Abstract
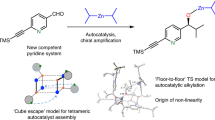
Asymmetric amplification is a curious phenomenon that is believed to play a key role in the emergence of biological homochirality, and thus of life itself. In asymmetric catalysis, it is achieved via positive nonlinear effects, which allow high product enantiomeric excesses with a non-enantiopure catalyst. However, it has also been proposed that non-enantiopure catalysts may be even more enantioselective than their enantiopure counterparts, although such a case has never been experimentally observed so far. Here, we present an example of such a hyperpositive nonlinear effect in asymmetric catalysis. We found that addition of dialkylzinc reagents to benzaldehyde gave higher product enantiomeric excesses with only partially resolved chiral N-benzyl-ephedrine ligands. A mechanistic study was carried out and our results point towards a two-component catalysis, where mononuclear as well as aggregated catalysts are in equilibrium and in competition. These results introduce an unprecedented class of asymmetric amplification in enantioselective catalysis.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Complete experimental procedures, compound characterization data and kinetic rate profiles are available in the Supplementary Information.
References
Carroll, J. D. A new definition of life. Chirality 21, 354–358 (2009).
Frank, F. C. On spontaneous asymmetric synthesis. Biochim. Biophys. Acta 11, 459–463 (1953).
Noorduin, W. L. et al. Complete chiral symmetry breaking of an amino acid derivative directed by circularly polarized light. Nat. Chem. 1, 729–732 (2009).
Viedma, C. Chiral symmetry breaking during crystallization: complete chiral purity induced by nonlinear autocatalysis and recycling. Phys. Rev. Lett. 94, 065504 (2005).
Soai, K., Shibata, T., Morioka, H. & Choji, K. Asymmetric autocatalysis and amplification of enantiomeric excess of a chiral molecule. Nature 378, 767–768 (1995).
Soai, K., Kawasaki, T. & Matsumoto, A. Asymmetric autocatalysis of pyrimidyl alkanol and related compounds. Self-replication, amplification of chirality and implication for the origin of biological enantioenriched chirality. Tetrahedron 74, 1973–1990 (2018).
Blackmond Donna, G. The origin of biological homochirality. Philos. Trans. R. Soc. B 366, 2878–2884 (2011).
Brewer, A. & Davis, A. P. Chiral encoding may provide a simple solution to the origin of life. Nat. Chem. 6, 569–574 (2014).
Bada, J. L. Origins of homochirality. Nature 374, 594–595 (1995).
Siegel, J. S. Chemistry: shattered mirrors. Nature 419, 346–347 (2002).
Guillaneux, D., Zhao, S.-H., Samuel, O., Rainford, D. & Kagan, H. B. Nonlinear effects in asymmetric catalysis. J. Am. Chem. Soc. 116, 9430–9439 (1994).
Girard, C. & Kagan, H. B. Nonlinear effects in asymmetric synthesis and stereoselective reactions: ten years of investigation. Angew. Chem. Int. Ed. 37, 2922–2959 (1998).
Satyanarayana, T., Abraham, S. & Kagan, H. B. Nonlinear effects in asymmetric catalysis. Angew. Chem. Int. Ed. 48, 456–494 (2009).
Puchot, C. et al. Nonlinear effects in asymmetric synthesis. Examples in asymmetric oxidations and aldolization reactions. J. Am. Chem. Soc. 108, 2353–2357 (1986).
Wynberg, H. & Feringa, B. Enantiomeric recognition and interactions. Tetrahedron 32, 2831–2834 (1976).
Kitamura, M., Okada, S., Suga, S. & Noyori, R. Enantioselective addition of dialkylzincs to aldehydes promoted by chiral amino alcohols. Mechanism and nonlinear effect. J. Am. Chem. Soc. 111, 4028–4036 (1989).
Noyori, R., Suga, S., Oka, H. & Kitamura, M. Self and nonself recognition of chiral catalysts: the origin of nonlinear effects in the amino-alcohol catalyzed asymmetric addition of diorganozincs to aldehydes. Chem. Rec. 1, 85–100 (2001).
Kitamura, M., Suga, S., Oka, H. & Noyori, R. Quantitative analysis of the chiral amplification in the amino alcohol-promoted asymmetric alkylation of aldehydes with dialkylzincs. J. Am. Chem. Soc. 120, 9800–9809 (1998).
Kitamura, M., Oka, H. & Noyori, R. Asymmetric addition of dialkylzincs to benzaldehyde derivatives catalyzed by chiral β-amino alcohols. Evidence for the monomeric alkylzinc aminoalkoxide as catalyst. Tetrahedron 55, 3605–3614 (1999).
Coote, S. J., Davies, S. G., Fletcher, A. M., Roberts, P. M. & Thomson, J. E. Enantiospecific stereodivergent synthesis of trans- and cis-N(2),3-dimethyl-4-phenyl-1,2,3,4-tetrahydroisoquinolines. Chem. Asian J. 5, 589–604 (2010).
Burés, J. A simple graphical method to determine the order in catalyst. Angew. Chem. Int. Ed. 55, 2028–2031 (2016).
Burés, J. Variable time normalization analysis: general graphical elucidation of reaction orders from concentration profiles. Angew. Chem. Int. Ed. 55, 16084–16087 (2016).
Nielsen, C. D.-T. & Burés, J. Visual kinetic analysis. Chem. Sci. 10, 348–353 (2019).
Neufeld, R. & Stalke, D. Accurate molecular weight determination of small molecules via DOSY-NMR by using external calibration curves with normalized diffusion coefficients. Chem. Eur. J. 6, 3354–3364 (2015).
Bachmann, S., Gernert, B. & Stalke, D. Solution structures of alkali metal cyclopentadienides in THF estimated by ECC-DOSY NMR-spectroscopy (incl. software). Chem. Commun. 52, 12861–12864 (2016).
Bachmann, S., Neufeld, R., Dzemski, M. & Stalke, D. New external calibration curves (ECCs) for the estimation of molecular weights in various common NMR solvents. Chem. Eur. J. 22, 8462–8465 (2016).
Kreyenschmidt, A.-K., Bachmann, S., Niklas, T. & Stalke, D. Molecular weight estimation of molecules incorporating heavier elements from van-der-Waals corrected ECC-DOSY. ChemistrySelect 2, 6957–6960 (2017).
Chaloner, P. A., Langadianou, E. & Perera, S. A. R. Enantioselective reaction of diethylzinc with arenecarbaldehydes in the presence of ephedrine derivatives. J. Chem. Soc. Perkin Trans. 1, 2731–2735 (1991).
Zhang, H. & Shing Chan, K. Dramatic temperature effect in asymmetric catalysis in the enantioselective addition of diethylzinc to aldehydes. J. Chem. Soc. Perkin Trans. 1, 381–382 (1999).
Kang, J., Kim, J. B., Kim, J. & Kim, D. The interaction of chiral amino thiols with organozinc reagents and aldehydes: a mechanism of amino thiol-catalyzed addition of organozinc reagents to aldehydes. Bull. Korean Chem. Soc. 19, 475–481 (1998).
Coates, G. E. & Ridley, D. Reactions between some organozinc compounds and 2-dimethylaminoethanol, acetoxime, phenylisocyanate and benzophenone. Some observations on the methylzinc methoxide tetramer. J. Chem. Soc. A 0, 1064–1069 (1966).
Boersma, J. & Noltes, J. G. Investigations on organozinc compounds X. Coordination chemistry of organozinc compounds RZnX: organozinc-oxygen derivatives of potentially bidentate ligands. J. Organomet. Chem. 13, 291–299 (1968).
van der Schaaf, P. A. et al. Organozinc complexes with monoanionic chelating phenolates or 2-pyridylmethanolates. Molecular structure of [Zn(CH2SiMe3){OCH2(2-Py)}]4. Organometallics 12, 3624–3629 (1993).
Fitzpatrick, K., Hulst, R. & Kellogg, R. M. Thiol and disulfide derivatives of ephedra alkaloids 2: a mechanistic study of their effect on the addition of diethyl zinc to benzaldehyde. Tetrahedron Asymmetry 6, 1861–1864 (1995).
Acknowledgements
We acknowledge the CNRS and the Ministère de l’Enseignement Supérieur et de la Recherche (MESR) for a PhD grant to Y.G. This work is part of the Strasbourg IDEX programme and we thank NIE Labex for support (ANR-11-LABX-0058_NIE). We thank L. H. Gade (University of Heidelberg) and J. Moran (University of Strasbourg) for helpful comments on this work.
Author information
Authors and Affiliations
Contributions
Y.G. performed the synthetic experiments and data analyses. A.M.-F. and T.A. participated in data analyses. S.B.-L. conceptualized and supervised the study and wrote the manuscript with Y.G.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, Figs. 1–7, Tables 1–5 and references.
Rights and permissions
About this article
Cite this article
Geiger, Y., Achard, T., Maisse-François, A. et al. Hyperpositive nonlinear effects in asymmetric catalysis. Nat Catal 3, 422–426 (2020). https://doi.org/10.1038/s41929-020-0441-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-020-0441-1
This article is cited by
-
Divergence of catalytic systems in the zinc-catalysed alkylation of benzaldehyde mediated by chiral proline-based ligands
Nature Synthesis (2024)
-
Symmetry Breaking by Consecutive Amplification: Efficient Paths to Homochirality
Origins of Life and Evolution of Biospheres (2022)
-
Unmasking unusual behaviour
Nature Reviews Chemistry (2020)