Abstract
Human exploration of deep space will involve missions of substantial distance and duration. To effectively mitigate health hazards, paradigm shifts in astronaut health systems are necessary to enable Earth-independent healthcare, rather than Earth-reliant. Here we present a summary of decadal recommendations from a workshop organized by NASA on artificial intelligence, machine learning and modelling applications that offer key solutions toward these space health challenges. The workshop recommended various biomonitoring approaches, biomarker science, spacecraft/habitat hardware, intelligent software and streamlined data management tools in need of development and integration to enable humanity to thrive in deep space. Participants recommended that these components culminate in a maximally automated, autonomous and intelligent Precision Space Health system, to monitor, aggregate and assess biomedical statuses.
Similar content being viewed by others
Main
Astronauts face unique hazards from spaceflight, including ionizing radiation, altered gravitational fields, altered day–night cycles, confined isolation, hostile closed environments, distance-duration from Earth1, planetary dust-regolith2,3 and extreme temperatures and atmospheres4,5. As astronauts experience these hazards, the body responds by adapting and deconditioning, with the potential for synergistic effects as the exposures persist1,2,6. As humanity readies itself for exploration of deep space and planetary-class missions (Mars and cis-lunar), astronauts would benefit from optimally scoped biomedical systems.
From the mid-twentieth century through to the present day, humanity has conducted missions in low Earth orbit (LEO) with access to nearly real-time support from mission-control-centre (MCC) flight surgeons. However, upcoming deep-space crews will face (1) high-latency communications that prohibit real-time support7,8,9,10, (2) data bandwidth and power constraints7,11, (3) infrequent resupply7,8,12, (4) the carrying of only essential and effective medications, which may degrade over time13, (5) an inability to evacuate or be quickly rescued7,8,12 and (6) greater exposure to solar and galactic cosmic radiation11,14,15,16. To address these challenges, there is an opportunity for spaceflight biology, engineering, health and medicine to leverage the powerful emerging computer-science approaches of artificial intelligence (AI) and machine learning (ML). AI/ML uses sample data to create a representation of a system that can predict an outcome of interest on future, previously unseen data17.
The basis for this Review is content from a workshop held in June 2021 organized by NASA (National Aeronautics and Space Administration) entitled ‘Workshop on Artificial Intelligence and Modeling for Space Biology’18. Biologists, clinicians and AI/ML experts in the workshop articulated the requirements necessary to create an AI-driven, proactive ‘precision space health’ (PSH) system to ensure that the future of space health is predictive, preventative, participatory and personalized19,20. In this Review, we first introduce the basic requirements for the PSH system, focusing on the importance of multilayered monitoring in support of astronaut health and habitat. We then summarize the workshop discussions, focusing on two essential topics for a successful PSH implementation: (1) biomarkers and habitat metrics necessary to monitor health and prevent health crisis using AI/ML; (2) AI/ML methods and hardware that can be used or that need further development to integrate and enable this stream of heterogeneous data. A parallel Review from the workshop reviews AI/ML challenges and opportunities to enable fundamental space biological research21.
Overview of Precision Space Health
In 2014, the National Academy of Sciences recommended an ethical framework for long-duration and deep-space exploration with health standards continually evolving, improving and informed by data22. The workshop discussion concluded that this framework will be best enabled by principles of Precision Medicine (PM) and Precision Health (PH)23. PM refers to personalized medical treatment tailored to the individual characteristics of each patient24, including genetic predisposition, behavioural influences (exercise, nutrition, stress) and environmental influences (physical and social determinants). Leveraging PM principles, PH emphasizes disease prevention and early detection via individualized, longitudinal monitoring25,26,27.
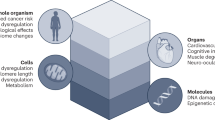
The workshop recommended a PSH system maximize AI/ML usage to integrate longitudinal, clinical, biomarker, ’omics, behavioural and microbiome data about an individual in a healthy state so as to facilitate automated and early detection of pathogenic changes (Fig. 1)28,29,30,31,32,33,34,35. It was noted that NASA and space agencies/companies are well-suited to lead in this domain, which will advance technology transfer for healthcare implementation on Earth36,37,38. In 2015–2016, the ‘Twins Study’39 monitored two genetically identical individuals on Earth and in space. It was a successful proof of concept for the ability to collect the type of rich data necessary to enable a PSH system. The experiment used several bioassays to measure effects from environmental space stressors (for example, ionizing radiation and carbon dioxide levels). There is constant monitoring of these and other stressors in the cabin/habitat during different phases of the mission (for example, pre-launch, launch and in-habitat) and these data, used as cofactors in AI/ML algorithms, can predict the onset of a medical crisis versus the temporary healthy adaptation of an individual to changes resulting from environmental stressors. One important finding in the study39 was that longitudinal data from the astronaut twin were more informative about his own health than comparisons to his twin’s synchronized data on Earth. Such a finding clearly highlights the importance of establishing a personalized baseline for each astronaut.
The AI/ML workshop generated this overall view of an ideal PSH system, with the requirement that it is a constantly learning system and has a feedback loop for improving the system (shown as bold arrows). Briefly, the research informs health and environmental monitoring, which informs appropriate countermeasures. The feedback loop shows how research and terrestrial support can be used to inform real-time health support and countermeasures, with outcome information flowing back into future monitoring strategies and biological research priorities.
The transition from LEO to deep-space missions (and cis-lunar) will present novel operational health requirements. Table 1 provides an overview of five key operational elements discussed at the workshop: a PSH must assess and aggregate and analyse multilayered data, adapt to out-of-distribution data, and provide actionable feedback. Although there have been initial onboard International Space Station (ISS) demonstrations of autonomous medical activities40, the proposed PSH system is a tremendous change from the current LEO medical planning model, which focuses on estimating the likelihood of specific medical conditions41 and provides weekly in-flight assessments in real time with MCC through audio-video streaming42, with limited or no onboard data analytics for crew-centred decision-making.
A multilayered, efficient and automated biomedical monitoring system, with predictive and clinical decision support, needs development and integration into an intelligent deep-space PSH system43,44. Redistribution of medical decisions, biological data and data management responsibilities must occur among the participants in this process (that is, the onboard Crew Health and Performance system, the crew themselves, the crew medical officer (CMO), the Environmental Control and Life Support System (ECLSS) and MCC45,46,47,48). The system must enable human crew members to assess themselves, and evaluate and act on the resulting data. It will need to identify, predict and provide health solutions to problems before they arise, as well as continuously manage and analyse the expanding accumulation of environmental, biological and health data. It should provide the CMO with explainable insights into the complexities of the biological environment, crew health and mission-specific requirements (for example, destination, duration, vehicle and habitat), as well as offer predictive outcomes depending on courses of treatment (or no treatment).
The workshop discussed repurposing and expanding terrestrial AI/ML approaches for PM/PH for spaceflight. Data collection, analysis and interpretation of health-related data and biomarkers are making substantial headway when coupled with AI/ML approaches37. In Table 2, we summarize the status of current technologies. Although in varying stages of maturity and requiring additional investment in development on Earth, many show relevance for detecting and mitigating spaceflight risks. These technologies need parallel development and integration toward a common goal so they can collectively support space exploration. We discuss these key technologies and science in the following sections.
Biomarkers and health-status assessment
A PSH system will require robust sets of biomarkers for early disease detection and prediction26,27,49,50,51. Of note, we need to identify markers of dysfunction and also of countermeasure success. However, clinically actionable biomarkers are still being discovered and implemented terrestrially52,53. Space agencies should invest in research to identify biomarkers to predict specific disorders. These biomarkers must also be safe, reliable and reproducible so as to assess biomedical function and assess environmental impacts on health54, and must be distinguishable between those that are causal versus indicative of a disease55. AI/ML can be leveraged in two ways. First, it can aid the optimization of prediction capabilities, facilitating the acceptance of monitoring thresholds on a personalized basis56. Second, AI/ML can learn from a constant stream of data, which opens the door to identifying new biomarkers with more predictive power than established ones. Another important aspect of biomarker discovery is the usage of model organism reference experiments and missions. Small animals provide access to many more data points, which is essential for AI/ML. Tissue-on-a-chip research provides an alternative to animal experiments, and remains simpler in its biological understanding compared to a full organism57,58,59. In the following we summarize the several data types the workshop identified as having the most potential for spaceflight health biomarkers.
Predictions using multi-omic, paired and phenotypic data
Considering the known altered immune function effects from spaceflight, the immune system is a key biological system to monitor and evaluate. Single-cell immune monitoring approaches such as single-cell RNA sequencing60,61 and mass cytometry62 are currently used to monitor populations in terrestrial clinical settings where the immune system is modulated63,64. These transcriptomics and proteomics data are challenging due to the high dimensionality (for example, there are over 20,000 genes measured in one transcriptomic sample) and low sample number. Dimensionality reduction will be key for combining and learning from these datasets. In contrast, physiological data such as levels of circulating cytokines have a much lower number of dimensions. The first step is to map a baseline immune configuration of individual astronauts. AI/ML models can then use departure-from-baseline to detect the most likely causal genes or proteins enabling physiological changes from spaceflight. Future approaches will most likely integrate comprehensive immune measurements65, combining omics, physiological, microbial66,67 and other relevant data such as microRNAs68, exosomes, cell-free DNA69, clonal haematopoiesis70 and DNA damage biomarkers71. Pairing of biomarkers based on behavioural phenotypes (speech patterns, semantic/sentiment breakdown, facial expressions) with multi-omics data can create useful omic-phenotypic connections72.
Predictions using longitudinal, individualized and baseline data
For biomarker identification and real-time space health monitoring, longitudinal measurements will be essential to detect individualized health changes. For example, continuous monitoring of temperature with wearables has shown that fever thresholds change between individuals, or with age, gender or ethnicity73. Similarly, systems-level analyses have shown variations in immune setpoints63 and microbiome composition74. Previous efforts have successfully demonstrated individualized monitoring of changes to self-baselines using blood31, digital devices32 and non-invasive saliva sampling34, enabling personalized coaching of individuals75 and microbiomes33. Such approaches can first establish baselines of various biomarkers for each astronaut individually on Earth. AI/ML methods for time-series analysis can identify potentially adverse medical events by monitoring deviations from a healthy baseline using longitudinal data31,32,33,34,75. When digital twin technology is mature, having an AI-enabled digital twin of each astronaut may eventually be a better health predictor76 than what we can achieve with real biological twins. When and how to label a biological response as unhealthy is important. For example, when exposed to ionizing radiation, an expected healthy response is the induction of DNA damage. However, when DNA damage is persistent or there is poor induction of DNA damage, there is a long-term radiation toxicity associated71,77,78. Characterizing the genetics of each astronaut by whole-genome sequencing will be useful to better interpret longitudinal data.
Non-invasive health-status assessment with AI/ML
The workshop discussed ways in which health-status assessment systems can incorporate AI/ML in a non-invasive manner. For example, AI/ML voice analysis can monitor stress or fatigue, with privacy considerations mitigated by avoiding semantic analysis of dialogue79. AI/ML can analyse sleep and locomotion activity, and can assess how inferred health status is affected. To mitigate stress, AI-generated personalized and private therapy programmes can be included in crew health resources80,81. A further example is the pairing of pre-flight structural ‘biomarker’ (that is, anatomical) analysis for monitoring vascular and tissue ocular/vision changes82,83 with assessments of adverse headward fluid shifts occurring in microgravity. Such spaceflight-based results linked with other physiological monitoring of blood, nutritional, immunological and performance measures would require AI/ML analysis and equipment miniaturization37,84,85. Assessments can also motivate the importance of acquiring more space data, rather than relying on terrestrial analogues that may not replicate key features of human responses86.
Space missions should expect the emergence of novel phenotypic manifestations and situations in deep space1,6. Reinforcement learning and n-of-1 studies49,50,87,88 may help provide statistical power to derive multi-targeted treatment, behavioural interventions, or activity interventions (specific to individuals) during a mission in order to address novel phenotypes.
Monitoring the spectrum of layered and integrated flight data
The workshop recommended an essential component of the PSH system be a multilayered monitoring-collection system, including both spacecraft/habitat/ecological environments and individual astronauts (Fig. 2). Its recommendations span from both contact and non-contact devices to monitor the health of individual astronauts, to novel semi-automated assays to monitor the entire spacecraft and all habitable environments (plants, crops, animals, microbes, physicochemical environment and so on). When realized, this holistic multilayered PSH monitoring system would provide, for the first time, a continuous picture of the health of the entire spacecraft/habitat, and the entire living ecosystem inside.
The initial layer of monitoring would be the continuous environmental sensing of physical (for example, vibrations, temperature, airflow, sounds and radiation), chemical (for example, carbon dioxide, dust particles, volatile organic and inorganic compounds) and biological components (for example, general microbiota and specific species with known risks). Many of these sensors are already standard instruments for LEO missions, but the data are not yet integrated into one database, nor analysable in situ. The NASA Open Science Data Repository (which includes both GeneLab and the Ames Life Sciences Data Archive) houses molecular ’omics and phenotypic data from spaceflight life sciences experiments, and has begun remediation of this issue89,90,91,92. Integrating sensor data will enable AI/ML models while keeping human in-the-loop inputs46. For example, radiation instruments currently in use on the ISS and planned for lunar missions gather real-time data on absorbed radiation dose and dose rates93,94,95. However, extrapolation from the absorbed dose to specific biological effects requires more detailed knowledge of the components of the space radiation field (for example, protons versus high-energy particles and neutrons). Also, of crucial importance for missions beyond Earth’s radioprotective Van Allen Belt, molecular and sophisticated dosimetry will be essential for the high-resolution detection of both DNA damage71 and gauging pharmaceutical stability96,97. Additional post-processing can achieve this on the ground with various types of active dosimeter93,98.
A second layer is traditional non-invasive physiological metrics, collected by ‘wearables’, point-of-care devices (for example, ultrasound, blood pressure, breath-analysis, ocular/visual and respiration), videos showing behavioural health, and self-administered tests, such as cognitive tests, exercise routines and sleep data54. Platforms should be minimally intrusive, and data should be easily decipherable by the crew and CMO. An example of non-intrusive monitoring shown recently used active sonar (speaker-microphone) to monitor heart rate and heart rhythms remotely99.
A third layer would be based on molecular physiological biomarkers and/or truly ‘invasive’ measures obtained from various swabs, blood draws, saliva sampling and other molecular assays. A ‘smart toilet’ (as well as a smart shower booth and a smart mirror) could preserve and prepare waste specimens for biochemistry assays and microbiome profiling25,100,101. Such platforms hold promise in expanding to include in situ and real-time analytical capabilities. Similarly, non-invasive high-frequency monitoring of molecular components from saliva over time can also provide immune signatures that may monitor deviations from a healthy immune baseline, using anomaly detection algorithms to assess change points as potential adverse medical events34.
Adaptations in computing, model-training and data communications in deep space
To enable real-time monitoring, in situ operations will play an increasingly important role in enabling immediate responses (Fig. 3). In addition, as the volume of health-related data grows until it is no longer viable to downlink datasets, AI/ML applications will offer real-time health capabilities, requiring full in situ computation for maximal autonomy. In this section we will dive deeper into the various aspects of this paradigm shift.
The life cycle of AI/ML in space biology research and spaceflight health enables full utilization of Open and FAIR space biological datasets through a data management environment. AI/ML methods leveraging neuromorphic processors and edge computing facilitate a layered and integrated spectrum of in-flight data acquisition to power a PSH system for deep-space missions.
Translational science and the AI/ML biomedical life cycle
Translational knowledge and data from both fundamental and applied space biomedical research are part of a crucial translational pipeline to inform a PSH system. Such research builds a wealth of evidence and statistical power upon which AI/ML biomedical predictions rely. The life cycle of AI/ML and the cross-cutting relationship between space biological research and a PSH system are shown in Fig. 3.
Current limitations for data communications and computing
There are several limitations to deploying computer, data-storage and communication systems on board any spacecraft102. These factors include volume, weight and power constraints, resilience to launch vibrations, chemically reactive planetary dusts and ionizing radiation103, and reliability in autonomous maintenance and repair. The pioneering development of the Spaceborne Computer-1 and -2104 aboard the ISS has made inroads towards addressing these challenges. Specialized hardware could reduce the footprint of computer needs while delivering very high AI/ML performance. For example, onboard graphics processing units would accelerate AI/ML capabilities for DNA sequencing requiring real-time base calling for detecting modified nucleic acids105. The workshop recommended more investment in the development of hardware enabling AI/ML. The following are strategies the workshop identified to circumvent the unique constraints of spaceflight: (1) developing software that relies less on accessing Earth databases, (2) using ML-based data compression106 and (3) deploying active learning systems based on pre-trained models on large Earth databases but constantly learning from the growing stream of data collected on board the spacecraft/habitat107. As missions move further from Earth, access to computing power will need to transition from terrestrial-based to spacecraft and habitat-based. Many challenges of building space-ready AI/ML are difficult to foresee, and there will be more challenges for Mars missions108.
Modelling with deep-space biomedical data
Most current terrestrial AI/ML techniques use large numbers of observations (and usually a large feature set) to train a model. The application of these methods to human health in spaceflight is challenging for several reasons. First, biomedical data collected and reusable from astronauts in-flight are historically and currently limited. Over 600 people worldwide have gone to space, and only 24 beyond LEO. Missions have had an average duration of fewer than 30 days, making it almost impossible to train sophisticated AI/ML models using only spaceflight human data. Second, the data collected during spaceflight have been narrow and inconsistent compared to what is typically available in clinical or research settings on Earth. Until recently, there was no standardized set of biomedical measures taken during NASA human spaceflight explicitly for research purposes. Finally, NASA’s mission of exploration means that the models needed for a PSH system must be extrapolated beyond the context in which we have spaceflight experience. Even though the laws of physics do not change over the course of a deep-space mission, the human body does, and potentially in nonlinear ways, thus lowering the accuracy of the ‘approximation’ training data. A key consideration for AI/ML medical applications is whether the system needs to be trained in situ using locally collected data, or whether the models can be trained using ground data prior to the mission, during the mission with an uplink of the updated model, or gradually developed into active learning systems107 to add autonomy degrees. These distinctions are important, because training a model is typically computationally intensive and requires large amounts of data, whereas performing inference with a trained model is far less demanding.
To address these considerations and limitations, Table 3 summarizes the workshop recommendations for methods to address the key requirements of an AI-enabled PSH system, and gives examples of applications. Neuromorphic processors are one exciting emerging technology to provide in situ computing that is well suited for performing AI/ML tasks in a spaceflight context, specifically, because of their radiation resilience, low power requirements and high efficacy for deep learning tasks. Indeed, NASA has recognized the potential of neuromorphic computing for these reasons109. Edge computing is a resilient approach that creates a redundant network to distribute computing needs based on sophisticated job management that prioritizes onboard activities. Finally, computing hardware, model software, and data management and communication strategies for PSH need to be maximally adaptive to accommodate constant reassessment of newly acquired data. Active learning algorithms expedite this approach, where humans can help AI/ML algorithms learn faster by injecting new data types and new labels into the system.
Federated learning was developed to deal with limited abilities for data transfer, where multiple model components are trained at the location of the data, and then merged into a single federated model. This would deal with large datasets generated in space, or NASA astronaut health data containing considerable privacy constraints. Regardless of how small and efficient computing resources and algorithms become, AI/ML in space works under limited conditions compared to that on Earth. Transfer learning is also a highly effective way to reduce spaceborne computing and data demands. Using this technique, AI/ML models are first trained on Earth using analogue datasets (for example, animal data or synthetic data). Subsequently, in space, the final computation to ‘transfer’ the model to the actual context of interest is performed, such as a classification model using astronaut health data. This reduces the computing and data requirements needed to achieve a model with acceptable efficacy. Dimensionality reduction is a central way to reduce the amount of data to consider in the prediction model, but there is more work to be done at the research level to be smarter about what features drive a response and thus only consider these factors.
Finally, probably the biggest challenge in a PSH system is the fact that such a system will operate using out-of-distribution data. Any training done with Earth analogues will only partially resemble what is truly happening in space. Methods that are more resilient to operating under different assumptions will be essential. The workshop listed several such methods, summarized into three categories: translation from animals to humans110,111; generalization across domains using approaches such as risk extrapolation or domain invariant methods55; and adapting models trained on Earth health data to perform well on space health data where the baseline normal is persistently different.
Further challenges for deep-space health
Integrated biomedical flight data acquisition, AI/ML modelling tools and techniques, as well as a PSH system will be crucial pillars in bridging the gap between the current LEO operational paradigm and that which is needed for successful cis-lunar and planetary-class missions. Discussed below are several other space biomedical challenges involving AI/ML covered by the workshop.
Crew confidence and fidelity of AI/ML
Through the lens of clinical significance, data are used to inform clinical decisions about preventive and acute care. It is important that we shift the role of diagnostic AI/ML from simply predicting labels, to interpreting context and providing iterative cues that guide diagnosticians112. The crew, flight surgeons and all related staff must wholeheartedly establish training and build confidence in an AI-based PSH system. Tools and techniques for AI/ML and modelling (or any type of data system) additionally ought to include comprehensive and potentially continuous assessment of its credibility, ethics and trustworthiness. This includes methods that address reference data or model prediction benchmarks. Fidelity and ethics assessments of AI modelling extend past specific technique validation. They encompass broader aspects of credibility, including a full provenance of life-cycle development and evaluation of the systemic assumptions, biases and deployment limitations (that is, toward predictions or knowledge-gained)113,114.
Ethics, genomics and de-identification
The spaceflight community has a responsibility to meet ethical obligations involved in a PSH system and related data privacy28,115. Whole-genome sequencing will probably be a pre-flight component required to enable an effective AI-driven PSH system. Handling privacy and the wishes of all spacefarers (NASA astronauts, but also international and private-commercial space travellers) requires sincere care, clear governance, and policy. De-identified data systems with decoupled federated learning systemic firewalls are one approach to ensure data are explicitly not traceable116. Broad data analysis must occur to support deep-space health, with completely untraceable data to protect any impacts to multi-generational offspring regarding their privacy and quality of life. The development of modified Genetic Information Nondiscrimination Act (GINA) guidelines28,117 and waivers for utilization of spaceflight genomics data should protect astronauts and their relatives.
Make data AI/ML-ready
Metadata and data curation-processing standards for PSH and the field of space biology need to be determined, ensuring data are ‘AI-ready’ and modelling accessible. A space ‘data readiness level’ metric can be implemented as a tool to encourage reliable data quality118. Basic synthetic datasets and model libraries for space health and biology also need development. Adaptations and innovations in statistics, algorithms, data and medical informatics are almost certainly going to be required and modified for spaceflight health and biology21,119.
Interdisciplinary teams
Deep-space missions will have one-of-a-kind space biology and health requirements. The interdisciplinary breadth of the teams required to develop, collaborate, refine and implement these systems is novel. Communities of programmers, biologists, data architects, human factors, operational clinicians and hardware engineers need to be interwoven (non-siloed), so as to implement a PSH and biomonitoring/research system120. Research is likely to flow more freely between researchers and clinical operations during deep-space missions, which could enable novel discovery outcomes. For example, a recent research team utilizing an ultrasound device was interested in blood-flow behaviour in the internal jugular vein in long-term ISS mission crew. The team noticed anomalies in venous blood flow, which led to a clinical team handover, who then characterized (and treated for) flow stasis and venous thrombosis121,122.
Recommendations and conclusion
The workshop brainstormed a decadal vision of what should be developed to leverage AI/ML for both spaceflight health support and space biological research. Various AI/ML technological needs were identified, and the workshop brought together (for the first time) an array of interdisciplinary AI/ML experts with space-oriented biologists and clinicians. Workshop participants agreed that it is difficult to predict the needed space capabilities as there is no platform today to serve as a developmental testbed for verification or validation of real-time, actionable, AI/ML model testing. During the workshop, time was spent understanding NASA’s data systems and technologies as they stand today, and the fundamental data management developments and designs needed to begin implementing AI/ML. Although no detailed roadmap, nor strategic framework, was produced, the general workshop goal of producing a decadal vision was achieved, through the identification of several broad focus areas over the next decade. Based on current capabilities, and with applications of AI/ML still in their infancy terrestrially in relation to clinical health, workshop participants arrived at a number of reasonable avenues to begin enabling this field:
-
Develop hardware and data systems to enable efficient data collection for in situ analytics, with an emphasis on baseline and longitudinal data
-
Develop stronger data management, harmonization, AI-readiness, and a data-readiness-level metric to allow in-mission and terrestrial-analogue research
-
Invest in an AI/ML/modelling ‘Zoo’, repurpose current AI/ML approaches for space-specific needs and research into novel algorithms, software, hardware and models
-
Transition crew health from treatment to prevention, which is essential for deep space, and requires a holistic, multilayered, biomonitoring approach of the entire spacecraft/habitat ecosystem, including multi-modal, multi-hierarchical data and research into targeted biomarkers
-
Integrate hardware, data systems, software/AI/ML and biomonitoring to enable the development of a PSH system for real-time, adaptable, maximally autonomous decision-making and analysis, in conjunction with the CMO and crew participation.
The workshop generated several far-reaching questions that cannot yet be fully answered and require focus in the next decade of research and development. How much degradation of crew health and capability should we expect? How much decision support and data analytics capability should be built into a mission to maximize the chances of success? How much long-term health risk will astronauts face post-career? These questions can only be answered by scientific research, and this is reliant on strong data systems in future missions and experiments.
The development of the AI/ML modelling systems discussed in this Review will be multi-year, interdisciplinary and involve far-reaching collaborations. As humanity explores beyond LEO, it will leave the confines of an immediately accessible, large and continuously supportive cohort of mission-control health and science staff, with systems that have been developed over the past ~60 years. Today’s exciting new spacecraft, with expansive payload abilities, greatly enable the field of space health and biology to develop and deploy AI/ML/edge technologies, toward in situ analytical capabilities. The relatively recent emergence of terrestrial AI, ML and modelling brings a key contribution towards making humanity multi-planetary through spacecraft and habitat biomedical science support and a PSH system.
References
Afshinnekoo, E. et al. Fundamental biological features of spaceflight: advancing the field to enable deep-space exploration. Cell 183, 1162–1184 (2020).
Loftus, D. J., Rask, J. C., McCrossin, C. G. & Tranfield, E. M. The chemical reactivity of lunar dust: from toxicity to astrobiology. Earth Moon Planets 107, 95–105 (2010).
Pohlen, M., Carroll, D., Prisk, G. K. & Sawyer, A. J. Overview of lunar dust toxicity risk. NPJ Microgravity 8, 55 (2022).
Paul, A.-L. & Ferl, R. J. The biology of low atmospheric pressure—implications for exploration mission design and advanced life support. Gravit. Space Res. 19, 3–17 (2005).
Council, N. R. Recapturing a Future for Space Exploration: Life and Physical Sciences Research for a New Era (National Academies Press, 2011).
Goswami, N. et al. Maximizing information from space data resources: a case for expanding integration across research disciplines. Eur. J. Appl. Physiol. 113, 1645–1654 (2013).
McGuire, K. et al. Using systems engineering to develop an integrated crew health and performance system to mitigate risk for human exploration missions. In Proc. 50th International Conference on Environmental Systems, 298, 1–11 (2021).
Antonsen, E., Hanson, A., Shah, R., Reed, R. D. & Canga, M. A. Conceptual drivers for an exploration medical system. In Proc. 67th International Astronautical Congress 1–10 (NASA Technical Reports Server, 2016).
Zhao, K. & Zhang, Q. Network protocol architectures for future deep-space internetworking. Sci. China Inf. Sci. 61, 040303 (2018).
Beaton, K. H. et al. Extravehicular activity operations concepts under communication latency and bandwidth constraints. In Proc. 2017 IEEE Aerospace Conference 1–20 (IEEE, 2017)
Ball, J. R. & Evans, C. H. Jr. Safe Passage: Astronaut Care for Exploration Missions (National Academies Press, 2014).
Antonsen, E. L. et al. Estimating medical risk in human spaceflight. NPJ Microgravity 8, 8 (2022).
McNulty, M. J. et al. Evaluating the cost of pharmaceutical purification for a long-duration space exploration medical foundry. Front. Microbiol. 12, 700863 (2021).
Blue, R. S. et al. Challenges in clinical management of radiation-induced illnesses during exploration spaceflight. Aerosp. Med. Hum. Perform. 90, 966–977 (2019).
Chancellor, J. C. et al. Limitations in predicting the space radiation health risk for exploration astronauts. NPJ Microgravity 4, 8 (2018).
Patel, Z. S. et al. Red risks for a journey to the red planet: the highest priority human health risks for a mission to Mars. NPJ Microgravity 6, 33 (2020).
Jordan, M. I. & Mitchell, T. M. Machine learning: trends, perspectives and prospects. Science 349, 255–260 (2015).
Costes, S. V., Sanders, L. M. & Scott, R. T. Workshop on Artificial Intelligence & Modeling for Space Biology https://zenodo.org/record/7508535#.Y9LwQITP23A (2023).
Hood, L. & Flores, M. A personal view on systems medicine and the emergence of proactive P4 medicine: predictive, preventive, personalized and participatory. N. Biotechnol. 29, 613–624 (2012).
Zitnik, M. et al. Machine learning for integrating data in biology and medicine: principles, practice and opportunities. Inf. Fusion 50, 71–91 (2019).
Sanders, L. M. et al. Biological research and self-driving labs in deep space supported by artificial intelligence. Nat. Mach. Intell. https://doi.org/10.1038/s42256-023-00618-4 (2023).
Kahn, J., Liverman, C. T. & McCoy, M. A. Health Standards for Long Duration and Exploration Spaceflight: Ethics Principles, Responsibilities and Decision Framework (National Academies Press, 2014).
Schmidt, M. A., Schmidt, C. M., Hubbard, R. M. & Mason, C. E. Why personalized medicine is the frontier of medicine and performance for humans in space. New Space 8, 63–76 (2020).
National Research Council (US) Committee on A Framework for Developing a New Taxonomy of Disease. Toward Precision Medicine: Building a Knowledge Network for Biomedical Research and a New Taxonomy of Disease (National Academies Press, 2012).
Park, S.-M., Ge, T. J., Won, D. D., Lee, J. K. & Liao, J. C. Digital biomarkers in human excreta. Nat. Rev. Gastroenterol. Hepatol. 18, 521–522 (2021).
Gambhir, S. S., Ge, T. J., Vermesh, O. & Spitler, R. Toward achieving precision health. Sci. Transl. Med. 10, eaao3612 (2018).
Gambhir, S. S., Ge, T. J., Vermesh, O., Spitler, R. & Gold, G. E. Continuous health monitoring: an opportunity for precision health. Sci. Transl. Med. 13, eabe5383 (2021).
Antonsen, E. L. & Reed, R. D. Policy considerations for precision medicine in human spaceflight. Hous. J. Health L. Policy 19, 1–37 (2020).
Schork, N. J. Personalized medicine: time for one-person trials. Nature 520, 609–611 (2015).
Arges, K. et al. The Project Baseline Health Study: a step towards a broader mission to map human health. NPJ Digit. Med. 3, 84 (2020).
Chen, R. et al. Personal omics profiling reveals dynamic molecular and medical phenotypes. Cell 148, 1293–1307 (2012).
Li, X. et al. Digital health: tracking physiomes and activity using wearable biosensors reveals useful health-related information. PLoS Biol. 15, e2001402 (2017).
Zhou, W. et al. Longitudinal multi-omics of host-microbe dynamics in prediabetes. Nature 569, 663–671 (2019).
Mias, G. I. et al. Longitudinal saliva omics responses to immune perturbation: a case study. Sci. Rep. 11, 710 (2021).
Haney, N. M., Urman, A., Waseem, T., Cagle, Y. & Morey, J. M. AI’s role in deep space. J. Med. Artif. Intell. 3, 11 (2020).
Yu, K.-H., Beam, A. L. & Kohane, I. S. Artificial intelligence in healthcare. Nat. Biomed. Eng. 2, 719–731 (2018).
Topol, E. J. Deep Medicine: How Artificial Intelligence Can Make Healthcare Human Again (Basic Books, 2019).
Topol, E. J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 25, 44–56 (2019).
Garrett-Bakelman, F. E. et al. The NASA Twins Study: a multidimensional analysis of a year-long human spaceflight. Science 364, eaau8650 (2019).
Thompson, D. E. Space Technology—Game Changing Development NASA Facts: Autonomous Medical Operations. NASA Technology Reports Server (NASA, 2018).
Walton, M. E. & Kerstman, E. L. Quantification of medical risk on the International Space Station using the Integrated Medical Model. Aerosp. Med. Hum. Perform. 91, 332–342 (2020).
Sipes, W., Holland, A. & Beven, G. in Handbook of Bioastronautics (eds Young, L. R. & Sutton, J. P.) 425–436 (Springer, 2021).
McGregor, C. A platform for real-time space health analytics as a service utilizing space data relays. In Proc. 2021 IEEE Aerospace Conference (50100) 1–14 (IEEE, 2021).
McGregor, C. A platform for real-time online health analytics during spaceflight. In Proc. 2013 IEEE Aerospace Conference 1–8 (IEEE, 2013).
Mindock, J. et al. Systems engineering for space exploration medical capabilities. In Proc. AIAA SPACE and Astronautics Forum and Exposition 139, 306–312 (American Institute of Aeronautics and Astronautics, 2017).
Schneider, W. F. et al. NASA environmental control and life support technology development and maturation for exploration: 2019 to 2020 overview. In Proc. International Conference on Environmental Systems 200, 1–12 (2021).
Broyan, J. L., Shaw, L., Mc Kinley, M., Meyer, C. & Ewert, M. K. NASA environmental control and life support technology development for exploration: 2020 to 2021 overview. In Proc. 50th International Conference on Environmental Systems 384, 1–12 (NASA, 2021).
Williams-Byrd, J. A. et al. Implementing NASA’s capability-driven approach: insight into NASA’s processes for maturing exploration systems. In AIAA SPACE 2015 Conference and Exposition (American Institute of Aeronautics and Astronautics, 2015).
Goel, N. & Dinges, D. F. Predicting risk in space: genetic markers for differential vulnerability to sleep restriction. Acta Astronaut. 77, 207–213 (2012).
Limkakeng, A. T. Jr. et al. Systematic molecular phenotyping: a path toward precision emergency medicine? Acad. Emerg. Med. 23, 1097–1106 (2016).
Clément, G. R. et al. Challenges to the central nervous system during human spaceflight missions to Mars. J. Neurophysiol. 123, 2037–2063 (2020).
Fitzgerald, J. et al. Future of biomarker evaluation in the realm of artificial intelligence algorithms: application in improved therapeutic stratification of patients with breast and prostate cancer. J. Clin. Pathol. 74, 429–434 (2021).
Weiss, J., Hoffmann, U. & Aerts, H. J. W. L. Artificial intelligence-derived imaging biomarkers to improve population health. Lancet Digit. Health 2, e154–e155 (2020).
Strangman, G. E. et al. Deep-space applications for point-of-care technologies. Curr. Opin. Biomed. Eng. 11, 45–50 (2019).
Budd, S. et al. Prototyping CRISP: a Causal Relation and Inference Search Platform applied to colorectal cancer data. In Proc. IEEE 3rd Global Conference on Life Sciences and Technologies (LifeTech) 517–521 (IEEE, 2021).
Schmidt, M. A. & Goodwin, T. J. Personalized medicine in human space flight: using Omics based analyses to develop individualized countermeasures that enhance astronaut safety and performance. Metabolomics 9, 1134–1156 (2013).
Low, L. A., Mummery, C., Berridge, B. R., Austin, C. P. & Tagle, D. A. Organs-on-chips: into the next decade. Nat. Rev. Drug Discov. 20, 345–361 (2021).
Tissue Chips in Space https://ncats.nih.gov/tissuechip/projects/space (NIH, 2016).
Yeung, C. K. et al. Tissue chips in space—challenges and opportunities. Clin. Transl. Sci. 13, 8–10 (2020).
Papalexi, E. & Satija, R. Single-cell RNA sequencing to explore immune cell heterogeneity. Nat. Rev. Immunol. 18, 35–45 (2018).
Gertz, M. L. et al. Multi-omic, single-cell, and biochemical profiles of astronauts guide pharmacological strategies for returning to gravity. Cell Rep. 33, 108429 (2020).
Spitzer, M. H. & Nolan, G. P. Mass cytometry: single cells, many features. Cell 165, 780–791 (2016).
Lakshmikanth, T. et al. Human immune system variation during 1 year. Cell Rep. 32, 107923 (2020).
Hartmann, F. J. et al. Comprehensive immune monitoring of clinical trials to advance human immunotherapy. Cell Rep. 28, 819–831 (2019).
Emerson, R. O. et al. Immunosequencing identifies signatures of cytomegalovirus exposure history and HLA-mediated effects on the T cell repertoire. Nat. Genet. 49, 659–665 (2017).
Tesei, D., Jewczynko, A., Lynch, A. M. & Urbaniak, C. Understanding the complexities and changes of the astronaut microbiome for successful long-duration space missions. Life 12, 495 (2022).
Cervantes, J. L. & Hong, B.-Y. Dysbiosis and immune dysregulation in outer space. Int. Rev. Immunol. 35, 67–82 (2016).
Malkani, S. et al. Circulating miRNA spaceflight signature reveals targets for countermeasure development. Cell Rep. 33, 108448 (2020).
Bezdan, D. et al. Cell-free DNA (cfDNA) and exosome profiling from a year-long human spaceflight reveals circulating biomarkers. iScience 23, 101844 (2020).
Mencia-Trinchant, N. et al. Clonal hematopoiesis before, during and after human spaceflight. Cell Rep. 33, 108458 (2020).
Pariset, E. et al. DNA damage baseline predicts resilience to space radiation and radiotherapy. Cell Rep. 33, 108434 (2020).
Bruce-Keller, A. J., Salbaum, J. M. & Berthoud, H.-R. Harnessing gut microbes for mental health: getting from here to there. Biol. Psychiatry 83, 214–223 (2018).
Obermeyer, Z., Samra, J. K. & Mullainathan, S. Individual differences in normal body temperature: longitudinal big data analysis of patient records. Brit. Med. J. 359, j5468 (2017).
Manor, O. et al. Health and disease markers correlate with gut microbiome composition across thousands of people. Nat. Commun. 11, 5206 (2020).
Price, N. D. et al. A wellness study of 108 individuals using personal, dense, dynamic data clouds. Nat. Biotechnol. 35, 747–756 (2017).
Masison, J. et al. A modular computational framework for medical digital twins. Proc. Natl Acad. Sci. USA 118, e2024287118 (2021).
Penninckx, S. et al. Dose, LET and strain dependence of radiation-induced 53BP1 foci in 15 mouse strains ex vivo introducing novel DNA damage metrics. Radiat. Res. 192, 1–12 (2019).
Pariset, E. et al. 53BP1 repair kinetics for prediction of in vivo radiation susceptibility in 15 mouse strains. Radiat. Res. 194, 485–499 (2020).
Or, F., Torous, J. & Onnela, J.-P. High potential but limited evidence: using voice data from smartphones to monitor and diagnose mood disorders. Psychiatr. Rehabil. J. 40, 320–324 (2017).
Gratzer, D. & Goldbloom, D. Therapy and E-therapy-preparing future psychiatrists in the era of apps and chatbots. Acad. Psychiatry 44, 231–234 (2020).
Gaffney, H., Mansell, W. & Tai, S. Conversational agents in the treatment of mental health problems: mixed-method systematic review. JMIR Ment. Health 6, e14166 (2019).
Gulshan, V. et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA 316, 2402–2410 (2016).
Lee, A. G. et al. Spaceflight associated neuro-ocular syndrome (SANS) and the neuro-ophthalmologic effects of microgravity: a review and an update. NPJ Microgravity 6, 7 (2020).
De Fauw, J. et al. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat. Med. 24, 1342–1350 (2018).
Poplin, R. et al. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nat. Biomed. Eng. 2, 158–164 (2018).
Taibbi, G. et al. Opposite response of blood vessels in the retina to 6° head-down tilt and long-duration microgravity. NPJ Microgravity 7, 38 (2021).
Wang, Y. & Schork, N. J. Power and design issues in crossover-based N-of-1 clinical trials with fixed data collection periods. Healthcare (Basel) 7, 84 (2019).
Schork, N. J. & Goetz, L. H. Single-subject studies in translational nutrition research. Annu. Rev. Nutr. 37, 395–422 (2017).
Ray, S. et al. GeneLab: omics database for spaceflight experiments. Bioinformatics 35, 1753–1759 (2019).
Berrios, D. C., Galazka, J., Grigorev, K., Gebre, S. & Costes, S. V. NASA GeneLab: interfaces for the exploration of space omics data. Nucleic Acids Res. 49, D1515–D1522 (2021).
Scott, R. T. et al. Advancing the integration of biosciences data sharing to further enable space exploration. Cell Rep. 33, 108441 (2020).
Scott, R. T. et al. Open science for the next decade of life and physical sciences research for deep space exploration. A White Paper Submitted on 23 December 2021 to the Committee on Biological and Physical Sciences in Space for the 2023–2032 Decadal Survey (National Academies Press, 2021).
Schimmerling, W. Space Radiation Dosimetry https://three.jsc.nasa.gov/articles/dosimetryposted3.pdf (NASA, 2009).
Yasuda, H. Effective dose measured with a life size human phantom in a low Earth orbit mission. J. Radiat. Res. 50, 89–96 (2009).
Kroupa, M. et al. A semiconductor radiation imaging pixel detector for space radiation dosimetry. Life Sci. Space Res. 6, 69–78 (2015).
Lassmann, M. & Eberlein, U. The relevance of dosimetry in precision medicine. J. Nucl. Med. 59, 1494–1499 (2018).
Blue, R. S. et al. Limitations in predicting radiation-induced pharmaceutical instability during long-duration spaceflight. NPJ Microgravity 5, 15 (2019).
Horneck, G. Biological monitoring of radiation exposure. Adv. Space Res. 22, 1631–1641 (1998).
Wang, A., Nguyen, D., Sridhar, A. R. & Gollakota, S. Using smart speakers to contactlessly monitor heart rhythms. Commun. Biol. 4, 319 (2021).
Park, S.-M. et al. A mountable toilet system for personalized health monitoring via the analysis of excreta. Nat. Biomed. Eng. 4, 624–635 (2020).
Ge, T. J., Chan, C. T., Lee, B. J., Liao, J. C. & Park, S.-M. Smart toilets for monitoring COVID-19 surges: passive diagnostics and public health. NPJ Digit. Med. 5, 39 (2022).
Hook, J. V. et al. Nebulae: a proposed concept of operation for deep space computing clouds. In Proc. 2020 IEEE Aerospace Conference 1–14 (IEEE, 2020).
Keys, A., Adams, J., Cressler, J., Johnson, M. & Patrick, M. A review of NASA’s radiation-hardened electronics for space environments project. In Proc. AIAA SPACE 2008 Conference & Exposition 1–7 (American Institute of Aeronautics and Astronautics, 2008).
Fernandez, M. HPC in space: an update on spaceborne computer after 1+ year on the ISS. In Proc. The International Conference for High Performance Computing, Networking, Storage, and Analysis (2018).
McIntyre, A. B. R. et al. Single-molecule sequencing detection of N6-methyladenine in microbial reference materials. Nat. Commun. 10, 579 (2019).
Azar, J., Makhoul, A., Barhamgi, M. & Couturier, R. An energy efficient IoT data compression approach for edge machine learning. Future Gener. Comput. Syst. 96, 168–175 (2019).
Monarch, R. (Munro). Human-in-the-Loop Machine Learning: Active Learning and Annotation for Human-centered AI (Manning, 2021).
Nangle, S. N. et al. The case for biotech on Mars. Nat. Biotechnol. 38, 401–407 (2020).
SBIR. Deep Neural Net and Neuromorphic Processors for In-Space Autonomy and Cognition (NASA Small Business Innovation Research, 2020).
Nelson, C. A. et al. Knowledge network embedding of transcriptomic data from spaceflown mice uncovers signs and symptoms associated with terrestrial diseases. Life 11, 42 (2021).
O’Donoghue, O. et al. Invariant risk minimisation for cross-organism inference: substituting mouse data for human data in human risk factor discovery. Preprint at https://arxiv.org/abs/2111.07348 (2021).
Adler-Milstein, J., Chen, J. H. & Dhaliwal, G. Next-generation artificial intelligence for diagnosis: from predicting diagnostic labels to ‘wayfinding’. JAMA 326, 2467–2468 (2021).
Erdemir, A. et al. Credible practice of modeling and simulation in healthcare: ten rules from a multidisciplinary perspective. J. Transl. Med. 18, 369 (2020).
IEEE Standards Association. IEEE Standard Model Process for Addressing Ethical Concerns during System Design. IEEE 1–82 (IEEE, 2021).
Joly, Y., Saulnier, K. M., Osien, G. & Knoppers, B. M. The ethical framing of personalized medicine. Curr. Opin. Allergy Clin. Immunol. 14, 404–408 (2014).
Li, T., Sahu, A. K., Talwalkar, A. & Smith, V. Federated learning: challenges, methods and future directions. IEEE Signal Process. Mag. 37, 50–60 (2020).
Green, R. C., Lautenbach, D. & McGuire, A. L. GINA, genetic discrimination and genomic medicine. N. Engl. J. Med. 372, 397–399 (2015).
Lavin, A. et al. Technology readiness levels for machine learning systems. Nat. Commun. 13, 6039 (2022).
Reynolds, R. J. & Shelhamer, M. In Beyond LEO (ed. Reynolds, R. J.) 1–7 (IntechOpen, 2020).
Hanson, A. et al. A model-based systems engineering approach to exploration medical system development. In Proc. 2019 IEEE Aerospace Conference 1–19 (IEEE, 2019).
Auñón-Chancellor, S. M., Pattarini, J. M., Moll, S. & Sargsyan, A. Venous thrombosis during spaceflight. N. Engl. J. Med. 382, 89–90 (2020).
Marshall-Goebel, K. et al. Assessment of jugular venous blood flow stasis and thrombosis during spaceflight. JAMA Netw. Open 2, e1915011 (2019).
Vyas, R. J. et al. Decreased vascular patterning in the retinas of astronaut crew members as new measure of ocular damage in spaceflight-associated neuro-ocular syndrome. Invest. Ophthalmol. Vis. Sci. 61, 34 (2020).
Lagatuz, M. et al. Vascular patterning as integrative readout of complex molecular and physiological signaling by VESsel GENeration Analysis. J. Vasc. Res. 58, 207–230 (2021).
Weng, S. F., Reps, J., Kai, J., Garibaldi, J. M. & Qureshi, N. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS ONE 12, e0174944 (2017).
Paschalidis, Y. How machine learning is helping us predict heart disease and diabetes. Harvard Business Review (30 May 2017).
Lee, S. M. C., Stenger, M. B., Laurie, S. S. & Macias, B. R. Evidence report: risk of cardiac rhythm problems during spaceflight. NASA Human Research Roadmap (NASA, 2017).
Strodthoff, N. & Strodthoff, C. Detecting and interpreting myocardial infarction using fully convolutional neural networks. Physiol. Meas. 40, 015001 (2019).
Hannun, A. Y. et al. Cardiologist-level arrhythmia detection and classification in ambulatory electrocardiograms using a deep neural network. Nat. Med. 25, 65–69 (2019).
Nasir, M., Baucom, B. R., Georgiou, P. & Narayanan, S. Predicting couple therapy outcomes based on speech acoustic features. PLoS ONE 12, e0185123 (2017).
Frankel, J. How artificial intelligence could help diagnose mental disorders. The Atlantic (23 August 2016).
Landon, L. B., Slack, K. J. & Barrett, J. D. Teamwork and collaboration in long-duration space missions: going to extremes. Am. Psychol. 73, 563–575 (2018).
Willams, R. S. & Davis, J. R. A critical strategy: ensuring behavioral health during extended-duration space missions. Aviat. Space Environ. Med. 76, B1–B2 (2005).
Nelson, C. A., Bove, R., Butte, A. J. & Baranzini, S. E. Embedding electronic health records onto a knowledge network recognizes prodromal features of multiple sclerosis and predicts diagnosis. J. Am. Med. Inform. Assoc. 29, 424–434 (2021).
Stingl, J. C., Welker, S., Hartmann, G., Damann, V. & Gerzer, R. Where failure is not an option—personalized medicine in astronauts. PLoS ONE 10, e0140764 (2015).
Blue, R. S. et al. Supplying a pharmacy for NASA exploration spaceflight: challenges and current understanding. NPJ Microgravity 5, 14 (2019).
Ashley, E. A. Towards precision medicine. Nat. Rev. Genet. 17, 507–522 (2016).
Wesseling, P. & Capper, D. WHO 2016 classification of gliomas. Neuropathol. Appl. Neurobiol. 44, 139–150 (2018).
Baribeau, Y. et al. Handheld point-of-care ultrasound probes: the new generation of POCUS. J. Cardiothorac. Vasc. Anesth. 34, 3139–3145 (2020).
Hashimoto, D. A., Rosman, G., Rus, D. & Meireles, O. R. Artificial intelligence in surgery: promises and perils. Ann. Surg. 268, 70–76 (2018).
Haidegger, T., Sándor, J. & Benyó, Z. Surgery in space: the future of robotic telesurgery. Surg. Endosc. 25, 681–690 (2011).
Akkus, Z. et al. A survey of deep-learning applications in ultrasound: artificial intelligence-powered ultrasound for improving clinical workflow. J. Am. Coll. Radiol. 16, 1318–1328 (2019).
Bowness, J., Varsou, O., Turbitt, L. & Burkett-St Laurent, D. Identifying anatomical structures on ultrasound: assistive artificial intelligence in ultrasound-guided regional anesthesia. Clin. Anat. 34, 802–809 (2021).
Mertens, C. J., Slaba, T. C. & Hu, S. Active dosimeter-based estimate of astronaut acute radiation risk for real-time solar energetic particle events. Space Weather 16, 1291–1316 (2018).
Toscano, W. et al. Wearable biosensor monitor to support autonomous crew health and readiness to perform. NASA Technology Reports Server (NASA, 2017).
Marković, D., Mizrahi, A., Querlioz, D. & Grollier, J. Physics for neuromorphic computing. Nat. Rev. Phys. 2, 499–510 (2020).
Liu, D., Yu, H. & Chai, Y. Low‐power computing with neuromorphic engineering. Adv. Intell. Syst. 3, 2000150 (2021).
Goecks, J., Jalili, V., Heiser, L. M. & Gray, J. W. How machine learning will transform biomedicine. Cell 181, 92–101 (2020).
Banbury, C. R. et al. Benchmarking TinyML systems: challenges and direction. Preprint at https://arxiv.org/abs/2003.04821 (2020).
Wang, Y., Yao, Q., Kwok, J. T. & Ni, L. M. Generalizing from a few examples: a survey on few-shot learning. ACM Comput. Surv. 53, 1–34 (2020).
Hu, F., Xie, D. & Shen, S. On the application of the Internet of Things in the field of medical and health care. In Proc. 2013 IEEE International Conference on Green Computing and Communications and IEEE Internet of Things and IEEE Cyber, Physical and Social Computing 2053–2058 (IEEE, 2013).
Cheng, X. & Liu, H. A novel post-processing method based on a weighted composite filter for enhancing semantic segmentation results. Sensors (Basel) 20, 5500 (2020).
Jiang, H. & Nachum, O. Identifying and correcting label bias in machine learning. In Proc. 23rd Intl. Conf. on AISTATS 1–10 (2020).
Krueger, D. et al. Out-of-distribution generalization via risk extrapolation (REx). In Proc. 38th Intl. Conf. on MachineLearning 139, 5815–5826 (PMLR, 2021).
Nguyen, A.T., Tran, T., Gal, Y. & Baydin, A. G. Domain invariant representation learning with domain density transformations. Preprint at https://arxiv.org/pdf/2102.05082.pdf (2021).
Jin, Z., Sun, Y. & Cheng, A. C. Predicting cardiovascular disease from real-time electrocardiographic monitoring: an adaptive machine learning approach on a cell phone. In Proc. 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society 6889–6892 (IEEE, 2009).
Acknowledgements
We thank all June 2021 participants and speakers at the ‘NASA Workshop on Artificial Intelligence & Modeling for Space Biology’. Thanks go to the NASA Space Biology Program, part of the NASA Biological and Physical Sciences Division within the NASA Science Mission Directorate, as well as the NASA Human Research Program (HRP). We also thank the Space Biosciences Division and Space Biology at Ames Research Center (ARC), especially D. Ly, R. Vik and P. Vaishampayan. We are grateful for the support provided by NASA GeneLab and the NASA Ames Life Sciences Data Archive. Additional thanks go to S. Bhattacharya (NASA Space Biology Program Scientist), K. Martin (ARC Lead of Exploration Medical Capability (an Element of HRP)), as well as L. Lewis (ARC NASA HRP Lead). S.V.C. is funded by NASA Human Research Program grant NNJ16HP24I. S.E.B. holds the Heidrich Family and Friends Endowed Chair in Neurology at UCSF. S.E.B. also holds the Distinguished Professorship I in Neurology at UCSF. S.E.B. is funded by an NSF Convergence Accelerator award (2033569) and NIH/NCATS Translator award (1OT2TR003450). G.I.M. was supported by the Translational Research Institute for Space Health, through NASA NNX16AO69A (project no. T0412). E.L.A. was supported by the Translational Research Institute for Space Health, through NASA NNX16AO69A. C.E.M. acknowledges NASA grants NNX14AH50G and NNX17AB26G. This work was also part of the DOE Agile BioFoundry, supported by the US Department of Energy, Energy Efficiency and Renewable Energy, Bioenergy Technologies Office, and the DOE Joint BioEnergy Institute, supported by the Office of Science, Office of Biological and Environmental Research, through contract no. DE-AC02-05CH11231 between Lawrence Berkeley National Laboratory and the US Department of Energy. S.V.K. is funded by the Canadian Space Agency (19HLSRM04) and Natural Sciences and Engineering Research Council (NSERC, RGPIN-288253). J.H.Y. is funded by NIH grant no. R00 GM118907 and the Agilent Early Career Professor Award.
Author information
Authors and Affiliations
Contributions
All authors contributed ideas and discussion during the joint workshop writing session or were speakers at the ‘NASA Workshop on Artificial Intelligence & Modeling for Space Biology’. R.T.S., L.M.S. and S.V.C. prepared the manuscript. All authors provided input and feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Machine Intelligence thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Scott, R.T., Sanders, L.M., Antonsen, E.L. et al. Biomonitoring and precision health in deep space supported by artificial intelligence. Nat Mach Intell 5, 196–207 (2023). https://doi.org/10.1038/s42256-023-00617-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42256-023-00617-5
This article is cited by
-
Physiological evidence of stress reduction during a summer Antarctic expedition with a significant influence of previous experience and vigor
Scientific Reports (2024)
-
Augmented and Virtual Reality-Based Cyber Twin Model for Observing Infants in Intensive Care: 6G for Smart Healthcare 4.0 by Machine Learning Techniques
Wireless Personal Communications (2024)
-
Biological research and self-driving labs in deep space supported by artificial intelligence
Nature Machine Intelligence (2023)