Abstract
Study design:
Retrospective cross-sectional study.
Objectives:
To investigate the characteristics of posttraumatic symptomatic syringomyelia after spinal cord injury (SCI).
Setting:
Swiss Paraplegic Centre, Nottwil, Switzerland.
Methods:
The patient database was screened for patients diagnosed with posttraumatic syringomyelia. Syrinx characteristics were determined on T2-weighted magnetic resonance images. Binary logistic regression analysis was used to investigate the effects of age, injury level, injury severity and syrinx location on early syrinx formation, syrinx length and syrinx extending cranial to the lesion.
Results:
The data of 138 patients were analyzed. The majority of the patients (78.3%) suffered from motor and sensory complete SCI (American Spinal Injury Association Impairment Scale (AIS) A). Syringomyelia was diagnosed a median 15.0 years after SCI at a median age of 42 years. The cervical spine was involved in >57% of the patients, and syringomyelia extended over a median seven vertebral levels. Complete SCI (P=0.035) and age (P=0.001) were significant predictors of early syrinx formation. Syringomyelia occurred significantly earlier in older (>30 years) patients (P⩽0.002) and those with complete SCI (P=0.027) compared with younger patients (⩽30 years) and those with incomplete SCI (AIS B–D), respectively. Age, injury level, injury severity (AIS A) and syrinx location did not have any significant (P>0.9) effect on syrinx extending cranially or syrinx length.
Conclusions:
Posttraumatic syringomyelia mainly occurs in patients with complete SCI (AIS A) and involves the cervical spine in 6 of the 10 patients. Patients with complete SCI and those age >30 years have an increased risk of syrinx formation within 5 years after injury.
Similar content being viewed by others
Introduction
Posttraumatic syringomyelia can cause neurological deterioration after spinal cord injury (SCI) and thus represents a potentially serious complication. Sensory disturbance, gradual loss of motor function, pain at or above the level of injury and increased spasticity are the characteristic and most common clinical signs.1, 2, 3, 4, 5, 6, 7 The reported incidences of posttraumatic syringomyelia vary greatly. The incidence of posttraumatic syringomyelia with clinical manifestation ranges from <1% to 7%.2, 3, 4, 5, 8, 9 However, the incidence in radiological investigations is considerably higher and ranges >50%.6, 10, 11 Clinically relevant posttraumatic syringomyelia does not occur frequently; nevertheless, progressive neurological deterioration in the wake of posttraumatic syringomyelia represents a serious complication with devastating consequences for the affected patients.
Posttraumatic syringomyelia has been observed more frequently in individuals with motor and sensory complete SCI compared with those with incomplete SCI.1, 2, 8 Furthermore, it has been reported that the onset of posttraumatic syringomyelia is earlier with older age at the time of SCI and with cervical and thoracic SCI compared with lumbar SCI.8 However, there is a lack of data concerning the relevant characteristics and risk factors of posttraumatic syringomyelia. On the basis of risk factors, screening procedures for the timely diagnosis of posttraumatic syringomyelia could be defined.
We have therefore investigated the characteristics of symptomatic posttraumatic syringomyelia in individuals with SCI and the effects of patient and syrinx characteristics on unfavorable syringomyelia presentation (early formation, cranial progression and elongation). The following hypotheses were tested: age at the time of SCI, injury level (that is, tetraplegic or paraplegic), injury severity (that is, motor and sensory complete or incomplete SCI), and syrinx location (that is, cervical or non-cervical) have a significant effect on early syrinx formation (that is, <5 years after SCI), syrinx length and syrinx extending cranial to the lesion.
Materials and methods
Setting
This investigation was carried out in a SCI rehabilitation center with 140 beds. All applicable institutional and governmental regulations concerning the ethical use of health-related data were followed during the course of this research.
Collected data
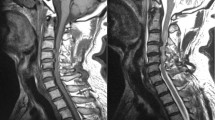
A 10-year time period (2002–2012) was evaluated. Individuals to be investigated were identified by searching our patient database with the terms ‘syringomyelia’, ‘syrinx’ and ‘hydromyelia’. Individuals with an etiology other than traumatic SCI and no magnetic resonance imaging of the affected area were excluded. Personal characteristics were collected from electronic patient records. The severity of SCI was classified using the American Spinal Injury Association Impairment Scale (AIS). The syrinx location and length were determined on T2-weighted magnetic resonance images of the spine in neutral position using the Phoenix PACS software (version 3.20.34233, Phoenix-PACS GmbH, Freiburg, Germany). Furthermore, the date of the diagnosis was collected.
Statistical analysis
The data were calculated as medians and 95% confidence intervals (CIs) if appropriate. The Chi-square or Fisher’s exact test was used to investigate the differences in observed frequencies between groups. Comparisons between independent samples were performed using Mann–Whitney U-test. The effects of age at the time of SCI, injury level (that is, tetraplegic or paraplegic), injury severity (that is, AIS A or AIS B–D) and syrinx location (that is, cervical or non-cervical) on early syrinx formation (that is, <5 years after SCI), syrinx length and syrinx extending cranial to the lesion were investigated using binary logistic regression analysis. The age was grouped using 15-year increments.12 The statistical analyses were performed using the SPSS software (version 18.0.3, IBM, Somers, NY, USA). A P-value of ⩽0.05 was considered significant.
Results
A total of 222 patients with symptomatic syringomyelia were identified. The data of 32 patients with non-traumatic SCI and 52 patients with myelomeningocele were excluded from analysis. Thus, the data of 138 patients were analyzed.
Patients’ characteristics, including gender, injury location and injury severity, are presented in Table 1. The majority of the patients (that is, 110/138, 79.7%) were male, and most patients (that is, 108/138, 78.3%) suffered from motor and sensory complete SCI (AIS A). The median age at the time of SCI was 25 years (95% CI 23–27 years, range 12–63 years). The symptoms associated with syringomyelia are presented in Table 2. Deterioration of motor and sensory function, pain and increased spasticity were the most commonly observed symptoms.
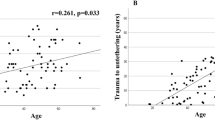
Syringomyelia was diagnosed a median 15.0 years (95% CI 10.6–16.7 years, range 0–40 years) after SCI had occurred at a median age of 42 years (95% CI 40–44 years, range 17–79 years). Syringomyelia was most commonly observed in the cervical–thoracic (that is, 58/138, 42.0%) or the thoracic spine (that is, 50/138, 36.2%). A total of 19 (13.8%) and 5 (3.6%) syrinx occurred in the cervical and thoraco-lumbar spine, respectively. In two patients, the syrinx stretched from the cervical to the lumbar spine. The median syrinx length was seven vertebral levels (95% CI 6–8 levels, range 1–19 levels). The direction of extension was cranial in 18 (14.4%) patients, caudal in 36 (28.8%) and both in 71 (56.8%) patients. The syrinxes were extending a median three (95% CI 3–4 levels, range 1–19 levels) and eight vertebral levels (95% CI 6–9 levels, range 2–18 levels) in the cranial and caudal direction, respectively.
Injury severity (P=0.035) and age (P=0.001) were significant predictors for the early (that is, <5 years after SCI) formation of syringomyelia (Table 3). The odds of early syrinx formation were increased more than nine times in patients with motor and sensory complete SCI (AIS A) compared with those with incomplete SCI (AIS B–D). Furthermore, the odds of early syrinx formation increased with age (Table 3). In patients aged >45 years, 53% experienced syrinx formation in <5 years after SCI compared with 15% in patients who were aged ⩽30 years (Table 4). Syringomyelia occurred significantly (P⩽0.002) earlier in older (>30 years) compared with younger patients (⩽30 years) (Table 5). In patients with motor and sensory complete SCI (AIS A), syringomyelia occurred significantly sooner (P=0.027) (Table 5) and more often earlier than 5 years after SCI (P=0.003) compared with patients with incomplete SCI (AIS B–D) (Table 4).
Age, injury level, injury severity and syrinx location did not have any significant (P>0.9) effect on cranial syrinx progression or syrinx length (Table 3).
Discussion
To our knowledge, this is the first investigation of possible predictors of early syrinx formation, cranial syrinx progression and syrinx length. Injury severity was a significant predictor of early syrinx formation. The odds of early syrinx formation were increased more than nine times in patients with motor and sensory complete SCI compared with those with incomplete SCI. A trauma resulting in complete SCI may cause a greater impairment of the cerebrospinal fluid circulation compared with one resulting in incomplete SCI and thus facilitate the development of syringomyelia.13 Other investigators have also observed earlier syrinx formation in patients with complete SCI.8 However, statistical significance was not reached, most likely because of insufficient sample size and statistical power. There is a lack of data concerning the effect of injury severity on the time course of syringomyelia after traumatic SCI. Previous reports contain data concerning the incidence and occurrence of syringomyelia in patients with complete and incomplete SCI. The majority of reports support the hypothesis that syringomyelia is more common in patients with complete SCI.1, 3, 4, 8, 14 In the present study, approximately 75% of the patients with syringomyelia suffered from complete SCI compared with 52–82% in these previous reports. Few authors have reported similar or lower (that is, 50–33%) proportions of patients with complete compared with those with incomplete SCI among patients suffering from syringomyelia.2, 9, 10 In contrast to present and other results, Ko et al.2 have reported that only 49% of patients with early syringomyelia suffered from complete SCI.
Age at the time of SCI was the second significant predictor of early syrinx formation. Syringomyelia occurred a median 3.5 and 5 years after SCI in patients aged >45 and 31–45 years, respectively, compared with 17 years in patients aged ⩽30 years. Other authors have also reported a significantly earlier onset of syringomyelia with increasing age at the time of SCI.8 The mean time after SCI was approximately 6, 10 and 13 years in patients aged >40 years, 31–40 years and up to 30 years, respectively, in this previous report.8 In individuals aged >30 years, age-related degenerative changes of the spinal canal and cord tissue affecting cerebrospinal fluid circulation seem to accelerate the development of posttraumatic syringomyelia.8
In the present study, the level of SCI (that is, tetraplegia vs paraplegia) did not have any significant effect on early syrinx formation, syrinx length or cranial syrinx progression. This is in accordance with other investigations, in which there was no association between the injury level and the time to syrinx formation.2, 4, 8 In contrast, some authors have observed a higher percentage of paraplegics among patients affected by syringomyelia.9, 10 In the present study, the time from SCI to syrinx formation was approximately 16 years in tetraplegic patients, compared with 7–9 years in previous reports.4, 8 The elapsed time after SCI in paraplegic patients was approximately 14 and 5–11 years in the present and previous investigations,4, 8 respectively.
In the present study, none of the investigated factors was a significant predictor of cranial syrinx progression or syrinx length. An earlier report corroborates that the direction of syrinx progression does not depend on the severity of SCI.7 Factors associated with the mechanism of syringomyelia formation in the spinal cord may affect syrinx length and cranial progression.13
In our patients, syringomyelia had been diagnosed a median 15 years after SCI at a median age of 42 years. Interestingly, there is a broad agreement concerning the occurrence of syringomyelia in patients in their forties1, 2, 7, 10 and thus between one and one-and-a-half decades after SCI.1, 8, 9, 15 Syringomyelia was most commonly observed in the cervical–thoracic or the thoracic spine (that is, 78%) and stretched over a median seven vertebral levels. The reported average extent of syringomyelia ranges from 7 to 12 vertebral levels.1, 10 In the present investigation, syringomyelia had progressed cranially, caudally and in both directions in 14, 29 and 57% of the patients, respectively. The main difference to previous data7, 10 is the greater percentage of progression in both direction (57% vs 33%) and the smaller percentage of cranial progression (14% vs 47%) in our study cohort. The extent of progression was a median 3 and 8 vertebral levels in the cranial and caudal direction, respectively, compared with 4.5 and 2 levels reported previously.10
The retrospective and cross-sectional study design pertains to the limitations of the present study. The odds ratio values should be considered with caution because of the wide CIs. Possible predictors of syringomyelia such as spinal canal stenosis,1, 2, 10 posttraumatic kyphosis of the vertebral column1 or pathophysiological changes in the spinal cord13, 16 have not been investigated. However, the aim of the present study was to investigate readily available and easily assessable predictors of syringomyelia. Further prospective longitudinal studies are needed to confirm the present results and to investigate other possible predictors of syringomyelia.
Progressive neurological deterioration in the wake of posttraumatic syringomyelia represents a serious complication with devastating consequences for the affected patients. Health professionals entrusted with the care of SCI individuals should be aware of the possibility of syringomyelia in motor and sensory complete SCI patients in their fortie s, approximately 10 years after injury, presenting with symptoms such as sensory and motor function loss, pain or increased spasticity. Individuals with motor and sensory complete SCI aged >30 years are at risk of early syrinx formation and should be screened for syringomyelia (annual magnetic resonance imaging). The extent and frequency of screening procedures should be investigated in future studies.
In conclusion, syringomyelia mainly occurs in patients with motor and sensory complete SCI and involves the cervical spine in three of five patients. Patients with motor and sensory complete SCI and those aged >30 years have an increased risk of syrinx formation within 5 years after injury.
Data archiving
There were no data to deposit.
References
Karam Y, Hitchon PW, Mhanna NE, He W, Noeller J . Post-traumatic syringomyelia: outcome predictors. Clin Neurol Neurosurg 2014; 124: 44–50.
Ko HY, Kim W, Kim SY, Shin MJ, Cha YS, Chang JH et al. Factors associated with early onset post-traumatic syringomyelia. Spinal Cord 2012; 50: 695–698.
Schurch B, Wichmann W, Rossier AB . Post-traumatic syringomyelia (cystic myelopathy): a prospective study of 449 patients with spinal cord injury. J Neurol Neurosurg Psychiatry 1996; 60: 61–67.
El Masry WS, Biyani A . Incidence, management, and outcome of post-traumatic syringomyelia. In memory of Mr Bernard Williams. J Neurol Neurosurg Psychiatry 1996; 60: 141–146.
Rossier AB, Foo D, Shillito J, Dyro FM . Posttraumatic cervical syringomyelia. Incidence, clinical presentation, electrophysiological studies, syrinx protein and results of conservative and operative treatment. Brain 1985; 108: 439–461.
Backe HA, Betz RR, Mesgarzadeh M, Beck T, Clancy M . Post-traumatic spinal cord cysts evaluated by magnetic resonance imaging. Paraplegia 1991; 29: 607–612.
Klekamp J . Treatment of posttraumatic syringomyelia. J Neurosurg Spine 2012; 17: 199–211.
Vannemreddy SS, Rowed DW, Bharatwal N . Posttraumatic syringomyelia: predisposing factors. Br J Neurosurg 2002; 16: 276–283.
Carroll AM, Brackenridge P . Post-traumatic syringomyelia: a review of the cases presenting in a regional spinal injuries unit in the north east of England over a 5-year period. Spine (Phila Pa 1976) 2005; 30: 1206–1210.
Perrouin-Verbe B, Lenne-Aurier K, Robert R, Auffray-Calvier E, Richard I, Mauduyt de la Greve I et al. Post-traumatic syringomyelia and post-traumatic spinal canal stenosis: a direct relationship: review of 75 patients with a spinal cord injury. Spinal Cord 1998; 36: 137–143.
Silberstein M, Hennessy O . Cystic cord lesions and neurological deterioration in spinal cord injury: operative considerations based on magnetic resonance imaging. Paraplegia 1992; 30: 661–668.
DeVivo MJ, Biering-Sorensen F, New P, Chen Y . Standardization of data analysis and reporting of results from the International Spinal Cord Injury Core Data Set. Spinal Cord 2011; 49: 596–599.
Greitz D . Unraveling the riddle of syringomyelia. Neurosurg Rev 2006; 29: 251–263 (Discussion 264).
Edgar R, Quail P . Progressive post-traumatic cystic and non-cystic myelopathy. Br J Neurosurg 1994; 8: 7–22.
Bonfield CM, Levi AD, Arnold PM, Okonkwo DO . Surgical management of post-traumatic syringomyelia. Spine 2010; 35: S245–S258.
Shields CB, Zhang YP, Shields LB . Post-traumatic syringomyelia: CSF hydrodynamic changes following spinal cord injury are the driving force in the development of PTSM. Handb Clin Neurol 2012; 109: 355–367.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Krebs, J., Koch, H., Hartmann, K. et al. The characteristics of posttraumatic syringomyelia. Spinal Cord 54, 463–466 (2016). https://doi.org/10.1038/sc.2015.218
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2015.218
This article is cited by
-
Spinal cord tethering and syringomyelia after trauma: impact of age and surgical outcome
Scientific Reports (2023)
-
Spinal cord untethering and midline myelotomy for delayed, symptomatic post-traumatic syringomyelia due to retained ballistic fragments: case report
Spinal Cord Series and Cases (2022)
-
Factors affecting long-term mortality rate after diagnosis of syringomyelia in disabled spinal cord injury patients: a population-based study
Spinal Cord (2020)
-
Neurosurgical untethering with or without syrinx drainage results in high patient satisfaction and favorable clinical outcome in post-traumatic myelopathy patients
Spinal Cord (2018)
-
Posttraumatische Syringomyelie
Trauma und Berufskrankheit (2018)