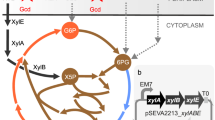
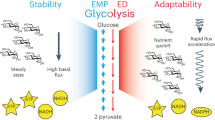
Abstract
The reprogramming of metabolism in response to switching the carbon source from glucose to non-preferred carbon sources is well-studied for yeast. However, understanding how metabolic networks respond to utilize a non-natural carbon source such as xylose is limited due to the incomplete knowledge of cellular response mechanisms. Here we applied a combination of metabolic engineering, systems biology and adaptive laboratory evolution to gain insights into how yeast can perform a global rewiring of cellular processes to efficiently accompany metabolic transitions. Through metabolic engineering, we substantially enhanced the cell growth on xylose after the growth on glucose. Transcriptome analysis of the engineered strains demonstrated that multiple pathways were involved in the cellular reprogramming. Through genome resequencing of the evolved strains and reverse engineering, we further identified that SWI/SNF chromatin remodelling, osmotic response and aldehyde reductase were responsible for the improved growth. Combined, our analysis showed that glycerol-3-phosphate and xylitol serve as two key metabolites that affect cellular adaptation to growth on xylose.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The RNA-seq raw data are available at the Genome Expression Omnibus website (https://www.ncbi.nlm.nih.gov/geo/) using series number GSE151478. The genome sequence data of the evolved strains used in this article are available at the Sequence Read Archive website (https://www.ncbi.nlm.nih.gov/sra) with the accession number PRJNA636080. All other data that support the findings in this study are available upon reasonable request. All plasmids and strains used in this study can be obtained from the corresponding author under a material transfer agreement. Source data are provided with this paper.
References
Botstein, D., Chervitz, S. A. & Cherry, M. Yeast as a model organism. Science 277, 1259–1260 (1997).
Nielsen, J. & Keasling, J. D. Engineering cellular metabolism. Cell 164, 1185–1197 (2016).
Kavšček, M., Stražar, M., Curk, T., Natter, K. & Petrovič, U. Yeast as a cell factory: current state and perspectives. Microb. Cell Fact. 14, 1–10 (2015).
Nielsen, J. Yeast systems biology: model organism and cell factory. Biotechnol. J. 14, 1800421 (2019).
Frendt, S. M. et al. Unraveling condition‐dependent networks of transcription factors that control metabolic pathway activity in yeast. Mol. Syst. Biol. 6, 432 (2010).
Kayikci, Ö. & Nielsen, J. Glucose repression in Saccharomyces cerevisiae. FEMS Yeast Res. 15, fov068 (2015).
Ramsey, S. A. et al. Dual feedback loops in the GAL regulon suppress cellular heterogeneity in yeast. Nat. Genet. 38, 1082–1087 (2006).
Zampar, G. G. et al. Temporal system‐level organization of the switch from glycolytic to gluconeogenic operation in yeast. Mol. Syst. Biol. 9, 651 (2013).
Li, J. et al. Molecular mechanism of environmental d-xylose perception by a XylFII–LytS complex in bacteria. Proc. Natl Acad. Sci. USA 114, 8235–8240 (2017).
Kwak, S. & Jin, Y.-S. Production of fuels and chemicals from xylose by engineered Saccharomyces cerevisiae: a review and perspective. Microb. Cell Fact. 16, 82 (2017).
Li, X., Chen, Y. & Nielsen, J. Harnessing xylose pathways for biofuels production. Curr. Opin. Biotechnol. 57, 56–65 (2019).
Jeffries, T. W. Engineering yeasts for xylose metabolism. Curr. Opin. Biotechnol. 17, 320–326 (2006).
Harhangi, H. R. et al. Xylose metabolism in the anaerobic fungus Piromyces sp. strain E2 follows the bacterial pathway. Arch. Microbiol. 180, 134–141 (2003).
Moon, J., Liu, Z. L., Ma, M. & Slininger, P. J. New genotypes of industrial yeast Saccharomyces cerevisiae engineered with YXI and heterologous xylose transporters improve xylose utilization and ethanol production. Biocatal. Agric. Biotechnol. 2, 247–254 (2013).
Lee, S.-M., Jellison, T. & Alper, H. S. Directed evolution of xylose isomerase for improved xylose catabolism and fermentation in the yeast Saccharomyces cerevisiae. Appl. Environ. Microbiol. 78, 5708–5716 (2012).
Brat, D., Boles, E. & Wiedemann, B. Functional expression of a bacterial xylose isomerase in Saccharomyces cerevisiae. Appl. Environ. Microbiol. 75, 2304–2311 (2009).
Ha, S.-J., Kim, S. R., Choi, J.-H., Park, M. S. & Jin, Y.-S. Xylitol does not inhibit xylose fermentation by engineered Saccharomyces cerevisiae expressing xylA as severely as it inhibits xylose isomerase reaction in vitro. Appl. Microbiol. Biotechnol. 92, 77–84 (2011).
Dos Santos, L. V. et al. Unraveling the genetic basis of xylose consumption in engineered Saccharomyces cerevisiae strains. Sci. Rep. 6, 38676 (2016).
de Figueiredo Vilela, L. et al. Functional expression of Burkholderia cenocepacia xylose isomerase in yeast increases ethanol production from a glucose–xylose blend. Bioresour. Technol. 128, 792–796 (2013).
Teunissen, A. W. R. H. & De Bont, J. A. M. Xylose isomerase genes and their use in fermentation of pentose sugars. US patent 9,334,488 (2016).
Träff, K., Cordero, R. O., Van Zyl, W. & Hahn-Hägerdal, B. Deletion of the GRE3 aldose reductase gene and its influence on xylose metabolism in recombinant strains of Saccharomyces cerevisiae expressing the xylA and XKS1 genes. Appl. Environ. Microbiol. 67, 5668–5674 (2001).
Hou, J., Jiao, C., Peng, B., Shen, Y. & Bao, X. Mutation of a regulator Ask10p improves xylose isomerase activity through up-regulation of molecular chaperones in Saccharomyces cerevisiae. Metab. Eng. 38, 241–250 (2016).
Xu, H. et al. PHO13 deletion-induced transcriptional activation prevents sedoheptulose accumulation during xylose metabolism in engineered Saccharomyces cerevisiae. Metab. Eng. 34, 88–96 (2016).
Sato, T. K. et al. Directed evolution reveals unexpected epistatic interactions that alter metabolic regulation and enable anaerobic xylose use by Saccharomyces cerevisiae. PLoS Genet. 12, e1006372 (2016).
Wenger, J. W., Schwartz, K. & Sherlock, G. Bulk segregant analysis by high-throughput sequencing reveals a novel xylose utilization gene from Saccharomyces cerevisiae. PLoS Genet. 6, e1000942 (2010).
Young, E., Poucher, A., Comer, A., Bailey, A. & Alper, H. Functional survey for heterologous sugar transport proteins, using Saccharomyces cerevisiae as a host. Appl. Environ. Microbiol. 77, 3311–3319 (2011).
Wang, C. et al. Cloning and characterization of heterologous transporters in Saccharomyces cerevisiae and identification of important amino acids for xylose utilization. Metab. Eng. 30, 79–88 (2015).
Apel, A. R., Ouellet, M., Szmidt-Middleton, H., Keasling, J. D. & Mukhopadhyay, A. Evolved hexose transporter enhances xylose uptake and glucose/xylose co-utilization in Saccharomyces cerevisiae. Sci. Rep. 6, 19512 (2016).
Madhani, H. D. & Fink, G. R. Combinatorial control required for the specificity of yeast MAPK signaling. Science 275, 1314–1317 (1997).
Domitrovic, T. et al. Structural and functional study of YER067W, a new protein involved in yeast metabolism control and drug resistance. PLoS ONE 5, e11163 (2010).
Guaragnella, N. & Butow, R. A. ATO3 encoding a putative outward ammonium transporter is an RTG-independent retrograde responsive gene regulated by GCN4 and the Ssy1-Ptr3-Ssy5 amino acid sensor system. J. Biol. Chem. 278, 45882–45887 (2003).
Edmunds, J. W. & Mahadevan, L. C. MAP kinases as structural adaptors and enzymatic activators in transcription complexes. J. Cell Sci. 117, 3715–3723 (2004).
Hinnebusch, A. G. Translational regulation of GCN4 and the general amino acid control of yeast. Annu. Rev. Microbiol. 59, 407–450 (2005).
Blom, J., De Mattos, M. J. T. & Grivell, L. A. Redirection of the respiro-fermentative flux distribution in Saccharomyces cerevisiae by overexpression of the transcription factor Hap4p. Appl. Environ. Microbiol. 66, 1970–1973 (2000).
Dabirian, Y. et al. Expanding the dynamic range of a transcription factor-based biosensor in Saccharomyces cerevisiae. ACS Synth. Biol. 8, 1968–1975 (2019).
Kim, J.-H., Polish, J. & Johnston, M. Specificity and regulation of DNA binding by the yeast glucose transporter gene repressor Rgt1. Mol. Cell. Biol. 23, 5208–5216 (2003).
Michael, D. G. et al. Model-based transcriptome engineering promotes a fermentative transcriptional state in yeast. Proc. Natl Acad. Sci. USA 113, E7428–E7437 (2016).
Demeke, M. M., Foulquie-Moreno, M. R., Dumortier, F. & Thevelein, J. M. Rapid evolution of recombinant Saccharomyces cerevisiae for xylose fermentation through formation of extra-chromosomal circular DNA. PLoS Genet. 11, e1005010 (2015).
Westfall, P. J., Ballon, D. R. & Thorner, J. When the stress of your environment makes you go HOG wild. Science 306, 1511–1512 (2004).
Dihazi, H., Kessler, R. & Eschrich, K. High osmolarity glycerol (HOG) pathway-induced phosphorylation and activation of 6-phosphofructo-2-kinase are essential for glycerol accumulation and yeast cell proliferation under hyperosmotic stress. J. Biol. Chem. 279, 23961–23968 (2004).
Larroy, C., Pares, X. & Biosca, J. A. Characterization of a Saccharomyces cerevisiae NADP(H)‐dependent alcohol dehydrogenase (ADHVII), a member of the cinnamyl alcohol dehydrogenase family. Eur. J. Biochem. 269, 5738–5745 (2002).
Intlekofer, A. M. & Finley, L. W. Metabolic signatures of cancer cells and stem cells. Nat. Metab. 1, 177–188 (2019).
Liberti, M. V. & Locasale, J. W. The Warburg effect: how does it benefit cancer cells? Trends Biochem. Sci. 41, 211–218 (2016).
Boehlke, K. & Friesen, J. Cellular content of ribonucleic acid and protein in Saccharomyces cerevisiae as a function of exponential growth rate: calculation of the apparent peptide chain elongation rate. J. Bacteriol. 121, 429–433 (1975).
Waldron, C. & Lacroute, F. Effect of growth rate on the amounts of ribosomal and transfer ribonucleic acids in yeast. J. Bacteriol. 122, 855–865 (1975).
Dutta, A. et al. Composition and function of mutant Swi/Snf complexes. Cell Rep. 18, 2124–2134 (2017).
Vuoristo, K. S., Mars, A. E., Sanders, J. P., Eggink, G. & Weusthuis, R. A. Metabolic engineering of TCA cycle for production of chemicals. Trends Biotechnol. 34, 191–197 (2016).
Zhou, Y. J. et al. Production of fatty acid-derived oleochemicals and biofuels by synthetic yeast cell factories. Nat. Commun. 7, 11709 (2016).
Verduyn, C., Postma, E., Scheffers, W. A. & Van Dijken, J. P. Effect of benzoic acid on metabolic fluxes in yeasts: a continuous‐culture study on the regulation of respiration and alcoholic fermentation. Yeast 8, 501–517 (1992).
Mans, R. et al. CRISPR/Cas9: a molecular Swiss army knife for simultaneous introduction of multiple genetic modifications in Saccharomyces cerevisiae. FEMS Yeast Res. 15, fov004 (2015).
Liu, Q. et al. Rewiring carbon metabolism in yeast for high level production of aromatic chemicals. Nat. Commun. 10, 4976 (2019).
Yu, T. et al. Reprogramming yeast metabolism from alcoholic fermentation to lipogenesis. Cell 174, 1549–1558 (2018).
Mikkelsen, M. D. et al. Microbial production of indolylglucosinolate through engineering of a multi-gene pathway in a versatile yeast expression platform. Metab. Eng. 14, 104–111 (2012).
Zhou, Y. J. et al. Modular pathway engineering of diterpenoid synthases and the mevalonic acid pathway for miltiradiene production. J. Am. Chem. Soc. 134, 3234–3241 (2012).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Trapnell, C. et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 28, 511 (2010).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Nat. Preced. https://doi.org/10.1038/npre.2010.4282.2 (2010).
Väremo, L., Nielsen, J. & Nookaew, I. Enriching the gene set analysis of genome-wide data by incorporating directionality of gene expression and combining statistical hypotheses and methods. Nucleic Acids Res. 41, 4378–4391 (2013).
Patil, K. R. & Nielsen, J. Uncovering transcriptional regulation of metabolism by using metabolic network topology. Proc. Natl Acad. Sci. USA 102, 2685–2689 (2005).
Oliveira, A. P., Patil, K. R. & Nielsen, J. Architecture of transcriptional regulatory circuits is knitted over the topology of bio-molecular interaction networks. BMC Syst. Biol. 2, 17 (2008).
Deatherage, D. E. et al. in Engineering and Analyzing Multicellular Systems: Methods and Protocols (eds Sun, L. & Shou, W.) 165–188 (Springer, 2014).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357 (2012).
Jenjaroenpun, P. et al. Complete genomic and transcriptional landscape analysis using third-generation sequencing: a case study of Saccharomyces cerevisiae CEN.PK113-7D. Nucleic Acids Res. 46, e38 (2018).
Acknowledgements
We thank C. Zhan, Z. Dai, H. Lu, T. Doughty, K. Campbell, R. Yu and L. F.-Y. Chao for helpful discussions. We thank J. Hellgren for the help with the RNA-seq data processing and analysis. We thank X. Chen, Z. Zhu and B. Ji for giving valuable advice on writing the manuscript. We thank X. Chen and L. F.-Y. Chao for help with the final polishing of the manuscript. This research was supported by The Novo Nordisk Foundation (NNF10CC1016517, J.N.), the Knut and Alice Wallenberg Foundation (J.N.), FORMAS (2015-01546, Y.C.), the Swedish Energy Agency (43548-1, J.N.), Carl Tryggers Stiftelse (Y.C.) and Ångpanneföreningens Forskningsstiftelse (Y.C.).
Author information
Authors and Affiliations
Contributions
J.N., Y.C. and X.L. conceived the study. X.L. performed most of the experiments. X.L., Y.C. and J.N. analysed all the experimental data. G.L. analysed the partial RNA-seq data. Y.W. and Q.L. assisted with the experimental performance. R.P. and X.L. analysed the genome sequence data. X.L., Y.C. and J.N. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Catalysis thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–7, Figs. 1–10, Tables 3, 4, 7 and 8, and references.
Supplementary Table
Supplementary Tables 1, 2, 5, 6 and 9.
Supplementary Data
Source data for Supplementary Figs. 2–10.
Source data
Source Data Fig. 1
Statistical Source Data for Fig. 1.
Source Data Fig. 2
Statistical Source Data for Fig. 2.
Source Data Fig. 3
Statistical Source Data for Fig. 3.
Source Data Fig. 4
Statistical Source Data for Fig. 4.
Source Data Fig. 5
Statistical Source Data for Fig. 5.
Source Data Fig. 6
Statistical Source Data for Fig. 6.
Source Data Fig. 7
Statistical Source Data for Fig. 7.
Rights and permissions
About this article
Cite this article
Li, X., Wang, Y., Li, G. et al. Metabolic network remodelling enhances yeast’s fitness on xylose using aerobic glycolysis. Nat Catal 4, 783–796 (2021). https://doi.org/10.1038/s41929-021-00670-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-021-00670-6
This article is cited by
-
Reprogramming methanol utilization pathways to convert Saccharomyces cerevisiae to a synthetic methylotroph
Nature Catalysis (2023)
-
De novo biosynthesis of rubusoside and rebaudiosides in engineered yeasts
Nature Communications (2022)
-
Rescuing yeast from cell death enables overproduction of fatty acids from sole methanol
Nature Metabolism (2022)