subanesthetic ketamine in rats
Nature Communications (2024)
-
Sexual differences in locus coeruleus neurons and related behavior in C57BL/6J mice
Biology of Sex Differences (2023)
-
Audible pain squeaks can mediate emotional contagion across pre-exposed rats with a potential effect of auto-conditioning
Communications Biology (2023)