Abstract
Blue carbon ecosystems (BCEs), including mangrove forests, seagrass meadows and tidal marshes, store carbon and provide co-benefits such as coastal protection and fisheries enhancement. Blue carbon sequestration has therefore been suggested as a natural climate solution. In this Review, we examine the potential for BCEs to act as carbon sinks and the opportunities to protect or restore ecosystems for this function. Globally, BCEs are calculated to store >30,000 Tg C across ~185 million ha, with their conservation potentially avoiding emissions of 304 (141–466) Tg carbon dioxide equivalent (CO2e) per year. Potential BCE restoration has been estimated in the range of 0.2–3.2 million ha for tidal marshes, 8.3–25.4 million ha for seagrasses and 9–13 million ha for mangroves, which could draw down an additional 841 (621–1,064) Tg CO2e per year by 2030, collectively amounting to ~3% of global emissions (based on 2019 and 2020 global annual fossil fuel emissions). Mangrove protection and/or restoration could provide the greatest carbon-related benefits, but better understanding of other BCEs is needed. BCE destruction is unlikely to stop fully, and not all losses can be restored. However, engineering and planning for coastal protection offer opportunities for protection and restoration, especially through valuing co-benefits. BCE prioritization is potentially a cost-effective and scalable natural climate solution, but there are still barriers to overcome before blue carbon project adoption will become widespread.
Key points
-
Blue carbon ecosystems (BCEs), including mangrove forests, tidal marshes and seagrass meadows, are gaining international recognition as a natural climate solution to contribute to climate change mitigation and adaptation targets.
-
Global distribution is estimated as ~36–185 million ha of BCEs, potentially storing ~8,970–32,650 Tg C and providing important co-benefits.
-
Protecting existing BCE could avoid emissions of 304 (141–466) Tg (95% CI bounds) carbon dioxide equivalent (CO2e) per year and large-scale restoration could remove an extra 841 (621–1,064) Tg CO2e per year by 2030, equivalent to ~3% (0.5–0.8% from protection and 2.3–2.5% from restoration) of annual global greenhouse gas emissions.
-
Blue carbon’s potential as a nature-based solution will depend on societal actions; restoring BCE should be a key focus of the UN Decade on Ecosystem Restoration (2021–2030).
-
Emerging blue carbon markets should aim to incorporate the value of co-benefits into financial frameworks to assist with the investments required for restoration and conservation.
Similar content being viewed by others
Introduction
To limit global average temperature rise to below 2 °C above pre-industrial levels, emissions must be reduced and excess greenhouse gases removed from the atmosphere. An emerging option to achieve both of these goals is natural climate solutions1,2, including carbon sequestration through ecosystem management and reforestation. Natural climate solutions have the potential to be more cost-effective and scalable than technological options3,4, such as direct air capture, geological sequestration and biochar production, many of which are yet to be deployed at large scale and face significant economic, social and environmental barriers5.
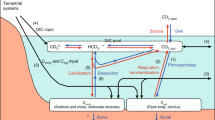
Originally, natural climate solutions were based almost exclusively on green carbon (terrestrial) ecosystems, largely ignoring coastal and ocean-based opportunities for carbon sequestration6. However, there has been increased appreciation of blue carbon ecosystems (BCEs), including seagrass meadows, mangrove forests, tidal marshes and, potentially, seaweed beds, since 2009. BCEs are widespread, highly productive coastal habitats that host diverse ecological communities and support human well-being, providing food and coastal protection against erosion and sea-level rise, influencing the livelihoods of millions7,8,9. These systems could substantially contribute to carbon drawdown due to their intense greenhouse gas removal, the long residence times of sequestered carbon and the large stocks accumulated (Fig. 1). Conversely, disturbance of BCEs could potentially act as large potential greenhouse gas sources10. Therefore, blue carbon strategies propose the conservation and restoration of these ecosystems as a strategy to mitigate and adapt to climate change11.
a | Key elements and processes in blue carbon cycling. Blue carbon ecosystems (BCEs) draw down CO2 from the atmosphere through photosynthesis, contributing to net accumulation of organic carbon within plant biomass and sediments. BCEs can also accumulate organic carbon from outside sources (such as via terrestrial run-off and plankton). Anoxic sediment conditions and positioning of BCEs in depositional settings make them well suited for carbon accumulation. Environmental factors such as climate change and coastal development can diminish stored blue carbon stocks by making them more susceptible to microbial degradation to inorganic carbon or greenhouse gases (such as CO2). b | Timeline showing evolution of blue carbon notable papers (classified as ‘ISI Highly Cited’ papers with ‘blue carbon’ title as at 3 June 2021, plus one paper from 1981 that reported carbon drawdown by BCE well before the ‘blue carbon’ term was coined), major moments in international climate change policy and key events in blue carbon policy and management. CDM, clean development mechanism.
BCEs occupy approximately 0.5% of the sea floor, from the upper intertidal zone down to about 50 m deep (depending on underwater light penetration), but contribute >50% of global carbon burial in the oceans12,13,14. Large amounts of autochthonous carbon are fixed in BCEs, and their position at the land–sea interface and high trapping capacity allows them to accumulate sediment and allochthonous carbon produced in other ecosystems11 (Fig. 1). The water-logged, anaerobic burial conditions then limit decomposition15. Compared with terrestrial systems, the general absence of fires in BCEs provides some confidence in the permanence of carbon stores over climatically relevant centennial to millennial timescales, despite BCE exposure to natural disturbances such as cyclones and flooding11,16. Unfortunately, around 50% of BCE global extent has been lost11, leading to greenhouse gas emissions and limiting their role in carbon sequestration17. Hence, halting and reversing the loss of BCEs is therefore urgent6.
Only about 1.5% of the global extent of BCEs is included in marine protected areas (MPAs)18, leaving ample scope for both conservation and restoration of BCEs. Restoration of BCEs at large scale is potentially feasible for mangroves and tidal marshes, with tenfold growth in the number of documented restoration projects of BCEs since the turn of the century19, although it remains challenging for seagrass meadows20. One of the largest ecosystem restoration projects ever undertaken was the restoration of ~1,487 km2 of mangroves in the Mekong Delta destroyed by the US Air Force during the Vietnam War19,21. To date, the planted forest in Can Gio Mangrove Forest Park, Vietnam, has since 1978 accumulated an estimated 152 Tg carbon dioxide equivalent (CO2e), across the 447 km2 of mangroves restored at this site22, even with the impact of typhoons. As the total carbon emissions of Vietnam for the year 2013 were 130 Tg CO2e, the total Mekong mangrove restoration area removed an equivalent of around three times Vietnam’s 2013 greenhouse gas emissions23.
An important aspect of BCE restoration, and natural climate solutions more broadly, is that unlike more technological climate change mitigation solutions, they also generate a range of other ecosystem services such as nutrient removal24, fisheries enhancement25 and coastal protection26 (Fig. 2). For example, the establishment of mangrove protected areas has been associated with long-term gains in fisheries production27. Mangroves are also increasingly valued for their contribution to protecting communities from the impacts of tropical storms and cyclones28,29. In short, BCE services increase the adaptive capacity of communities to cope with natural hazards and climate change, while helping achieve multiple United Nations Sustainable Development Goals. As such, the conservation and restoration of BCEs can be considered a win–win solution that contributes to both climate change mitigation and adaptation, while enhancing coastal livelihoods30,31.
In this Review, we discuss the global potential of blue carbon contributions to climate change mitigation and adaptation, focusing on the large-scale restoration opportunities. Global estimates of blue carbon stocks are described, and the main constraints and feasibility of large-scale restoration of BCEs are outlined. The role of BCEs in helping countries achieve their international emission reduction targets is examined, and future steps are identified to improve blue carbon estimates on distribution extent, stocks, sequestration and mitigation potential.
Global blue carbon stocks
Robust information on geographical extent and estimates of carbon storage are key to including BCEs in national and global climate mitigation accounting. Based on current mapping efforts, BCEs encompass ~36–185 million ha within the world’s coastal zone32,33,34 (with the wide range due to large uncertainties in the distribution of seagrass meadows34,35 and tidal marshes32) and could potentially hold ~8,970–32,650 Tg C in their soils and biomass. However, one major caveat associated with these global estimates is that all data supporting this Review have been extracted from existing literature, which usually did not provide error propagation or uncertainty estimates. Therefore, upper and lower interval bounds for soil and biomass carbon stocks were estimated based on available information (including spatial data or country-level estimates; Supplementary Table 1) and are not definitive. A full description of existing data sets and their use here is available in Supplementary Methods.
Amongst BCEs, mangrove forests are the most studied (in terms of publication numbers) and best mapped, occurring across ~13.7 million ha within tropical, subtropical and warm temperate coastal zones33 (Fig. 3). Different carbon pools have different turnover times, with fresh plant litter rapidly decomposing over years, stabilized soil organic matter persisting for thousands of years to centuries and poorly defined intermediate carbon pools with turnover times of years to centuries13,36. Mangrove forests potentially hold ~70% of their carbon in the soil37, with global estimates ranging from 1,900 Tg C (ref.38) to 8,400 Tg C (ref.39) for the top metre of soil (Supplementary Table 1). Carbon stocks in living biomass are estimated between 1,230 Tg C (ref.37) and 3,900 Tg C (ref.40).
Systems include mangrove forests (data from ref.33), tidal marshes (data from ref.32) and both mapped48 and modelled34 distributions of seagrass meadows (data from refs34,48). Blue carbon ecosystems (BCEs) encompass ~36–185 million ha along coastal zones globally, with large uncertainties in the distribution of seagrass meadows and tidal marshes.
Here, the data available from ref.41 and ref.40 were used for lower-bound and upper-bound biomass stock estimates, respectively, and those from ref.42 and ref.43 for soil stocks (Supplementary Methods; Supplementary Tables 1–3). Based on these data, ~40–50% of global soil and biomass carbon stocks within mangrove forests were in Indonesia (soil ranging from ~830 to 1,780 Tg C; biomass ranging from ~570 to 1,040 Tg C), Brazil (soil ranging from ~235 to 514 Tg C; biomass ranging from ~98 to 313 Tg C), Australia (soil ranging from ~83 to 464 Tg C; biomass ranging from ~113 to 115 Tg C) and Malaysia (ranging from ~200 to 365 Tg C; biomass ranging from ~95 to 255 Tg C), regardless of the data set used to estimate carbon stocks (Supplementary Tables 2, 3). Indonesia alone holds more than 20% of this total (Fig. 4; Supplementary Tables 2, 3). Mangrove soils alone have lost an estimated 30.4–122 Tg C due to land-use changes that occurred between 2000 and 2015, with Indonesia, Malaysia and Myanmar contributing to more than 75% of these losses43.
a | Upper bound of blue carbon soil stocks per country (teragrams of carbon). b | Lower bound of blue carbon soil stocks per coastline length (teragrams of carbon). c | Upper bound of blue carbon soil stocks per coastline length (teragrams of carbon per kilometre). d | Lower bound of blue carbon soil stocks per coastline length (teragrams of carbon per kilometre). Blue carbon ecosystems (BCEs) potentially hold ~8,970– 32,650 Tg C in their soils and biomass, with most of their carbon stored in soil. Full description of methods and data sources used available in Supplementary Methods. Blue carbon stocks are along the coastline, with colour coding by country for display purposes only.
Mangrove forests have been the main focus of research on the distribution of blue carbon stocks in BCEs. In comparison, seagrass meadows and tidal marshes still lack robust and spatial-explicit estimates of soil carbon within their geographical extents, although there have been efforts to map the habitat extent and soil carbon at local and regional scales for seagrass44,45,46 and tidal marshes32,38. In addition, the Coastal Carbon Research Network, which was created by the Smithsonian Environmental Research Centre in 2018, is developing publicly available interactive maps of existing soil core data47.
Considering their mapped35,48 and potential34 distribution, seagrass meadows potentially cover 16–165 million ha globally, with these estimates derived from aggregating mapped seagrass areas and modelling the ocean area suitable to support seagrass growth, respectively. Soil carbon stocks are therefore estimated to vary from ~1,732 to 21,000 Tg C (Supplementary Methods). Such an order of magnitude range is associated with the large uncertainties in seagrass mapping, the methodological differences and seagrass bed variability. For example, compilations35,48 of existing data sets of mapped seagrass meadows have estimated with low confidence that seagrass meadows could occupy 27 million ha35. Using global occurrence records of seagrass and environmental variables to model the potential distribution of seagrass meadows results in estimates of 165 million ha34. Regardless, there is still a lack of data on the eastern and western coast of South America, Africa, the Indian Ocean and the Indo-Pacific region, and vast meadows of deep-water seagrasses (depths >50 m) are only beginning to be discovered49,50,51,52. Based on current information, Australia, Indonesia and the USA are the countries with the largest area of seagrass meadows, regardless of the data set used here (Supplementary Table 4). If considering the upper and lower bounds for seagrass distribution extent (Supplementary Tables 1, 4)34,48 and the global estimates of sediment carbon stocks available in ref.46, carbon stocks could vary from ~3,760 to 21,000 Tg C with Australia (carbon stocks varying from ~37 to 2,320 Tg C), the USA (from ~6 to 1,700 Tg C) and Indonesia (from ~41 to 1,320 Tg C) accounting for ~2–25% of the total carbon stocks within seagrass meadows (Fig. 4; Supplementary Methods). Even within these well-studied regions, however, there are some limitations that could potentially underestimate and/or overestimate carbon inventories. For example, Australia is one of the few countries with a full published inventory of BCE carbon stocks, which shows that this ecosystem would hold ~762–1,051 Tg C (ref.53). Using Australia as an example, it is apparent that seagrass extent layers from either ref.34 or ref.48 could lead to incorrect carbon inventories, highlighting that global maps and estimates are a first step towards estimating blue carbon stocks; however, countries that invest in BCE mapping and carbon sampling will have a better evaluation of its carbon storage capacity, required to support policy actions.
As with seagrass meadows, global distribution maps of tidal marshes remain incomplete. The global area of tidal marshes has been suggested to be in the range of 2.2–40 million ha12,54, with the most comprehensive study to date estimating ~5.5 million ha across 43 countries32. However, this estimate is likely conservative and incomplete32. For example, there are known areas of tidal marshes in Canada, Russia, South America and Africa that are not reflected on current maps, and so are not included in the global area estimate. In addition, despite their importance, a global and spatially explicit map of soil carbon is still lacking for tidal marshes. Based on the global tidal marsh distribution mapped in ref.32, the global soil map55 and mean carbon stocks (Supplementary Tables 1, 5), it is estimated here that potential blue carbon stocks in tidal marsh soil could vary from ~862 to 1,350 Tg C (Supplementary Tables 1, 5). The USA (354–640 Tg C), Australia (112–223 Tg C) and Russia (180–277 Tg C) hold approximately 77–86% of the global soil tidal marsh carbon stocks (Fig. 4; Supplementary Table 5).
Improved global-scale mapping of BCEs, particularly seagrasses and tidal marshes, is required to better understand the contribution of BCEs to the global carbon cycle. Furthermore, it is important to highlight that the range of carbon stock estimates for each ecosystem and carbon pool (biomass and soil) provided in this Review reflects the large uncertainties associated with global blue carbon estimates (for example, linked to poorly constrained total seagrass and tidal marsh habitat extent and the large variability in carbon storage among blue carbon habitats across and within countries). Although large uncertainties remain (such as propagation and uncertainty estimates for each country and global values), the ranges provided here encompass the known variability in BCE extent and stocks across countries (Supplementary Table 1). Other substantial research gaps include understanding the spatial variability — and drivers of such variability — for key carbon parameters. For example, although there have been improvements in understanding how and why soil carbon stocks vary globally in tidal marshes56 and mangroves57,58, global drivers of seagrass carbon stock remain largely unexplored. Less still is known about patterns and drivers of carbon fluxes (carbon burial rates, atmospheric and/or lateral greenhouse gas fluxes) beyond particular sites or regional studies59,60. Better understanding of these factors will improve attempts to estimate and model BCE contributions to global carbon cycling, and enhance capacity to predict the greenhouse gas benefits of restoration activities at national scales61.
The scope for large-scale restoration
Delivering the full potential of blue carbon restoration as a natural climate solution requires returning BCEs to their historical extent, knowledge of which remains poorly constrained in many cases. Coastal development has modified BCEs in some locations to an extent that restoration is not possible because it is either economically, legally and/or logistically unfeasible. Nevertheless, wetland conversion to an urban area represents a relatively minor component of the area lost. For example, this conversion accounted for just 3% of mangrove loss globally between 2000 and 2016 (ref.62), although it might have been much greater during the nineteenth and twentieth centuries. In many regions, BCEs have largely been claimed for rice paddies (in Asia), aquaculture ponds (Asia and Central America) and pasture lands (temperate tidal marshes)63,64 — a conversion that in some cases was initiated centuries ago. Compared with urban lands, it is more feasible to restore these lands to BCEs once they fall out of production, given appropriate engineering and land tenure considerations. Successful tidal marsh and mangrove restoration also depends on sediment supply in minerogenic systems, as well as current and future soil elevation. Mapping converted areas that can be returned to the original habitat has shown that there is the potential for >800,000 ha globally to be biophysically suitable for restoration back to mangrove forests65. Likewise, restoring tidal flows can lead to rapid tidal marsh restoration, with tidal marsh restoration successfully expanding tidal marshes even in some of the most densely urbanized cities in the world, such as New York City, USA19. However, restoration failures have also occurred, providing opportunities to learn19,66,67,68.
The scope for global-scale coastal wetland restoration is constrained by multiple socio-economic considerations. This constraint is especially important in countries where a large proportion of the restorable habitat is on small agricultural land holdings, where restoration efforts could conflict with the livelihoods and food security of local communities69,70. Restoration efforts in Southeast Asia highlight the impact of socio-economic constraints on wetland restoration — only 5.5–34.2% of the area biophysically suitable for mangrove restoration is ultimately restorable after various socio-economic (such as livelihoods, food security and land rights) and operational (including deforestation risk, site accessibility and proximity to seed sources) constraints are considered71.
Compared with other marine ecosystems (such as mangrove forests, tidal marshes, coral reefs and oyster reefs), seagrass restoration is costly and has a lower success rate20,72. Furthermore, carbon stocks in seagrass biomass and marsh grass leaves are usually not included in carbon inventories, as the residence time varies from only a few months to a few decades and, therefore, is irrelevant to climate change mitigation. Around 29% of the known seagrass global extent has disappeared since the 1940s at a mean rate of 1.5% per year, with large-scale losses reported in the USA, Australia, New Zealand and Europe73. Yet there is potential to restore areas formerly occupied by seagrass if the pressures that led to the loss (namely, deteriorated water quality with eutrophication74) are removed75. However, natural restoration is a very slow process. For example, more than 70 million seeds of eelgrass (Zostera marina) have been distributed in the western Atlantic coastal lagoons since 1999, resulting in the recovery of 3,600 ha after >20 years76,77.
Overall, seagrass restoration has been largely unsuccessful to date78, requiring improved restoration practices. Various tools and methods (for example, buoy-deployed seeding, dispenser injection seeding, artificial in-water structures and community involvement) can enhance the cost-effectiveness, efficiency and scalability of restoration activities75. Active restoration can catalyse natural recovery processes24,72,79. These processes will enhance the recovery of the associated ecosystem services such as carbon sequestration, which can otherwise be slow if left to natural recovery, as demonstrated for seagrass meadows in coastal waters in Virginia, USA24. Restoration of seagrass meadows is complex but possible, and future efforts should combine actions to improve the suitability of the habitat to allow for natural recovery processes in seagrasses, in addition to state-of-the-art methods that enhance the success of active restoration programmes77.
Blue carbon losses from human activities
Quantifying BCE losses has been hampered by a lack of understanding of their historical and current spatial extent, in particular for seagrasses and tidal marshes. The lack of global time series for tidal marsh areas comparable with those for mangroves33,64,80 is surprising, as marshes can also be detected with remote sensing. However, tidal marsh mapping can be challenging due to their spatial, temporal and spectral complexity, particularly when bordering other grassland and wetland types81,82. High-enough spatial resolution of satellite products for this purpose has only just been achieved. Moreover, resolving seagrass area and distribution is still difficult with remote sensing, as it is challenging to detect seagrass meadows underwater and distinguish them from other communities such as macroalgae83 in satellite images. Furthermore, the lack of remote sensing data before the 1970s precludes a robust assessment of global BCE extent prior to the expansion of coastal settlements.
Despite these difficulties, global losses of tidal marshes have been estimated at ~1–2% per year84 and seagrass loss rates since the 1940s have been estimated at 1.5% per year73, although conservation actions have resulted in the deceleration and reversal of declining trends in seagrasses in some regions85. Remote sensing methods for terrestrial forests have been adapted for mangroves, allowing for a country by country estimate of ecosystem change from 2000 to 2016 (ref.62). Globally, annual loss rates of mangroves have decreased sixfold from 0.99% in the 1980s to an average of 0.16–0.39% per year between 2000 and 2012 (ref.86). However, loss rates are still substantial in places such as Southeast Asia, which contains ~50% of the global mangrove forest area. Some Southeast Asian countries such as Myanmar experienced loss rates as high as 0.5% per year between 2000 and 2016 (with 44,485 ha deforested between 2000 and 2016)62.
The major drivers of BCE loss vary by ecosystem and region, but generally include physical modification (including ecosystem change and drainage), pollution, non-native species and climate change87. In most cases, the primary drivers of BCE loss are indirectly driven by socio-economic factors that centre around coastal development, energy, food, infrastructure expansion, and recreation and/or tourism. For example, mangrove forests continue to be converted to expand aquaculture and agriculture (such as rice and oil palm), often to meet national food and economic security targets. Commodity production accounted for 47% of global mangrove loss in the early twenty-first century, with 92% of commodity production occurring in Southeast Asia alone62,63. Similarly, extensive use of fertilizers on agricultural fields and the subsequent eutrophication of coastal ecosystems has been identified as a major driver of large-scale seagrass and tidal marsh loss73,74,88.
The effects of climate change (including increased temperatures, sea-level rise, and increased frequency and severity of storms and heatwaves) on BCE loss have been increasing over time62,89,90. For example, increases in the occurrence and extent of marine heatwaves have led to extensive losses (36–80% loss in seagrass cover) in temperate and subtropical seagrasses in Chesapeake Bay, USA, the western Mediterranean and Western Australia’s Shark Bay91,92,93,94. Some mangrove forests, such as those in the Gulf of Carpentaria, Australia, have experienced dieback from factors associated with extreme El Niño events (for example, elevated temperatures, reduced inundation and drought)95. Cyclones have also led to widespread damage and losses in mangrove forests16,96,97.
Although it is difficult to show causality98, increasing air temperature has been related to the poleward range extension of mangroves in many instances99,100,101,102,103. For example, in Western Port, Australia, mangrove encroachment was related to increasing annual maximum temperature at sites with lower elevations and closer to known mangrove–tidal marsh boundaries100. This expansion has generally come at the expense of tidal marshes as they become displaced by mangroves, and is associated with increases in blue carbon as a carbon-poor herbaceous ecosystem is replaced by a woody ecosystem104,105.
Sea-level rise combined with coastal squeeze — habitat loss in the intertidal area due to anthropogenic structures or actions, which prevent the landward migration of coastal habitats that would otherwise occur in response to sea-level rise — and reduced sediment loads to coastal systems from rivers damming threatens the future persistence of many mangrove and tidal marsh ecosystems106,107,108,109. Although the effects of sea-level rise on BCE loss might not begin to truly manifest for several decades, 5–30% of coastal wetlands could be submerged by 2080 if upslope migration pathways, through lateral accommodation space, are precluded100,110,111. Conversely, moderate rates of sea-level rise can be beneficial to the burial of BCE soil carbon in areas that have experienced stable relative sea level over the past millennia56 — such as much of the southern hemisphere — constraining the soil space available to accumulate sediments by BCEs.
Blue carbon ecosystem restoration
The protection and restoration of BCEs have the potential to add substantially to climate mitigation efforts and nationally determined contributions (NDCs). Under business as usual operations, BCEs are expected to decline further. If protection is prioritized, however, then blue carbon trajectories should stabilize112 and even reverse towards recovery19. Rehabilitation and restoration of BCEs will increase the contribution of blue carbon to natural climate solutions, as will planning for sea-level rise to maximize accommodation space for BCEs.
Protection of BCEs could avoid an estimated 304 (141–466) Tg CO2e per year113, whereas large-scale restoration could draw down an extra 841 (621–1,064) Tg CO2e per year by 2030 through further avoided emissions and additional carbon sequestration from growth (mangroves only) and soil carbon sequestration (Fig. 5). For example, the maximum mitigation potential from avoided coastal impacts has been estimated for mangrove forests113, showing that countries in Southeast Asia would have the highest mitigation potential at up to 65 Tg CO2e per year (Fig. 5). Considering the estimated global annual emissions from fossil fuels for 2019 and 2020 (ref.114) (36,400 and 34,100 Tg CO2e, respectively), the mitigation potential equates to potential abatement of ~3% of global emissions (0.5–0.8% from protection and 2.3–2.5% from restoration; further details on this estimate are available in Supplementary Methods). Although mangroves and seagrasses contribute equally to avoided emissions potential through protection of the existing habitat, mangroves contribute the largest proportion to mitigation potential from the restoration of disturbed or lost habitats113 (Fig. 5). The substantial contribution of mangroves to mitigation potential through restoration compared with seagrasses and tidal marshes, in part, likely reflects the broader understanding of ecosystem loss, and thus the opportunities for restoration in mangroves compared with the other systems. As the full extent of habitat loss for seagrasses and tidal marshes is unknown, the potential for their restoration to contribute to climate mitigation efforts estimated by Griscom et al.113 is more uncertain. The estimated maximum extent of available habitat for restoration ranged from 0.2 to 3.2 million ha for tidal marshes, and from 8.3 to 25.4 million ha for seagrasses113. In contrast, the restorable habitat for mangroves is much more resolved, with estimates between 9 and 13 million ha65,113. Such large differences in the estimated restorable habitat for tidal marshes and seagrasses cause potentially avoidable greenhouse gas fluxes and additional carbon sequestration to vary by orders of magnitude.
Protecting existing blue carbon ecosystems (BCEs) through avoided emissions and large-scale restoration could be equivalent to ~3% of annual global greenhouse gas emissions. a | Maximum mitigation potential at country level for avoided coastal impacts in mangrove forests (teragrams of carbon dioxide equivalent (CO2e) per year)113. b | Estimated annual loss rates (%) for seagrass (between 1879 and 2006)73, tidal marshes84 and mangroves (between 2000 and 2012)86. c | Global average soil carbon stocks (for mineral soils) per area (megagrams of carbon per hectare)134. d | Avoidable flux for avoided coastal wetland impacts the pathway (megagrams of carbon per hectare)113. e | Avoidable flux and additional sequestration rates (95% CI) for the coastal wetland restoration pathway (megagrams of carbon per hectare per year)113. f | Potential area extent available to restoration (millions of hectares)113. g | Maximum mitigation potential from avoided emissions due to conversion (dark blue; mineralization of carbon stocks in biomass and soils, and loss of carbon sequestration potential (teragrams of CO2e per year))113 and maximum mitigation potential from restoration (orange; mineralization of carbon stocks in biomass and soils, and recovery of carbon sequestration potential (teragrams of CO2e per year))113. Maximum mitigation potential for avoided emissions assumes that 100% of carbon stocks are lost following habitat loss113 (1 Mg ha = 1,000,000 ha and 1 Tg = 1,000,000 Mg). Uncertainty estimates (grey bars) represent 95% CIs. Tidal marshes and seagrass meadows do not have information available on potential for avoided emissions per country.
It is not realistic to assume that all BCEs that have been lost can be restored. In cases such as the conversion of mangroves to a port or of tidal marsh to urban development, it is improbable that these lands can be converted to their natural state. For example, it has been estimated that 17% of the 973,640 ha of mangroves lost between 1996 and 2016 is not restorable due to urbanization, erosion and, possibly, restoration costs65. However, the creation and expansion of new BCEs through engineering solutions (including modification of hydrodynamics to restore historical tidal exchange) and planting has the potential to add to NDCs (assuming plantations do not occur on or convert other BCEs), while deriving additional benefits such as coastal defence against sea-level rise115. Thus, future studies should aim to identify BCEs for restoration as well as identify potential new areas for the expansion of BCEs. Furthermore, the time taken for restored ecosystems to achieve carbon stocks that are equivalent to natural ecosystems and data on the trajectory of carbon accumulation with time since restoration are limited, with an opportunity for future studies to answer this fundamental question in blue carbon science116. Whether restoration and conservation measures are effective for long-term survival depends on numerous contributing factors. To ensure success, restoration policies should be based on globally accepted best practice, with particular attention to site suitability, while taking into account local conditions and the management policies that are relevant to the local community67,117,118,119,120,121.
Practical aspects of restoration
Whereas conservation of remaining BCEs is a cost-effective way of reducing greenhouse gas emissions, restoration requires substantial investments and is, therefore, less cost-effective113. However, both conservation and restoration contribute to climate change adaptation, and therefore restoration offers opportunities to develop market-based mechanisms that take advantage of existing frameworks for carbon offsets122. For example, application of the Verified Carbon Standard methodology for tidal wetland and seagrass restoration (VM0033) has created an opportunity for projects to gain financial help through carbon offsets. Projects can be developed in partnership with those wanting to purchase offsets, whether in the public or private sector, with the chance to make additional contributions from other financial streams30. For instance, 11,000 ha of mangrove forest are being protected and restored in Cispatá Bay, Colombia, which will reduce potential emissions by 17,000 metric tons of CO2 within 2 years123. Conservation International, along with Colombia community partners, intend to use the carbon value generated through the project to contribute to a long-term sustainable financing strategy for the region, and Apple is investing in the project as part of their emissions reduction strategy123. Alongside the growing experience of restoration has come refinement of the Verified Carbon Standard methodology, so that projects that follow these procedures should be successful as experience grows. In many cases, simple restoration methods such as natural or purposeful reseeding or replanting can be cost-effective.
BCE restoration provides ecosystem service benefits besides carbon drawdown that support coastal communities and their economies7,124. Many open up opportunities for developing payment for ecosystem service schemes that are complementary to carbon management. These schemes can provide flexibility in financing and a broader outlook on ecosystem service provision. Moreover, ecosystem service co-benefits from BCE restoration are supported by a wide range of policy goals — contributing to multiple Sustainable Development Goals, the Convention on Biodiversity Aichi targets, the Ramsar Convention and UNESCO (Biosphere Reserves, Natural World Heritage sites), amongst others — and community goals for development125,126. Achieving the restoration scale required to deliver maximum climate change mitigation benefits from BCEs (Fig. 5) needs strong governance, and support from governments, beneficiary industries, corporations and communities committed to the UN Decade on Ecosystem Restoration127.
MPAs and other listed sites can be used to help achieve the Aichi and Durban targets (which call for protecting 10% of marine areas within natural jurisdictions and an expansion of MPAs by 30%, respectively)128,129 and Sustainable Development Goals (Target 14.1). A common feature of MPA establishment is to protect and conserve biodiversity, rather than carbon, meaning that some proposed locations for new MPAs might not be able to serve both goals. An economic analysis of MPA development to reach the expansion targets concluded that although benefits exceed costs, regardless of the degree of human impact in an area, expansion into more biodiverse low-usage areas would be the most cost-effective130. In terms of climate change mitigation, use of the same criteria to locate MPA expansion might not yield the same cost benefit, as projects can only claim carbon credits if they can verify that the carbon stored there would be at risk131. In this context, established MPAs might not meet the requirements of ‘additionality’ criteria of existing blue carbon methodologies132 — that is, projects are generally unable to claim credits for carbon abatement achieved through conservation or restoration actions that are not already prescribed by existing legislation or management requirements. Additionality is an area of current policy debate and development, with approaches likely to vary among jurisdictions and accounting mechanisms133.
Relevance to nationally determined contributions
NDCs to the Paris Agreement (2016) encourage nations to decide what activities they will undertake to lower their greenhouse gas emissions. Currently, blue carbon commitments within NDCs are limited. An estimated 64 countries have included a reference to coastal and marine ecosystems in terms of climate adaptation and mitigation in their NDCs. The majority of these (45 countries) list commitments related to mangroves, with fewer (10 countries) for seagrass122. Tidal marshes are not specifically mentioned, although many NDCs refer generally to wetlands or commit to using the Intergovernmental Panel on Climate Change (IPCC) Wetlands Supplement134. A few countries, such as the Bahamas and Belize, have ambitious, quantified targets for BCEs in their NDCs, but many countries still lack any measurable targets135.
The IPCC Wetlands Supplement134 provides methodologies and default emission factors such that nations can include, in their NDCs, emissions from a limited range of management activities for conservation and removals through restoration activities. To date, only three countries have reported greenhouse emissions and removals associated with land-use change of coastal wetlands (Australia, the United Arab Emirates and the USA). Market-based methodologies are linked to the underlying IPCC guidance, and focus on generating carbon credits for conservation and restoration of coastal wetlands61,117,132 However, their use is still limited with few pilot projects, mostly within mangroves117. The Livelihoods Carbon Funds are partnering with communities in India (The Sundarbans Mangrove Restoration Project), Indonesia (Yagasu) and Senegal (Océanium) to restore and protect mangrove forests. Sixteen million trees in Sundarbans, 18 million trees in Indonesia and 8,000 ha in Senegal have been planted, with the carbon credits generated being used to help repay the project costs69. Together, these initiatives will sequester a predicted 2.7 million tons of CO2 over 20 years. On a smaller scale, Tahiry Honko (Madagascar) and Mikoko Pamoja (Kenya) have been among the first projects where Plan Vivo has certified blue carbon credits117. The conservation and restoration of more than 1,200 ha of mangrove forest in Tahiry Honko generates more than 1,300 carbon credits per year, where half of the funds generated go to help support the local communities. The Mikoko Pamoja project helps conserve 117 ha and has established 10 ha of mangrove forest117. In 2019, the project sold 1,912 credits with 65% of the revenue going to support community development projects.
Numerous countries can benefit from the inclusion of their coastal ecosystems in NDCs, including small island states with low emissions and small land area relative to the length of coastlines and/or shallow shelf, which could support extensive blue carbon habitats (for example, in Madagascar and Solomon Islands). In larger countries (such as the USA and Canada), there are opportunities to include restoration or habitat creation (including landward retreat with sea-level rise). In terms of climate change mitigation, the main beneficiaries would be those countries with extensive, carbon-rich BCEs that have experienced high rates of loss37. In Indonesia, for example, conversion of mangroves to shrimp ponds accounts for up to 20% of greenhouse gas emissions associated with land-cover change136. However, many nations do not have the necessary data to assess either the extent of BCE or carbon stocks, particularly in the case of tidal marsh and seagrass ecosystems.
Increasing the inclusion of blue carbon within NDCs can be supported in numerous ways (Fig. 6). First, the use of existing61,131 or new frameworks can aid in the identification of blue carbon restoration opportunities at national or subnational scales. In Australia, for example, this process allowed prioritization of leading opportunities that are now being included within the domestic carbon accounting framework61. Resourcing of field data collection and the development of new tools, including spatial data products137,138, are integral to understanding the scope and scale of such opportunities at the national scale. Second, the improvement of standardized protocols for the accounting of greenhouse gas benefits132 and accessibility to low-cost data collection approaches such as remote sensing and affordable field sensors139 will enhance feasibility and uptake of carbon crediting projects. Finally, further demonstration projects are needed, to showcase successes and failures across a broader range of geographic settings, land use and ecosystem types, to build data sets, insights and awareness for future restoration projects116,117.
Other considerations
There have been unsuccessful or poorly planned attempts at long-term restoration, which have utilized non-native or inappropriate species, monoculture without consideration of plant traits and functional diversity, and/or unsuitable sites for planting, including planting on top of other BCEs67,120,140. The capacity of many BCE plant species to repopulate via vegetative growth and/or transportation of propagules and seeds on the tide means that passive revegetation techniques (such as improvements in tidal connectivity and water quality, and/or alterations to hydrodynamic energy) can be preferred in many circumstances61.
Positive steps can be taken to mitigate sea-level rise through restoration projects that include a mix of species and at increased densities and scale, while taking into account local hydrology, the potential for landward expansion of mangroves and tidal marshes, and relevance to the socio-economic factors affecting the local communities109,111,141,142. Anthropogenic warming can also have an impact; for example, increasing temperatures can result in changing gas fluxes in BCEs that lead to enhanced emissions93,118,143. The effect of temperature increase on greenhouse gas emissions in seagrass meadows has indicated the potential negative effects of climate change. An unprecedented heatwave in 2010–2011 led to a 22% loss of seagrass cover in Shark Bay, Australia, resulting in an estimated increase in national CO2 emissions from land-use change by 4–21% per annum93. Experimentally increasing the temperature up to 37 °C led to a fourfold increase in methane fluxes if compared with the community held at 25 °C from Red Sea seagrass sediments143. Temperature increase is likely to lead to a range of different responses in BCEs, including an increase or decrease in primary productivity, faster decomposition of organic matter and changes in BCE spatial distribution, but there are still many uncertainties regarding the nature and extent of these changes and how they might differ with latitude and environmental setting108,118. Conversely, enhanced removals can occur when mangroves expand into tidal marshes or invasive species colonize tidal marsh and seagrass meadows144,145,146,147. Biological invasions of this type will require their own type of management actions based on site-specific conservation strategies104,148.
Summary and future directions
Restoring and managing BCEs for climate change mitigation and adaptation is exciting and feasible28, with potential to draw down an extra 841 Tg CO2e per year. It is complex, however, and must take into account a range of issues including social factors, existing policy, variation in geomorphological settings, human impacts and climate change. For example, monitoring and management of restored areas is needed to ensure their permanence, but many projects fail to consider sea-level rise and/or socio-economic or other human-related impacts in their initial planning117,118,120. These complexities are not insurmountable, but they do require further research and planning, including reconsideration of how international policies can better reward long-term carbon drawdown through BCEs (as opposed to rewarding cheaper, shorter-term abatement). As BCE losses are progressively halted by removing the pressures that led to their losses (for example, one third of seagrass loss in Europe was due to disease, poor water quality and coastal development)85 and protecting BCEs, the goal should shift to restoring BCEs back to their historical extent19.
The development and uptake of blue carbon financing mechanisms have been supported in recent years through national and international initiatives, comprising government and non-government groups122,149. The incorporation of blue carbon within NDCs in the Paris Agreement has seen numerous countries explore domestic mechanisms to promote blue carbon projects and include emissions abatement in their national accounts61,125. Novel approaches such as blended financing (whereby a commercial project provides societal benefits plus financial returns to investors), and/or pairing with co-benefits such as reduction of insurance premiums related to coastal protection146,147, can help enhance uptake and progression to verification in coming years. Importantly, divergent views remain on the application of market-based approaches to quantifying ecosystem service provision to finance environmental protection and restoration, including for coastal wetlands148,150.
Scientific research has multiple roles in facilitating the development of blue carbon accounting frameworks (Fig. 6). Ongoing research of carbon cycling in the coastal zone underpins quantification and accounting for emissions abatement achieved at local through to national scales53,151. However, further research is required to fill substantial gaps108 — geographic biases in the coverage of blue carbon data must be addressed (including imbalances between high, middle and low-income nations); robust information on the scope of small-scale restoration projects at the global or country scale should be developed, including improved estimates of the influence of disturbances and interventions on greenhouse gas fluxes116; and the impacts of climate change and sea-level rise on BCEs and the permanence of carbon sequestration need to be understood109. Future studies should focus on developing new spatial products with similar resolutions, particularly for seagrasses and tidal marshes, and also assess propagation and uncertainty estimates in global blue carbon estimates to constrain the large variability in the estimates provided in our review — thereby providing more meaningful information for policy and other initiatives aiming to implement blue carbon into climate change mitigation strategies. At the project scale, demonstration is required to generate knowledge and data specific to natural, degraded, restored and created wetlands. Demonstration projects will also help identify and ameliorate policy and governance roadblocks, which might include issues of land tenure, project boundaries and rights to carbon152, economic barriers to uptake (such as high transaction costs), demonstrating additionality and issues with double-counting of carbon gains61. Carbon export and storage beyond the boundaries of BCEs, including macroalgae, are potentially an important but unquantified carbon sink14. However, future research needs to overcome scientific and policy barriers that currently preclude the inclusion of carbon export and sequestration beyond BCE extent in blue carbon accounting and abatement associated with management activities.
The success of the blue carbon strategy since it was first put forward in 2009 (ref.6) is reflected in its growing uptake within NDCs and has led to an effort to ascertain additional pathways for climate change mitigation through ocean-based natural climate solutions153. A growing repertoire of options is available, including carbon sequestration from alkalinity release with carbonate dissolution, organic carbon preservation in anoxic conditions within BCEs60,116,154, seaweed farm management155,156 and natural kelp forest conservation14 for carbon sequestration155,156, management of sediments beyond BCEs to avoid emissions and rebuilding populations of large marine animals116. The success of these emerging blue carbon options requires robust scientific evidence along with accountability of their contribution to greenhouse gas mitigation, including meeting the requirements of additionality and permanence of this benefit.
There is an opportunity to further catalyse these actions through synergies between the concurrent UN Decade of Ocean Science (2021–2030) and UN Decade on Ecosystem Restoration (2021–2030)127, as both robust scientific knowledge and more cost-effective technologies to further restoration efforts are needed. The potential for BCEs to offset global emissions (~3% per year, or approximately the combined global emissions from landfill and wastewater sectors)157, while also providing numerous additional ecosystem services for coastal adaptation and local livelihoods, justifies the attention that blue carbon is receiving as a natural climate solution. Our current challenge is to break through financial, scientific governance and social barriers to enable widespread implementation of blue carbon projects.
References
Nesshöver, C. et al. The science, policy and practice of nature-based solutions: an interdisciplinary perspective. Sci. Total Environ. 579, 1215–1227 (2017).
Chausson, A. et al. Mapping the effectiveness of nature-based solutions for climate change adaptation. Glob. Chang. Biol. 26, 6134–6155 (2020).
Pires, J. C. M. Negative emissions technologies: a complementary solution for climate change mitigation. Sci. Total Environ. 672, 502–514 (2019).
McLaren, D. A comparative global assessment of potential negative emissions technologies. Process Saf. Environ. Prot. 90, 489–500 (2012).
Anderson, K. & Peters, G. The trouble with negative emissions. Science 354, 182–183 (2016).
Nellemann, C. et al. Blue Carbon — The Role of Healthy Oceans in Binding Carbon (UN Environment, 2009).
Barbier, E. B. et al. The value of estuarine and coastal ecosystem services. Ecol. Monogr. 81, 169–193 (2011).
Himes-Cornell, A., Grose, S. O. & Pendleton, L. Mangrove ecosystem service values and methodological approaches to valuation: where do we stand? Front. Mar. Sci. 5, 376 (2018).
Friess, D. A. et al. in Oceanography and Marine Biology Vol. 58 Ch. 3 (CRC, 2020).
Lovelock, C. E. & Duarte, C. M. Dimensions of blue carbon and emerging perspectives. Biol. Lett. 15 https://doi.org/10.1098/rsbl.2018.0781 (2019).
Duarte, C. M., Losada, I. J., Hendriks, I. E., Mazarrasa, I. & Marbà, N. The role of coastal plant communities for climate change mitigation and adaptation. Nat. Clim. Chang. 3, 961–968 (2013).
Duarte, C. M., Middelburg, J. J. & Caraco, N. Major role of marine vegetation on the oceanic carbon cycle. Biogeosciences 2, 1–8 (2005).
Mcleod, E. et al. A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front. Ecol. Environ. 9, 552–560 (2011).
Krause-Jensen, D. & Duarte, C. M. Substantial role of macroalgae in marine carbon sequestration. Nat. Geosci. 9, 737–742 (2016).
Macreadie, P. I. et al. Vulnerability of seagrass blue carbon to microbial attack following exposure to warming and oxygen. Sci. Total Environ. 686, 264–275 (2019).
Sippo, J. Z., Lovelock, C. E., Santos, I. R., Sanders, C. J. & Maher, D. T. Mangrove mortality in a changing climate: an overview. Estuar. Coast. Shelf Sci. 215, 241–249 (2018).
Lovelock, C. E. et al. Assessing the risk of carbon dioxide emissions from blue carbon ecosystems. Front. Ecol. Environ. 15, 257–265 (2017).
Zhao, Q. et al. Where marine protected areas would best represent 30% of ocean biodiversity. Biol. Conserv. 244, 108536 (2020).
Duarte, C. M. et al. Rebuilding marine life. Nature 580, 39–51 (2020).
Bayraktarov, E. et al. The cost and feasibility of marine coastal restoration. Ecol. Appl. 26, 1055–1074 (2016).
Van, T. T. et al. Changes in mangrove vegetation area and character in a war and land use change affected region of Vietnam (Mui Ca Mau) over six decades. Acta Oecol. 63, 71–81 (2015).
Dung, L. V., Tue, N. T., Nhuan, M. T. & Omori, K. Carbon storage in a restored mangrove forest in Can Gio Mangrove Forest Park, Mekong Delta, Vietnam. For. Ecol. Manage. 380, 31–40 (2016).
Nam, V. N., Sasmito, S. D., Murdiyarso, D., Purbopuspito, J. & MacKenzie, R. A. Carbon stocks in artificially and naturally regenerated mangrove ecosystems in the Mekong Delta. Wetl. Ecol. Manag. 24, 231–244 (2016).
Reynolds, L. K., Waycott, M., McGlathery, K. J. & Orth, R. J. Ecosystem services returned through seagrass restoration. Restor. Ecol. 24, 583–588 (2016).
Das, S. Ecological restoration and livelihood: contribution of planted mangroves as nursery and habitat for artisanal and commercial fishery. World Dev. 94, 492–502 (2017).
Kiesel, J. et al. Effective design of managed realignment schemes can reduce coastal flood risks. Estuar. Coast. Shelf Sci. 242, 106844 (2020).
McNally, C. G., Uchida, E. & Gold, A. J. The effect of a protected area on the tradeoffs between short-run and long-run benefits from mangrove ecosystems. Proc. Natl Acad. Sci. USA 108, 13945–13950 (2011).
Chow, J. Mangrove management for climate change adaptation and sustainable development in coastal zones. J. Sustain. For. 37, 139–156 (2018).
Dasgupta, S., Islam, M. S., Huq, M., Huque Khan, Z. & Hasib, M. R. Quantifying the protective capacity of mangroves from storm surges in coastal Bangladesh. PLoS ONE 14, e0214079 (2019).
Sutton-Grier, A. E. & Moore, A. Leveraging carbon services of coastal ecosystems for habitat protection and restoration. Coast. Manag. 44, 259–277 (2016).
Owuor, M. A., Mulwa, R., Otieno, P., Icely, J. & Newton, A. Valuing mangrove biodiversity and ecosystem services: a deliberative choice experiment in Mida Creek, Kenya. Ecosyst. Serv. 40, 101040 (2019).
Mcowen, C. J. et al. A global map of saltmarshes. Biodivers. Data J. 5, e11764 (2018).
Bunting, P. et al. The global mangrove watch — a new 2010 global baseline of mangrove extent. Remote Sens. 10, 1669 (2018).
Jayathilake, D. R. M. & Costello, M. J. A modelled global distribution of the seagrass biome. Biol. Conserv. 226, 120–126 (2018).
McKenzie, L. J. et al. The global distribution of seagrass meadows. Environ. Res. Lett. 15, 74041 (2020).
Trumbore, S. E. Potential responses of soil organic carbon to global environmental change. Proc. Natl Acad. Sci. USA 94, 8284–8291 (1997).
Hamilton, S. E. & Friess, D. A. Global carbon stocks and potential emissions due to mangrove deforestation from 2000 to 2012. Nat. Clim. Chang. 8, 240–244 (2018).
Ouyang, X. & Lee, S. Y. Improved estimates on global carbon stock and carbon pools in tidal wetlands. Nat. Commun. 11, 317 (2020).
Kauffman, J. B. et al. Total ecosystem carbon stocks of mangroves across broad global environmental and physical gradients. Ecol. Monogr. 90, e01405 (2020).
Simard, M. et al. Mangrove canopy height globally related to precipitation, temperature and cyclone frequency. Nat. Geosci. 12, 40–45 (2019).
Hutchison, J., Manica, A., Swetnam, R., Balmford, A. & Spalding, M. Predicting global patterns in mangrove forest biomass. Conserv. Lett. 7, 233–240 (2014).
Atwood, T. B. et al. Global patterns in mangrove soil carbon stocks and losses. Nat. Clim. Chang. 7, 523–528 (2017).
Sanderman, J. et al. A global map of mangrove forest soil carbon at 30 m spatial resolution. Environ. Res. Lett. 13, 55002 (2018).
Traganos, D. et al. Towards global-scale seagrass mapping and monitoring using Sentinel-2 on Google Earth Engine: the case study of the Aegean and Ionian Seas. Remote Sens. 10, 1227 (2018).
Hossain, M. S. & Hashim, M. Potential of Earth Observation (EO) technologies for seagrass ecosystem service assessments. Int. J. Appl. Earth Obs. Geoinf. 77, 15–29 (2019).
Atwood, T. B., Witt, A., Mayorga, J., Hammill, E. & Sala, E. Global patterns in marine sediment carbon stocks. Front. Mar. Sci. 7, 165 (2020).
Coastal carbon atlas. Coastal Carbon Research Coordination Network. CCRCN https://ccrcn.shinyapps.io/CoastalCarbonAtlas/_w_8595a9b5/#tab-6425-6 (2019).
UNEP-WCMC. Ocean data viewer: global distribution of seagrasses. UNEP https://doi.org/10.34892/x6r3-d211 (2018).
Hammerstrom, K. K., Kenworthy, W. J., Fonseca, M. S. & Whitfield, P. E. Seed bank, biomass, and productivity of Halophila decipiens, a deep water seagrass on the west Florida continental shelf. Aquat. Bot. 84, 110–120 (2006).
Pergent-Martini, C. et al. Descriptors of Posidonia oceanica meadows: use and application. Ecol. Indic. 5, 213–230 (2005).
Esteban, N., Unsworth, R. K. F., Gourlay, J. B. Q. & Hays, G. C. The discovery of deep-water seagrass meadows in a pristine Indian Ocean wilderness revealed by tracking green turtles. Mar. Pollut. Bull. 134, 99–105 (2018).
York, P. H. et al. Dynamics of a deep-water seagrass population on the Great Barrier Reef: annual occurrence and response to a major dredging program. Sci. Rep. 5, 13167 (2015).
Serrano, O. et al. Australian vegetated coastal ecosystems as global hotspots for climate change mitigation. Nat. Commun. 10, 4313 (2019).
Chmura, G. L., Anisfeld, S. C., Cahoon, D. R. & Lynch, J. C. Global carbon sequestration in tidal, saline wetland soils. Glob. Biogeochem. Cycles 17, 1111 (2003).
Hengl, T. et al. SoilGrids250m: global gridded soil information based on machine learning. PLoS ONE 12, e0169748 (2017).
Rogers, K. et al. Wetland carbon storage controlled by millennial-scale variation in relative sea-level rise. Nature 567, 91–95 (2019).
Rovai, A. S. et al. Global controls on carbon storage in mangrove soils. Nat. Clim. Chang. 8, 534–538 (2018).
Worthington, T. A. et al. A global biophysical typology of mangroves and its relevance for ecosystem structure and deforestation. Sci. Rep. 10, 14652 (2020).
Maher, D. T., Call, M., Santos, I. R. & Sanders, C. J. Beyond burial: lateral exchange is a significant atmospheric carbon sink in mangrove forests. Biol. Lett. 14, 20180200 (2018).
Santos, I. R., Maher, D. T., Larkin, R., Webb, J. R. & Sanders, C. J. Carbon outwelling and outgassing vs. burial in an estuarine tidal creek surrounded by mangrove and saltmarsh wetlands. Limnol. Ocean 64, 996–1013 (2019).
Kelleway, J. J. et al. A national approach to greenhouse gas abatement through blue carbon management. Glob. Environ. Chang. 63, 102083 (2020).
Goldberg, L., Lagomasino, D., Thomas, N. & Fatoyinbo, T. Global declines in human-driven mangrove loss. Glob. Chang. Biol. 68, 5844–5855 (2020).
Richards, D. R. & Friess, D. A. Rates and drivers of mangrove deforestation in Southeast Asia, 2000–2012. Proc. Natl. Acad. Sci. 113, 344–349 (2016).
Thomas, N. et al. Distribution and drivers of global mangrove forest change, 1996–2010. PLoS ONE 12, e0179302 (2017).
Worthington, T. & Spalding, M. Mangrove restoration potential: a global map highlighting a critical opportunity (OECD, 2018).
Kearney, M. S., Riter, J. C. A. & Turner, R. E. Freshwater river diversions for marsh restoration in Louisiana: twenty-six years of changing vegetative cover and marsh area. Geophys. Res. Lett. 38, 16405 (2011).
Lee, S. Y., Hamilton, S., Barbier, E. B., Primavera, J. & Lewis, R. R. Better restoration policies are needed to conserve mangrove ecosystems. Nat. Ecol. Evol. 3, 870–872 (2019).
Lovelock, C. E. & Brown, B. M. Land tenure considerations are key to successful mangrove restoration. Nat. Ecol. Evol. 3, 1135 (2019).
Herr, D., Blum, J., Himes-Cornell, A. & Sutton-Grier, A. An analysis of the potential positive and negative livelihood impacts of coastal carbon offset projects. J. Environ. Manag. 235, 463–479 (2019).
Mojica Vélez, J. M., Barrasa García, S. & Espinoza Tenorio, A. Policies in coastal wetlands: key challenges. Environ. Sci. Policy 88, 72–82 (2018).
Zeng, Y. et al. Economic and social constraints on reforestation for climate mitigation in Southeast Asia. Nat. Clim. Chang. 10, 842–844 (2020).
van Katwijk, M. M. et al. Global analysis of seagrass restoration: the importance of large-scale planting. J. Appl. Ecol. 53, 567–578 (2016).
Waycott, M. et al. Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proc. Natl Acad. Sci. USA 106, 12377–12381 (2009).
Orth, R. J. et al. A global crisis for seagrass ecosystems. Bioscience 56, 987–996 (2006).
Tan, Y. M. et al. Seagrass restoration is possible: insights and lessons from Australia and New Zealand. Front. Mar. Sci. 7, 617 (2020).
Greiner, J. T., McGlathery, K. J., Gunnell, J. & McKee, B. A. Seagrass restoration enhances ‘blue carbon’ sequestration in coastal waters. PLoS ONE 8, e72469 (2013).
Orth, R. J. et al. Restoration of seagrass habitat leads to rapid recovery of coastal ecosystem services. Sci. Adv. 6, eabc6434 (2020).
Cunha, A. H. et al. Changing paradigms in seagrass restoration. Restor. Ecol. 20, 427–430 (2012).
Rezek, R. J., Furman, B. T., Jung, R. P., Hall, M. O. & Bell, S. S. Long-term performance of seagrass restoration projects in Florida, USA. Sci. Rep. 9, 15514 (2019).
Worthington, T. A. et al. Harnessing big data to support the conservation and rehabilitation of mangrove forests globally. One Earth 2, 429–443 (2020).
Kandus, P. et al. Remote sensing of wetlands in South America: status and challenges. Int. J. Remote Sens. 39, 993–1016 (2018).
Gallant, A. L. The challenges of remote monitoring of wetlands. Remote Sens. 7, 10938–10950 (2015).
Unsworth, R. K. F. et al. Sowing the seeds of seagrass recovery using hessian bags. Front. Ecol. Evol. 7, 311 (2019).
Duarte, C. M., Dennison, W. C., Orth, R. J. W. & Carruthers, T. J. B. The charisma of coastal ecosystems: addressing the imbalance. Estuaries Coasts 31, 233–238 (2008).
de los Santos, C. B. et al. Recent trend reversal for declining European seagrass meadows. Nat. Commun. 10, 3356 (2019).
Hamilton, S. E. & Casey, D. Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (CGMFC-21). Glob. Ecol. Biogeogr. 25, 729–738 (2016).
Pendleton, L. et al. Estimating global “blue carbon” emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 7, e43542 (2012).
Deegan, L. A. et al. Coastal eutrophication as a driver of salt marsh loss. Nature 490, 388–392 (2012).
Cardoso, P. G., Raffaelli, D. & Pardal, M. A. The impact of extreme weather events on the seagrass Zostera noltii and related Hydrobia ulvae population. Mar. Pollut. Bull. 56, 483–492 (2008).
Rogers, K. Accommodation space as a framework for assessing the response of mangroves to relative sea-level rise. Singap. J. Trop. Geogr. 42, 163–183 (2021).
Marbà, N. & Duarte, C. M. Mediterranean warming triggers seagrass (Posidonia oceanica) shoot mortality. Glob. Chang. Biol. 16, 2366–2375 (2010).
Lefcheck, J. S., Wilcox, D. J., Murphy, R. R., Marion, S. R. & Orth, R. J. Multiple stressors threaten the imperiled coastal foundation species eelgrass (Zostera marina) in Chesapeake Bay, USA. Glob. Chang. Biol. 23, 3474–3483 (2017).
Arias-Ortiz, A. et al. A marine heatwave drives massive losses from the world’s largest seagrass carbon stocks. Nat. Clim. Chang. 8, 338–344 (2018).
Kendrick, G. A. et al. A systematic review of how multiple stressors from an extreme event drove ecosystem-wide loss of resilience in an iconic seagrass community. Front. Mar. Sci. 6, 455 (2019).
Duke, N. C. et al. Large-scale dieback of mangroves in Australia’s Gulf of Carpentaria: a severe ecosystem response, coincidental with an unusually extreme weather event. Mar. Freshw. Res. 68, 1816–1829 (2017).
Taillie, P. J. et al. Widespread mangrove damage resulting from the 2017 Atlantic mega hurricane season. Environ. Res. Lett. 15, 64010 (2020).
Asbridge, E., Lucas, R., Rogers, K. & Accad, A. The extent of mangrove change and potential for recovery following severe Tropical Cyclone Yasi, Hinchinbrook Island, Queensland, Australia. Ecol. Evol. 8, 10416–10434 (2018).
Hickey, S. M. et al. Is climate change shifting the poleward limit of mangroves? Estuaries Coasts 40, 1215–1226 (2017).
Saintilan, N., Wilson, N. C., Rogers, K., Rajkaran, A. & Krauss, K. W. Mangrove expansion and salt marsh decline at mangrove poleward limits. Glob. Chang. Biol. 20, 147–157 (2014).
Whitt, A. A. et al. March of the mangroves: drivers of encroachment into southern temperate saltmarsh. Estuar. Coast. Shelf Sci. 240, 106776 (2020).
Cavanaugh, K. C. et al. Sensitivity of mangrove range limits to climate variability. Glob. Ecol. Biogeogr. 27, 925–935 (2018).
Cavanaugh, K. C. et al. Poleward expansion of mangroves is a threshold response to decreased frequency of extreme cold events. Proc. Natl Acad. Sci. USA 111, 723–727 (2014).
Coldren, G. A., Langley, J. A., Feller, I. C. & Chapman, S. K. Warming accelerates mangrove expansion and surface elevation gain in a subtropical wetland. J. Ecol. 107, 79–90 (2019).
Yando, E. S. et al. Salt marsh–mangrove ecotones: using structural gradients to investigate the effects of woody plant encroachment on plant–soil interactions and ecosystem carbon pools. J. Ecol. 104, 1020–1031 (2016).
Doughty, C. L. et al. Mangrove range expansion rapidly increases coastal wetland carbon storage. Estuaries Coasts 39, 385–396 (2016).
Lovelock, C. E. et al. Sea level and turbidity controls on mangrove soil surface elevation change. Estuar. Coast. Shelf Sci. 153, 1–9 (2015).
Woodroffe, C. D. et al. Mangrove sedimentation and response to relative sea-level rise. Ann. Rev. Mar. Sci. 8, 243–266 (2016).
Lovelock, C. E. & Reef, R. Variable impacts of climate change on blue carbon. One Earth 3, 195–211 (2020).
Saintilan, N. et al. Thresholds of mangrove survival under rapid sea level rise. Science 368, 1118–1121 (2020).
Nicholls, R. J. Coastal flooding and wetland loss in the 21st century: changes under the SRES climate and socio-economic scenarios. Glob. Environ. Chang. 14, 69–86 (2004).
Schuerch, M. et al. Future response of global coastal wetlands to sea-level rise. Nature 561, 231–234 (2018).
Adame, M. F. et al. Future carbon emissions from global mangrove forest loss. Glob. Chang. Biol. 27, 2856–2866 (2021).
Griscom, B. W. et al. Natural climate solutions. Proc. Natl Acad. Sci. USA 114, 11645–11650 (2017).
Friedlingstein, P. et al. Global Carbon Budget 2020. Earth Syst. Sci. Data 12, 3269–3340 (2020).
Morris, R. L., Boxshall, A. & Swearer, S. E. Climate-resilient coasts require diverse defence solutions. Nat. Clim. Chang. 10, 485–487 (2020).
Macreadie, P. I. et al. The future of blue carbon science. Nat. Commun. 10, 3998 (2019).
Wylie, L., Sutton-Grier, A. E. & Moore, A. Keys to successful blue carbon projects: lessons learned from global case studies. Mar. Policy 65, 76–84 (2016).
Howard, J. F. et al. Clarifying the role of coastal and marine systems in climate mitigation. Front. Ecol. Environ. 15, 42–50 (2017).
Lenihan, H. S. & Peterson, C. H. How habitat degradation through fishery disturbance enhances impacts of hypoxia on oysters reefs. Ecol. Appl. 8, 128–140 (1998).
Ellison, A. M., Felson, A. J. & Friess, D. A. Mangrove rehabilitation and restoration as experimental adaptive management. Front. Mar. Sci. 7, 327 (2020).
Lester, S. E., Dubel, A. K., Hernan, G., McHenry, J. & Rassweiler, A. Spatial planning principles for marine ecosystem restoration. Front. Mar. Sci. 7, 328 (2020).
Herr, D. & Landis, E. Coastal blue carbon ecosystems: opportunities for nationally determined contributions. Policy brief (IUCN, 2016).
Apple Newsroom. Conserving mangroves, a lifeline for the world. Apple (22 April 2019) https://www.apple.com/newsroom/2019/04/conserving-mangroves-a-lifeline-for-the-world
Hochard, J. P., Hamilton, S. & Barbier, E. B. Mangroves shelter coastal economic activity from cyclones. Proc. Natl Acad. Sci. USA 116, 12232–12237 (2019).
Herr, D., von Unger, M., Laffoley, D. & McGivern, A. Pathways for implementation of blue carbon initiatives. Aquat. Conserv. Mar. Freshw. Ecosyst. 27, 116–129 (2017).
Friess, D. A. et al. in Sustainable Development Goals: Their Impacts on Forests and People Ch. 14 (eds Katila, P. et al.) 445–481 (Cambridge Univ. Press, 2019).
Waltham, N. J. et al. UN Decade on Ecosystem Restoration 2021–2030 — what chance for success in restoring coastal ecosystems? Front. Mar. Sci. 7, 71 (2020).
Convention on Biological Diversity. Conference of the Parties Decision X/2: strategic plan for biodiversity 2011–2020. CBD https://www.cbd.int/decision/cop/?id=12268 (2011).
United Nations. Transforming our world: the 2030 Agenda for Sustainable Development (UN, 2015).
Brander, L. M. et al. The global costs and benefits of expanding marine protected areas. Mar. Policy 116, 103953 (2020).
Howard, J. F. et al. The potential to integrate blue carbon into MPA design and management. Aquat. Conserv. 27, 100–115 (2017).
Needelman, B. A. et al. The science and policy of the Verified Carbon Standard methodology for tidal wetland and seagrass restoration. Estuaries Coasts 41, 2159–2171 (2018).
Michaelowa, A., Hermwille, L., Obergassel, W. & Butzengeiger, S. Additionality revisited: guarding the integrity of market mechanisms under the Paris Agreement. Clim. Policy 19, 1211–1224 (2019).
Intergovernmental Panel on Climate Change. 2013 Supplement to the 2006 IPCC guidelines for national greenhouse gas inventories: wetlands (IPCC, 2014).
United Nations Environment Programme. Out of the blue: the value of seagrasses to the environment and to people (UNEP, 2020).
Murdiyarso, D. et al. The potential of Indonesian mangrove forests for global climate change mitigation. Nat. Clim. Chang. 5, 1089–1092 (2015).
Jones, T. et al. Madagascar’s mangroves: quantifying nation-wide and ecosystem specific dynamics, and detailed contemporary mapping of distinct ecosystems. Remote Sens. 8, 106 (2016).
Holmquist, J. R. et al. Uncertainty in United States coastal wetland greenhouse gas inventorying. Environ. Res. Lett. 13, 115005 (2018).
Maher, D. T., Drexl, M., Tait, D. R., Johnston, S. G. & Jeffrey, L. C. iAMES: an inexpensive, automated methane ebullition sensor. Environ. Sci. Technol. 53, 6420–6426 (2019).
Primavera, J. H. & Esteban, J. M. A. A review of mangrove rehabilitation in the Philippines: successes, failures and future prospects. Wetl. Ecol. Manag. 16, 345–358 (2008).
Silliman, B. R. et al. Facilitation shifts paradigms and can amplify coastal restoration efforts. Proc. Natl Acad. Sci. USA 112, 14295–14300 (2015).
Enwright, N. M., Griffith, K. T. & Osland, M. J. Barriers to and opportunities for landward migration of coastal wetlands with sea-level rise. Front. Ecol. Environ. 14, 307–316 (2016).
Burkholz, C., Garcias-Bonet, N. & Duarte, C. M. Warming enhances carbon dioxide and methane fluxes from Red Sea seagrass (Halophila stipulacea) sediments. Biogeosciences 17, 1717–1730 (2020).
Bianchi, T. S. et al. Historical reconstruction of mangrove expansion in the Gulf of Mexico: linking climate change with carbon sequestration in coastal wetlands. Estuar. Coast. Shelf Sci. 119, 7–16 (2013).
Apostolaki, E. T. et al. Exotic Halophila stipulacea is an introduced carbon sink for the eastern Mediterranean Sea. Sci. Rep. 9, 9643 (2019).
Bell, J. & Lovelock, C. E. Insuring mangrove forests for their role in mitigating coastal erosion and storm-surge: an Australian case study. Wetlands 33, 279–289 (2013).
Reguero, B. G. et al. Financing coastal resilience by combining nature-based risk reduction with insurance. Ecol. Econ. 169, 106487 (2020).
Thomas, S. Blue carbon: knowledge gaps, critical issues, and novel approaches. Ecol. Econ. 107, 22–38 (2014).
International Partnership for Blue Carbon. Blue carbon partnership. IPBC https://bluecarbonpartnership.org (2017).
Boon, P. I. & Prahalad, V. Ecologists, economics and politics: problems and contradictions in applying neoliberal ideology to nature conservation in Australia. Pac. Conserv. Biol. 23, 115–132 (2017).
Adame, M. F. et al. The undervalued contribution of mangrove protection in Mexico to carbon emission targets. Conserv. Lett. 11, e12445 (2018).
Bell-James, J. & Lovelock, C. E. Legal barriers and enablers for reintroducing tides: an Australian case study in reconverting ponded pasture for climate change mitigation. Land Use Policy 88, 104192 (2019).
Gattuso, J.-P. et al. Ocean solutions to address climate change and its effects on marine ecosystems. Front. Mar. Sci. 5, 337 (2018).
Saderne, V. et al. Role of carbonate burial in blue carbon budgets. Nat. Commun. 10, 1106 (2019).
Duarte, C. M., Wu, J., Xiao, X., Bruhn, A. & Krause-Jensen, D. Can seaweed farming play a role in climate change mitigation and adaptation? Front. Mar. Sci. 4, 100 (2017).
Froehlich, H. E., Afflerbach, J. C., Frazier, M. & Halpern, B. S. Blue growth potential to mitigate climate change through seaweed offsetting. Curr. Biol. 29, 3087–3093.e3 (2019).
Ritchie, H. & Roser, M. CO2 and greenhouse gas emissions. Our World in Data https://ourworldindata.org/co2-and-other-greenhouse-gas-emissions (2017).
Smith, S. V. Marine macrophytes as a global carbon sink. Science 211, 838–840 (1981).
Intergovernmental Panel on Climate Change. Special report on the ocean and cryosphere in a changing climate (IPCC, 2019).
Verified Carbon Standard. VM0007 REDD+ methodology framework (REDD+MF) (VCS, 2020).
Carnell, P. E. et al. Mapping ocean wealth Australia: the value of coastal wetlands to people and nature. The Nature Conservancy https://doi.org/10.21153/carnell2019mapping (2019).
Jänes, H. et al. Stable isotopes infer the value of Australia’s coastal vegetated ecosystems from fisheries. Fish Fish. 21, 80–90 (2020).
Jänes, H. et al. Quantifying fisheries enhancement from coastal vegetated ecosystems. Ecosyst. Serv. 43, 101105 (2020).
Huang, B. et al. Quantifying welfare gains of coastal and estuarine ecosystem rehabilitation for recreational fisheries. Sci. Total Environ. 710, 134680 (2020).
Acknowledgements
The authors acknowledge funding by Deakin University (to P.I.M. and M.D.P.C.), Qantas (to P.I.M. and M.D.P.C.), HSBC (to P.I.M. and M.D.P.C.), Australian Research Council Discovery Grants (DP200100575; to P.I.M. and C.M.D.), King Abdullah University of Science and Technology under KAUST’s Circular Carbon Economy Initiative (to C.M.D.) and the Early Career Research Fellowship from the Gulf Research Program of the National Academies of Sciences, Engineering, and Medicine (to T.B.A.; the content is solely the responsibility of the authors and does not necessarily represent the official views of the Gulf Research Program of the National Academies of Sciences, Engineering, and Medicine). They also thank N. Yilmaz who helped with creation of the figures.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Earth & Environment thanks R. Bhomia and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Macreadie, P.I., Costa, M.D.P., Atwood, T.B. et al. Blue carbon as a natural climate solution. Nat Rev Earth Environ 2, 826–839 (2021). https://doi.org/10.1038/s43017-021-00224-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43017-021-00224-1
This article is cited by
-
A global meta-analysis on the drivers of salt marsh planting success and implications for ecosystem services
Nature Communications (2024)
-
Good fisheries management is good carbon management
npj Ocean Sustainability (2024)
-
Substantial kelp detritus exported beyond the continental shelf by dense shelf water transport
Scientific Reports (2024)
-
Methane oxidation minimizes emissions and offsets to carbon burial in mangroves
Nature Climate Change (2024)
-
Assessing Tidal Hydrodynamics in a Tropical Seascape Using Structure‐from‐Motion Photogrammetry and 2D Flow Modelling
Estuaries and Coasts (2024)