Abstract
Knowledge of the effect of foods on gut microbiota composition and functionality is expanding. To isolate the effect of single foods and/or single nutrients (i.e., fiber, polyphenols), this protocol describes an in vitro batch fermentation procedure to be carried out after an in vitro gastrointestinal digestion. Therefore, this is an extension of the previous protocol described by Brodkorb et al. (2019) for studying in vitro digestion. The current protocol uses an oligotrophic fermentation medium with peptone and a high concentration of fecal inoculum from human fecal samples both to provide the microbiota and as the main source of nutrients for the bacteria. This protocol is recommended for screening work to be performed when many food samples are to be studied. It has been used successfully to study gut microbiota fermentation of different foodstuffs, giving insights into their functionality, community structure or ability to degrade particular substances, which can contribute to the development of personalized nutrition strategies. The procedure does not require a specific level of expertise. The protocol takes 4–6 h for preparation of fermentation tubes and 20 h for incubation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
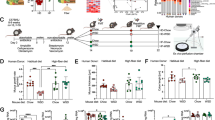
The data shown in Fig. 4 are available from the supporting primary research paper previously published by Pérez-Burillo et al.17. The data presented in Figs. 2 and 3 were generated for this protocol. Source data are provided with this paper.
References
Flint, H. J., Duncan, S. H., Scott, K. P. & Louis, P. Links between diet, gut microbiota composition and gut metabolism. Proc. Nutr. Soc. 74, 13–22 (2015).
Salazar, N. et al. Exopolysaccharides produced by Bifidobacterium longum IPLA E44 and Bifidobacterium animalis subsp. lactis IPLA R1 modify the composition and metabolic activity of human faecal microbiota in pH-controlled batch cultures. Int. J. Food Microbiol. 135, 260–267 (2009).
Power, S. E., O’Toole, P. W., Stanton, C., Ross, R. P. & Fitzgerald, G. F. Intestinal microbiota, diet and health. Br. J. Nutr. 111, 387–402 (2014).
Shankar, V. et al. Differences in gut metabolites and microbial composition and functions between Egyptian and U.S. children are consistent with their diets. mSystems 2, e00169–16 (2017).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2013).
De Filippo, C. et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl Acad. Sci. USA 107, 14691–14696 (2010).
Venema, K. & van den Abbeele, P. Experimental models of the gut microbiome. Best Pract. Res. Clin. Gastroenterol. 27, 115–126 (2013).
Molly, K., Vande Woestyne, M. & Verstraete, W. Development of a 5-step multi-chamber reactor as a simulation of the human intestinal microbial ecosystem. Appl. Microbiol. Biotechnol. 39, 254–258 (1993).
Agans, R. et al. Dietary fatty acids sustain the growth of the human gut microbiota. Appl. Environ. Microbiol. 84, e01525–18 (2018).
Wiese, M. et al. CoMiniGut—a small volume in vitro colon model for the screening of gut microbial fermentation processes. PeerJ 6, e4268 (2018).
Ludwig, I. A., Paz de Peña, M., Concepción, C. & Alan, C. Catabolism of coffee chlorogenic acids by human colonic microbiota: colonic catabolism of coffee chlorogenic acids. BioFactors 39, 623–632 (2013).
Coles, L. T., Moughan, P. J. & Darragh, A. J. In vitro digestion and fermentation methods, including gas production techniques, as applied to nutritive evaluation of foods in the hindgut of humans and other simple-stomached animals. Anim. Feed Sci. Technol. 123–124, 421–444 (2005).
Wang, M. et al. In vitro colonic fermentation of dietary fibers: fermentation rate, short-chain fatty acid production and changes in microbiota. Trends Food Sci. Technol. 88, 1–9 (2019).
Mould, F. L., Morgan, R., Kliem, K. E. & Krystallidou, E. A review and simplification of the in vitro incubation medium. Anim. Feed Sci. Technol. 123–124, 155–172 (2005).
Brodkorb, A. et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 14, 991–1014 (2019).
Pérez-Burillo, S., Rufián-Henares, J. A. & Pastoriza, S. Towards an improved global antioxidant response method (GAR+): physiological-resembling in vitro digestion-fermentation method. Food Chem 239, 1253–1262 (2018).
Pérez-Burillo, S. et al. Effect of food thermal processing on the composition of the gut microbiota. J. Agric. Food Chem. 66, 11500–11509 (2018).
Pérez-Burillo, S. et al. Effect of in vitro digestion-fermentation on green and roasted coffee bioactivity: the role of the gut microbiota. Food Chem. 279, 252–259 (2019).
Pérez-Burillo, S. et al. Potential probiotic salami with dietary fiber modulates antioxidant capacity, short chain fatty acid production and gut microbiota community structure. LWT 105, 355–362 (2019).
Pérez-Burillo, S. et al. Spent coffee grounds extract, rich in mannooligosaccharides, promotes a healthier gut microbial community in a dose-dependent manner. J. Agric. Food Chem. 67, 2500–2509 (2019).
Pérez-Burillo, S., Rajakaruna, S., Pastoriza, S., Paliy, O. & Rufián-Henares, J. A. Bioactivity of food melanoidins is mediated by gut microbiota. Food Chem. 316, 126309 (2020).
Rocchetti, G. et al. Transformation of polyphenols found in pigmented gluten-free flours during in vitro large intestinal fermentation. Food Chem. 298, 125068 (2019).
Jin, J. B. et al. Supplementation with Chlorella vulgaris, Chlorella protothecoides, and Schizochytrium sp. increases propionate-producing bacteria in in vitro human gut fermentation. J. Sci. Food Agric. 100, 2938–2945 (2020).
Selma, M. V., Espín, J. C. & Tomás-Barberán, F. A. Interaction between phenolics and gut microbiota: role in human health. J. Agric. Food Chem. 57, 6485–6501 (2009).
Rowland, I. et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur. J. Nutr. 57, 1–24 (2018).
Tomas‐Barberan, F. et al. In vitro transformation of chlorogenic acid by human gut microbiota. Mol. Nutr. Food Res. 58, 1122–1131 (2014).
Saura‐Calixto, F. et al. Proanthocyanidin metabolites associated with dietary fibre from in vitro colonic fermentation and proanthocyanidin metabolites in human plasma. Mol. Nutr. Food Res. 54, 939–946 (2010).
Roowi, S. et al. Green tea flavan-3-ols: colonic degradation and urinary excretion of catabolites by humans. J. Agric. Food Chem. 58, 1296–1304 (2010).
Jaganath, I. B., Mullen, W., Lean, M. E. J., Edwards, C. A. & Crozier, A. In vitro catabolism of rutin by human fecal bacteria and the antioxidant capacity of its catabolites. Free Radic. Biol. Med. 47, 1180–1189 (2009).
Serra, A. et al. Metabolic pathways of the colonic metabolism of flavonoids (flavonols, flavones and flavanones) and phenolic acids. Food Chem. 130, 383–393 (2012).
Pinta, M. N. et al. In vitro gut metabolism of [U-13C]-quinic acid, the other hydrolysis product of chlorogenic acid. Mol. Nutr. Food Res. 62, 1800396 (2018).
Hidalgo, M. et al. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. J. Agric. Food Chem. 60, 3882–3890 (2012).
Marín, L., Miguélez, E. M., Villar, C. J. & Lombó, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: antimicrobial properties. Biomed. Res. Int. https://doi.org/10.1155/2015/905215 (2015).
Stevens, J. F. & Maier, C. S. The chemistry of gut microbial metabolism of polyphenols. Phytochem. Rev. 15, 425–444 (2016).
Fuertes, Á. et al. Adaptation of the human gut microbiota metabolic network during the first year after birth. Front. Microbiol. 10, 848 (2019).
Ríos-Covián, D. et al. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 7, 185 (2016).
Shen, Q., Chen, Y. A. & Tuohy, K. M. A comparative in vitro investigation into the effects of cooked meats on the human faecal microbiota. Anaerobe 16, 572–577 (2010).
Poelaert, C. et al. Cooking has variable effects on the fermentability in the large intestine of the fraction of meats, grain legumes, and insects that is resistant to digestion in the small intestine in an in vitro model of the pig’s gastrointestinal tract. J. Agric. Food Chem. 65, 435–444 (2017).
Lee, D. K. et al. The combination of mixed lactic acid bacteria and dietary fiber lowers serum cholesterol levels and fecal harmful enzyme activities in rats. Arch. Pharm. Res. 34, 23–29 (2011).
Molska, M. & Regula, J. Potential mechanisms of probiotics action in the prevention and treatment of colorectal cancer. Nutrients 11, 2453 (2019).
Pham, V. T. & Mohajeri, M. H. The application of in vitro human intestinal models on the screening and development of pre- and probiotics. Benef. Microbes 9, 725–742 (2018).
Gu, F. et al. In vitro fermentation behavior of isomalto/malto-polysaccharides using human fecal inoculum indicates prebiotic potential. Mol. Nutr. Food Res. 62, e1800232 (2018).
Fehlbaum, S. et al. In vitro fermentation of selected prebiotics and their effects on the composition and activity of the adult gut microbiota. Int. J. Mol. Sci. 19, 3097 (2018).
Pérez-Burillo, S. et al. Potential probiotic salami with dietary fiber modulates metabolism and gut microbiota in a human intervention study. J. Funct. Foods 66, 103790 (2020).
Kolodziejczyk, A. A., Zheng, D. & Elinav, E. Diet–microbiota interactions and personalized nutrition. Nat. Rev. Microbiol. 17, 742–753 (2019).
Magnúsdóttir, S. et al. Generation of genome-scale metabolic reconstructions for 773 members of the human gut microbiota. Nat. Biotechnol. 35, 81–89 (2017).
Coyte, K. Z. & Rakoff-Nahoum, S. Understanding competition and cooperation within the mammalian gut microbiome. Curr. Biol. 29, R538–R544 (2019).
Payne, A. N., Zihler, A., Chassard, C. & Lacroix, C. Advances and perspectives in in vitro human gut fermentation modeling. Trends Biotechnol. 30, 17–25 (2012).
Cohen, S. M. Human relevance of animal carcinogenicity studies. Regul. Toxicol. Pharmacol. 21, 75–80 (1995). discussion 81-86.
Eckburg, P. B. et al. Diversity of the human intestinal microbial flora. Science 308, 1635–1638 (2005).
Ley, R. E. et al. Obesity alters gut microbial ecology. Proc. Natl Acad. Sci. USA 102, 11070–11075 (2005).
Belkaid, Y. & Hand, T. W. Role of the microbiota in immunity and inflammation. Cell 157, 121–141 (2014).
Shankar, V. et al. The networks of human gut microbe-metabolite associations are different between health and irritable bowel syndrome. ISME J. 9, 1899–1903 (2015).
Aguirre, M. & Venema, K. Challenges in simulating the human gut for understanding the role of the microbiota in obesity. Benef. Microbes 8, 31–53 (2017).
Gratton, J. et al. Optimized sample handling strategy for metabolic profiling of human feces. Anal. Chem. 88, 4661–4668 (2016).
Gorzelak, M. A. et al. Methods for improving human gut microbiome data by reducing variability through sample processing and storage of stool. PLoS ONE 10, e0134802 (2015).
Long, W. et al. Differential responses of gut microbiota to the same prebiotic formula in oligotrophic and eutrophic batch fermentation systems. Sci. Rep. 5, 1–11 (2015).
Paliy, O. & Shankar, V. Application of multivariate statistical techniques in microbial ecology. Mol. Ecol. 25, 1032–1057 (2016).
Pérez-Burillo, S., Rufián-Henares, J. A. & Pastoriza, S. Effect of home cooking on the antioxidant capacity of vegetables: relationship with Maillard reaction indicators. Food Res. Int. 121, 514–523 (2019).
Quince, C., Walker, A. W., Simpson, J. T., Loman, N. J. & Segata, N. Shotgun metagenomics, from sampling to analysis. Nat. Biotechnol. 35, 833–844 (2017).
Morales, F. J., Somoza, V. & Fogliano, V. Physiological relevance of dietary melanoidins. Amino Acids 42, 1097–1109 (2012).
Bauer, E. & Thiele, I. From network analysis to functional metabolic modeling of the human gut microbiota. mSystems 3, 00209–00217 (2018).
Fernandes, J., Su, W., Rahat-Rozenbloom, S., Wolever, T. M. S. & Comelli, E. M. Adiposity, gut microbiota and faecal short chain fatty acids are linked in adult humans. Nutr. Diabetes 4, e121–e121 (2014).
Bjerrum, J. T. et al. Metabonomics of human fecal extracts characterize ulcerative colitis, Crohn’s disease and healthy individuals. Metabolomics 11, 122–133 (2015).
Nistal, E. et al. Differences in faecal bacteria populations and faecal bacteria metabolism in healthy adults and celiac disease patients. Biochimie 94, 1724–1729 (2012).
Ze, X., Duncan, S. H., Louis, P. & Flint, H. J. Ruminococcus bromii is a keystone species for the degradation of resistant starch in the human colon. ISME J 6, 1535–1543 (2012).
Zhou, L., Xie, M., Yang, F. & Liu, J. Antioxidant activity of high purity blueberry anthocyanins and the effects on human intestinal microbiota. LWT 117, 108621 (2020).
Gonçalves, G. A. et al. Effects of in vitro gastrointestinal digestion and colonic fermentation on a rosemary (Rosmarinus officinalis L) extract rich in rosmarinic acid. Food Chem. 271, 393–400 (2019).
Chen, Y., Chang, S. K. C., Zhang, Y., Hsu, C.-Y. & Nannapaneni, R. Gut microbiota and short chain fatty acid composition as affected by legume type and processing methods as assessed by simulated in vitro digestion assays. Food Chem. 312, 126040 (2020).
del Hierro, J. N. et al. In vitro colonic fermentation of saponin-rich extracts from quinoa, lentil, and fenugreek. effect on sapogenins yield and human gut microbiota. J. Agric. Food Chem. 68, 106–116 (2020).
Zhang, X. et al. Phytochemical profile, bioactivity, and prebiotic potential of bound phenolics released from rice bran dietary fiber during in vitro gastrointestinal digestion and colonic fermentation. J. Agric. Food Chem. 67, 12796–12805 (2019).
Wang, M. et al. Purified fraction of polysaccharides from Fuzhuan brick tea modulates the composition and metabolism of gut microbiota in anaerobic fermentation in vitro. Int. J. Biol. Macromol. 140, 858–870 (2019).
Rui, Y. et al. Simulated digestion and fermentation in vitro by human gut microbiota of intra- and extra-cellular polysaccharides from Aspergillus cristatus. LWT 116, 108508 (2019).
Chen, L. et al. Simulated digestion and fermentation in vitro by human gut microbiota of polysaccharides from Helicteres angustifolia L. Int. J. Biol. Macromol. 141, 1065–1071 (2019).
Huang, F. et al. Structural characterization and in vitro gastrointestinal digestion and fermentation of litchi polysaccharide. Int. J. Biol. Macromol. 140, 965–972 (2019).
Acknowledgements
This study was supported by the EU project Stance4Health (contract no. 816303) and by the Plan propio de Investigación y Transferencia of the University of Granada under the program ‘Intensificación de la Investigación, modalidad B’ to J.A.R.-H.
Author information
Authors and Affiliations
Contributions
S.P.-B., S.M., and J.A.R.-H. wrote the manuscript. B.N.-P., A.J.V.-M., D.H.-N., A.L.-M. and S.P. contributed to the writing of the manuscript. S.P.-B., S.M., B.N.-P., A.J.V.-M., D.H.-N. and A.L.-M. contributed to formal analysis and investigation; S.P.-B. developed the methodology. S.P. and J.A.R.-H. supervised the work. J.A.R.-H. obtained funding and coordinates the EU project Stance4Health.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks Francisco Tomás-Barberán and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Pérez-Burillo, S. et al. Food Chem. 239, 1253–1262 (2018): https://doi.org/10.1016/j.foodchem.2017.07.024
Pérez-Burillo, S. et al. Food Chem. 279, 252–259 (2019): https://doi.org/10.1016/j.foodchem.2018.11.137
Pérez-Burillo, S., et al. Food Chem. 316, 126309 (2020): https://doi.org/10.1016/j.foodchem.2020.126309
This protocol is an extension to: Nat. Protoc. 14, 991–1014 (2019): https://doi.org/10.1038/s41596-018-0119-1
Supplementary information
Source data
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Rights and permissions
About this article
Cite this article
Pérez-Burillo, S., Molino, S., Navajas-Porras, B. et al. An in vitro batch fermentation protocol for studying the contribution of food to gut microbiota composition and functionality. Nat Protoc 16, 3186–3209 (2021). https://doi.org/10.1038/s41596-021-00537-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-021-00537-x
This article is cited by
-
Study of High Molecular Weight Hemicelluloses Recovered from Black Spruce Mill Waste for Potential Value-Added Applications in Foods
Waste and Biomass Valorization (2024)
-
Human immune and gut microbial parameters associated with inter-individual variations in COVID-19 mRNA vaccine-induced immunity
Communications Biology (2023)
-
Beneficial metabolic transformations and prebiotic potential of hemp bran and its alcalase hydrolysate, after colonic fermentation in a gut model
Scientific Reports (2023)
-
Designs and methodologies to recreate in vitro human gut microbiota models
Bio-Design and Manufacturing (2023)
-
Prediction of degradation pathways of phenolic compounds in the human gut microbiota through enzyme promiscuity methods
npj Systems Biology and Applications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.