Key Points
-
Highlights the importance of establishing drainage for patients attending with an acute dentoalveolar abscess
-
Challenges the need to 'complete the full course' of antibiotics, classically 5-7 days, when a 3 day course of antibiotics has been shown to be efficacious.
-
Emphasises to the GDP the impact of over-prescribing antibiotics on global antibiotic resistance and the need for change in prescribing habits.
Abstract
Objective An audit to ascertain the effectiveness of drainage combined with a three day standard dose antimicrobial regime for patients with acute dentoalveolar abscess and associated systemic symptoms.
Method Patients attending the Primary Care Department at Bristol Dental Hospital with an acute dentoalveolar abscess associated with systemic involvement underwent drainage and removal of the cause of their infection, followed by a three day course of antibiotics. The antibiotic issued was of standard dosage and the choice of antibiotic prescribed varied depending on the type of infection present. The patients were followed up by either telephone or clinical review.
Results From a sample size of 188 patients, an overall review was obtained for 80.3% of patients. When departmental guidelines were followed all reviewed patients achieved a successful outcome. An overall antibiotic prescribing rate of 2.9% was achieved for adult patients attending the emergency department in pain.
Conclusion Following drainage and removal of the cause of infection, a three day standard dose antibiotic regime was effective in the management of the acute dentoalveolar abscess in all reviewed patients showing associated signs of systemic symptoms.
Similar content being viewed by others
Introduction
Awareness of international concerns relating to the appropriateness and overprescribing of antibiotics in the dental setting led to a rigorous review of antibiotic prescribing for the management of the acute dentoalveolar abscess at Bristol Dental Hospital in 2004/5. Following a literature search of MEDLINE, EMBASE and the COCHRANE library (using the search criteria 'antibiotic' and 'dental'), minimal evidence-based usage of antibiotic prescribing was found for the management of this group of patients.1
Considering best practice, available evidence and a thorough understanding of current empirical treatment regimes, prescribing guidelines for the management of the patient with an acute dentoalveolar abscess were drawn up for use in the primary care department at the University of Bristol Dental Hospital and School.1,2,3,4
This study set out to investigate the effectiveness of a three day course of standard dose antibiotics in the management of patients with systemic symptoms related to their dentoalveolar abscess following effective drainage.
Method
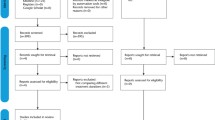
Prescribing guidelines, drawn up for the Primary Care Department at the University of Bristol Dental Hospital and School, for patients with acute dentoalveolar infections and associated signs of systemic involvement were agreed within the Division on the basis of best practice and available evidence. The aim was to produce evidence-based prescribing guidelines for this group of patients (Fig. 1).1 These guidelines were implemented from 1 July 2005 and the patient outcome was audited retrospectively.
The majority of patients presenting with a dentoalveolar abscess have localised swelling which can be managed by local drainage methods. A smaller number of patients present showing signs of spreading infection or with a systemic response to their infection.
For the purposes of this audit, clinical signs of pyrexia (aural temperature >36.8°C taken on the contralateral side), trismus, significant regional lymphadenopathy, gross facial swelling, closure of the eye, dysphagia, tachycardia (pulse rate >100 beats per minute) and rigors were regarded as indicators of systemic response to infection.
All adult patients found to exhibit signs of systemic involvement underwent drainage followed by removal of the cause of their infection. They were then prescribed a three day course of standard dose antibiotics as shown in Figure 1 and followed up either clinically or by telephone on completion of their antibiotic course. All patients were advised that if their symptoms had not resolved or had worsened, they should re-attend the department.
The decision to review patients by telephone was taken as a large 'failure to attend' for review was anticipated if all patients were given a formal follow-up appointment. However, for patients showing the most severe signs of systemic involvement, who were on the borderline for admission but managed as an outpatient, or for those patients who were immunocompromised, a formal follow-up appointment was made, allowing an opportunity to correlate clinical findings with the patient's perception of clinical improvement.
Immunocompromised patients included those with unstable diabetes mellitus, patients on immunosuppressant therapy, those undergoing chemotherapy or patients who had had previous radiotherapy to the head and neck.
Results
Over a 24 month period (1 July 2005 to 30 June 2007) 6,586 adult patients attended the Primary Care Department in pain. One hundred and eighty-eight patients showed signs of systemic involvement associated with a dentoalveolar abscess and were prescribed antibiotics in accordance with Figure 1 following drainage and removal of the source of infection. This resulted in an overall antiobiotic prescribing rate of 2.9%, contrasting very favourably with other studies showing much higher rates (23-74%) of antimicrobial prescribing for emergency patients.4,5,6,7
In total 22 patients were reviewed clinically; all had resolution of their systemic symptoms and the verbal:clinical correlation was 100%.
Despite repeated attempts at contact, only 129 out of the remaining 166 patients were reviewed by telephone, giving a response rate of 77.7%. Combined with those patients seen clinically a review rate of 80.3% was achieved. Overall, seven patients failed to achieve resolution of their systemic symptoms following their three day antibiotic course, giving a success rate of 95.3%. The clinical notes for these patients were re-examined in an attempt to explain the apparent failure. These records revealed that there was failure to achieve drainage in four patients and two patients failed to wait for their drainage/extractions to be carried out. The final patient re-attended after his telephone review when he was diagnosed as having a dry socket rather than an on-going infection. The socket was irrigated, dressed and healed uneventfully.
Thus, in all cases where a review was obtained and the patient had successful drainage, there was a 100% resolution in systemic symptoms.
Discussion
Antibiotics are the most widely prescribed category of drugs issued on prescription by general dental practitioners (GDPs), accounting for 7-10% of all prescriptions issued for antibiotics.8,9 A number of studies and surveys have revealed that there is widespread variation in the prescribing habits of GDPs with inconsistency in dosage, length of treatment and often inappropriate prescribing.4,5,6,10,11,12,13 A postal questionnaire by Lewis et al.14 in 1989 showed that dental practitioners estimated that only the minority of patients (approximately 5%) had an acute infection present when they issued a prescription for antibiotics. A similar picture is seen when looking at the prescribing patterns of general medical practitioners presented with oral pain.7
A number of audits have been carried out looking into the prescribing habits of general dental and general medical practitioners.15,16,17,18,19,20 Overwhelmingly these show that the antimicrobial prescribing habits are high when managing patients with acute dental pain, whether or not there is frank infection involved, and that there is wide variation in the type of antimicrobial prescribed, its dose and duration. They also highlight the lack of guidelines suggesting appropriateness in prescribing and illustrate how effective education is in reducing unnecessary prescriptions. Despite significant reductions in prescribing habits following education, it is still apparent that excessive prescriptions are being issued.16,18,19
Note has also been made of the vulnerability of general dental and medical practitioners in relation to such inappropriate prescribing in terms of potential litigation.21
A study by Kuriyama et al.15 highlights the excellent success rates in achieving stabilisation and improvement in the clinical situation following surgical drainage of the dentoalveolar infection along with rational prescribing. The definitive treatment for a patient with an acute dentoalveolar abscess is drainage followed by removal of the cause of the infection.15,22,23,24,25,26,27,28,29,30,31,32,33,34 This allows a release of pus reducing the overall number of bacteria, increasing oxygen diffusion and decreasing tissue pH.22 The predominant organisms isolated from dentoalveolar abscesses derived from the periodontal tissues are obligate anaerobes22,23,35,36,37,38,39,40,41 whereas those derived from the periapical tissues are mixed infections, with strictly anaerobic species exceeding facultative anaerobes by a factor of three to four.15,22,24,25,26,27,28,29,30,31,32,33,34,35,42,43,44,45
Following development of an abscess, the host response is to aid drainage of pus by the path of least resistance. Dependent on the anatomical site of the abscess, spread of infection may involve the muscles of mastication leading to a reduction in inter-incisal opening, presenting clinically as trismus. Alternatively the pus may spread deep to the buccinator muscle, through fascial planes, and spread beneath the skin, with the patient presenting with facial swelling. Both of these clinical signs should be regarded as signs of systemic involvement.47
As bacterial metabolites, endotoxins and exotoxins enter the bloodstream, the thermoregulatory centre in the hypothalamus responds by increasing body temperature and patients experience pyrexia.22 Pyrexia, along with regional lymphadenopathy, malaise, dysphagia, rigors and tachycardia are also signs of systemic reaction and antibiotics are needed to prevent progression to septicaemia.12,22,24,27,28,29,30,31,32,33,35,46,47,48,49 Between 2000-2005, the Office for National Statistics in England and Wales show a death rate from dentoalvolar abscess of 8-16 patients per year.50
For patients who exhibit signs of systemic infection related to their abscess, treatment with antibiotics is appropriate. The antibiotic is needed only until resolution of these systemic symptoms occurs. This usually takes 2-3 days.22,27,28,29,41,42,43,46
Resistance of micro-organisms to antibiotics is becoming increasingly important and a number of bacteria are now resistant to multiple antibiotics.51 Such is the case with methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Staphylococcus aureus (VRSA) and multiple drug-resistant Mycobacterium tuberculosis.52,53,54
Radical changes in prescribing habits and recognition of the increasing levels of resistant micro-organisms are needed to slow this ever-increasing trend.8,51,53,54,55,56,57,58,59,60,61,62 It is now clear that indiscriminate usage of antibiotics has contributed to this massive increase of resistant bacteria. As a consequence the European Centre for Disease Prevention launched the first European Antibiotics Awareness Day on 18 November 2008.63
The majority of micro-organisms isolated from dentoalveolar abscesses are Gram-negative anaerobes. Eick et al.61 demonstated that these were highly susceptible to metronidazole and clindamycin, but 22% of isolates were resistant to penicillin. Other studies have shown similar trends.60,64
The resurgence of clindamycin must also be considered. It has an excellent spectrum of activity against bacterial isolates from dentoalveloar abscesses, has superior bone penetration, stimulatory effects on the immune system and is well absorbed orally.24,42,59,65,66,67,68,69,70,71 Its historical link with pseudomembranous colitis (PMC) has been overestimated and is in fact no higher than other antimicrobials, including amoxicillin, when used in isolation.24,54,65,66,72,73,74 Caution is, however, needed in the elderly, the chronically ill, patients with a history of gastrointestinal disease, those on long-term antibiotics and those who have been hospitalised.73,75,76,77,78,79 Sandor et al22 and others have advocated its use as a primary therapeutic modality for the management of dentoalveolar abscesses.22,35,47,65,69,72,80
There is overwhelming evidence that the rise in resistant bacteria is due in part to the overprescribing of antibiotics. In order for antibiotics to continue to be effective at the time of definitive need, this rise in resistance needs to be slowed. A report by the Standing Medical Advisory Committee6 urged reduced prescribing in order to protect the future beneficial effects of antibiotics.
Historically, we as dental practitioners have been taught that antibiotics should be prescribed for 5-7 days and that patients must complete the course. It is now evident that this idea is misguided and that it actually leads to an increase in colonisation resistance.3,22 When short courses of antimicrobials are used, microbial conjugation is discouraged and transfer of resistant genes is minimised.3 When antibiotics are required, the most appropriate antibiotic should be prescribed in terms of its spectrum of activity. This optimises the therapeutic benefits of the antibiotic to the patient while minimising the risks of increasing microbial resistance. There is increasing evidence that many of the responsible oral flora are becoming resistant to penicillin59,60,61,62 and a number of studies have advocated the benefits of clindamycin as the first choice antibiotic for dentoalveolar abscess management in patients with evidence of systemic involvement.24,35,47,62,65,69,71,80
Conclusion
In the current climate of evidence-based medicine, an attempt has been made to rationalise the use of antibiotic prescribing for adult patients attending with acute dentoalveolar infections. Most can be successfully treated with surgical drainage followed by removal of the cause of the infection.15,22,23,24,25,26,27,28,29,30,31,32,33,34 For those patients who have become systemically unwell as a result of their infection, the same principles are followed along with antibiotic therapy to control and contain the systemic involvement.
This study has shown that a three day course of standard dose antibiotics, as per Figure 1, has been effective in managing these infections.
Given the annual costs to the National Health Service involved in the prescribing of antibiotics, the increasing levels of bacterial resistance, the emergence of bacterial strains resistant to multiple antimicrobial agents and the never-ending increase in litigation, extreme care should be taken when prescribing antibiotics for acute dentoalveolar infections and more emphasis should be placed on the provision of adequate drainage.
References
Ellison S J . The role of phenoxymethylpenicillin, amoxicillin, metronidazole and clindamycin in the management of acute dentoalveolar abscesses – a review. Br Dent J 2009; 206: 357–362.
Scottish Dental Clinical Effectiveness Programme. Emergency dental care: dental clinical guidance. Dundee: SDCEP, 2007. ISBN 978 1 905829 04 0.
Martin M V . Antimicrobials and dentistry: a rationale for their use. Faculty Dent J 2010; 1: 15–19.
Palmer N A, Pealing R, Ireland R S, Martin M V . A study of therapeutic antibiotic prescribing in National Health Service general dental practice in England. Br Dent J 2000; 188: 554–558.
Dailey Y M, Martin M V . Are antibiotics being used appropriately for emergency dental treatment? Br Dent J 2001; 191: 391–393.
Thomas D W, Satterthwaite J, Absi E G, Lewis M A, Shepherd J P . Antibiotic prescription for acute dental conditions in the primary care setting. Br Dent J 1996; 182: 401–404.
Anderson R, Calder L, Thomas D W . Antibiotic prescribing for dental conditions: general medical practitioners and dentists compared. Br Dent J 2000; 188: 398–400.
Standing Medical Advisory Committee, Department of Health. The path of least resistance. London: Department of Health, 1998.
Poveda Roda R, Bagan J V, Sanchis Bielsa J M, Carbonell Pastor E . Antibiotic use in dental practice. A review. Med Oral Patol Oral Cir Bucal 2007; 12; E186–E192.
Palmer N O, Martin M V, Pealing R, Ireland R S . An analysis of antibiotic prescriptions from general dental practitioners in England. J. Antimicrob Chemother 2000; 46: 1033–1035.
Roy K M, Bagg J . Antibiotic prescribing by general dental practitioners in the Greater Glasgow Health Board, Scotland. Br Dent J 2000; 188: 674–676.
Palmer N O, Martin M V, Pealing R et al. Antibiotic prescribing knowledge of National Health Service general dental practitioners in England and Scotland. J Antimicrob Chemother 2001; 47: 233–237.
Palmer N, Martin M V . An investigation of antibiotic prescribing by general dental practitioners: a pilot study. Prim Dent Care 1998; 5: 11–14.
Lewis M A, Meechan C, MacFarlane T W, Lamey P J, Kay E . Presentation and antimicrobial treatment of acute orofacial infections in general dental practice. Br Dent J 1989; 166: 41–45.
Kuriyama T, Absi E G, Williams D W, Lewis M A . An outcome audit of the treatment of acute dentoalveolar infection: impact of penicillin resistance. Br Dent J 2005; 198: 759–763.
Chate R A, White S, Hale L R et al. The impact of clinical audit on antibiotic prescribing in general dental practice. Br Dent J 2006; 201: 635–641.
Palmer N A, Dailey Y M . General dental practitioners' experiences of a collaborative clinical audit on antibiotic prescribing: a qualitative study. Br Dent J 2002; 193: 46–49.
Palmer N A, Dailey Y M, Martin M V . Can audit improve antibiotic prescribing in general dental practice? Br Dent J 2001; 191: 253–255.
Steed M, Gibson J . An audit of antibiotic prescribing in general dental practice. Prim Dent Care 1997; 4: 66–70.
Muthukrishnan A, Walters H, Douglas P S . An audit of antibiotic prescribing by general practitioners in the initial management of acute dental infection. Dent Update 1996; 23: 316–318.
Pogrel M A . The risk management of infections. Br Dent J 1992; 172: 354–355.
Marsh P, Martin M V . Oral microbiolgy. 4th ed. Edinburgh: Wright, 1999.
Stefanopoulos P K, Kolokotronis A E . The significance of anaerobic bacteria in acute orofacial odontogenic infections. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2004; 98: 398–408.
Sandor G K, Low D E, Judd P L, Davidson R J . Antimicrobial treatment options in the management of odontogenic infections. J Can Dent Assoc 1999; 65: 508–514.
Klein P B . Antibiotic therapy: maximize the benefits, minimize the risks. Dent Today 1994; 13: 42, 44–47.
Johnson B S . Principles and practice of antibiotic therapy. Infect Dis Clin North Am 1999; 13: 851–870.
Faculty of General Dental Practitioners (UK) of the Royal College of Surgeons of England. Primary dental care for general dental practitioners. London: FGDP(UK), 2000.
Martin M V, Longman L P, Hill J B, Hardy P . Acute dentoalveolar infections: an investigation of the duration of antibiotic therapy. Br Dent J 1997; 183: 135–137.
Pogrel M A . Antibiotics in general practice. Dent Update 1994; 21: 272–280.
Haas D A, Epstein J B, Eggert F M . Antimicrobial resistance: dentistry's role. J Can Dent Assoc 1998; 64: 496–502.
Palmer N A . Revisiting the role of dentists in prescribing antibiotics. Dent Update 2003; 30: 570–574.
Abbott P V, Hume W R, Pearman J W . Antibiotics and endodontics. Aust Dent J 1990; 35: 50–60.
Longman L P, Preston A J, Martin M V, Wilson N H . Endodontics in the adult patient: the role of antibiotics. J Dent 2000; 28: 539–548.
Lewis M A O, MacFarlane T W . Short-course high-dosage amoxicillin in the treatment of acute dento-alveolar abscess. Br Dent J 1986; 161: 299–302.
Dirks S J, Terezhalmy G T . The patient with an odontogenic infection. Quintessence Int 2004; 35: 482–502.
Rothwell B R . Odontogenic infections. Emerg Med Clin North Am 1985; 3: 161–178.
Johnson B D, Engel D . Acute necrotizing ulcerative gingivitis. A review of diagnosis, aetiology and treatment. J Periodontol 1986; 57: 141–149.
Loesche W J, Giordano J R . Metronidazole in periodontitis V: debridement should precede medication. Compendium 1994; 15: 1198–1217.
Blakey G H, White R P, Offenbacher S, Phillips C, Olutayo Delano E, Maynor G . Clinical/Biological outcomes of treatment for pericoronitis. J Oral Maxillofac Surg 1996; 54: 1150–1160.
Rosenberg E S, Torosian J P, Hammond B F, Cutler S A . Routine anaerobic bacterial culture and systemic antibiotic usage in the treatment of adult periodontitis: a 6 year longitudinal study. Int J Periodontics Restorative Dent 1993; 13: 213–243.
Walton R E, Zerr M, Peterson L . Antibiotics in dentistry – a boon or a bane? Alliance for the prudent use of Antibiotics Newsletter 1997; 15: 1–5.
Lewis M A O, MacFarlane T W, McGowan D A . Antibiotic susceptibilities of bacteria isolated from acute dentoalveolar abscesses. J Antimicrob Chemother 1989; 23: 69–77.
Lewis M A, MacFarlane T W, McGowan D A . Quantitative bacteriology of acute dento-alveolar abscesses. J Med Microbiol 1986; 21: 101–104.
Lewis M A, MacFarlane T W, McGowan D A . A microbiological and clinical review of the acute dentoalveolar abscess. Br J Oral Maxillofac Surg 1990; 28: 359–366.
Gill Y, Scully C . The microbiology and management of acute dentoalveolar abscess: views of British oral and maxillofacial surgeons. Br J Oral Maxillofac Surg 1988; 26: 452–457.
Pallasch T J . How to use antibiotics effectively. J Calif Dent Assoc 1993; 21: 46–50.
Flynn T R, Shanti R M, Levi M H, Adamo A K, Kraut R A, Treiger N . Severe odontogenic infections, part 1: prospective report. J Oral Maxillofac Surg 2006; 64: 1093–1103.
Swift J Q, Gulden W S . Antibiotic therapy – managing odontogenic infections. Dent Clin North Am 2002; 46: 623–633.
Martindale. The extra pharmacopoeia. 33rd ed. London: Pharmaceutical Press, 2002.
Office for National Statistics. Mortality statistics: deaths registered in England and Wales, 2000. Series DH2 No 27–32. London: Office for National Statistics, 2001.
House of Lords Select Committee on Science and Technology. Seventh report. Resistance to antibiotics and other antimicrobial agents. London: UK Parliament, 1998.
Pallasch T J . Global antibiotic resistance and its impact on the dental community. J Calif Dent Assoc 2000; 28: 215–233.
Samaranayake L P, Johnson N W . Guidelines for the use of antimicrobial agents to minimise development of resistance. Int Dent J 1999; 49: 189–195.
Standing Medical Advisory Committee. Report on impact of clinical prescribing on antimicrobial resistance. London: Department of Health, 1998.
Sweeney L C, Dave J, Chambers P A, Heritage J . Antibiotic resistance in general dental practice – a cause for concern? J Antimicrob Chemother 2004; 53: 567–576.
Fine D H, Hammond B F, Loesche W J . Clinical use of antibiotics in dental practice. Int J Antimicrob Agents 1998; 9: 235–238.
American Dental Association Council on Scientific Affairs. Antibiotics in dentistry. J Am Dent Assoc 1997; 128: 648.
Lewis M A, Parkhurst C L, Douglas C W et al. Prevelance of penicillin resistant bacteria in acute suppurative oral infection. J Antimicrob Chemother 1995; 35: 785–791.
Lewis M A O, Carmichael F, MacFarlane T W, Milligan S G . A randomised trial of co-amoxiclav versus penicillin V in the treatment of acute dentoalveolar abscess. Br Dent J 1993; 175: 169–174.
Flynn T R, Shanti R M, Hayes C . Severe odontogenic infections, part 2: prospective outcomes study. J Oral Maxillofac Surg 2006; 64: 1104–1113.
Eick S, Pfister W, Straub E . Antimicrobial susceptibility of anaerobic and capnophilic bacteria isolated from odontogenic abscesses and rapidly progressive periodontitis. Int J Antimicrob Agents 1999; 12: 41–46.
Kuriyama T, Williams D W, Yanagisawa M et al. Antimicrobial susceptibility of 800 anaerobic isolates from patients with dentoalveolar infection to 13 oral antibiotics. Oral Microbiol Immunol 2007; 22: 285–288.
Lewis M A O . Why we must reduce dental prescription of antibiotics: European Union Antibiotic Awareness Day. Br Dent J 2008; 205: 537–538.
Addy L D, Martin M V . Clindamycin and dentistry. Br Dent J 2005; 199: 23–26.
Stefanopoulos P K, Kolokotronis A E . Controversies in antibiotic choices for odontogenic infections. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2006; 101: 697–698.
Zetner K, Schmidt H, Pfeiffer S . Concentrations of clindamycin in the mandibular bone of companion animals. Int J Antimicrob Agents 2003; 4: 166–171.
Kirkwood K L . Update on antibiotics used to treat orofacial infections. Alpha Omega 2003; 96: 28–34.
Mueller S C, Henkel K, Neumann J et al. Perioperative antibiotic prophylaxis in maxillofacial surgery: penetration of clindamycin into various tissues. J Craniomaxillofac Surg 1999; 27: 172–176.
Gilbert D N, Moellering R C Jr, Eliopoulos G M, Sande M A (eds). The Sanford guide to antimicrobial therapy 2005. p 33. Sperryville, VA: Antimicrobial Therapy, Inc., 2005.
Reese R E, Betts R F, Gumustop B . Clindamycin. In Handbook of antibiotics. 3rd ed. pp 435–440. Philadelphia: Lippincott Williams & Wilkins, 2000.
Brook I, Lewis M A, Sandor G K et al. Clindamycin in dentistry: more than just effective prophylaxis for endocarditis? Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005; 100: 550–558.
Dajani A S, Taubert K A, Wilson W et al. Prevention of bacterial endocarditis. Recommendations by the American Heart Association. JAMA 1997; 277: 1794–1801.
Johnson S, Gerding D N . Clostridium difficile – associated diarrhea. Clin Infect Dis 1998; 26: 1027–1036.
Lewis J S, Jorgensen J H . Inducible clindamycin resistance in staphylococci: should clinicians and microbiologists be concerned? Clin Infect Dis 2005; 40: 280–285.
Baxter R, Ray G T, Fireman B H . Case-control study of antibiotic use and subsequent Clostridium difficle-associated diarrhea in hospitalized patients. Infect Control Hosp Epidemiol 2008; 29: 44–50.
Thibault A, Miller M A, Gaese C . Risk factors for the development of Clostridium difficle-associated diarrhea during a hospital outbreak. Infect Control Hosp Epidemiol 1991; 12: 345–348.
Andrejak M, Schmit J L, Tondriaux A . The clinical significance of antibiotic-associated pseudomembranous colitis in the 1990s. Drug Saf 1991; 6: 339–349.
Greenwood D, O'Grady F . Antibiotics and host defences. In The scientific basis of antimicrobial chemotherapy. Cambridge: Cambridge University Press, 1985.
Brause B D, Romankiewicz J A, Gotz V et al. Comparative study of diarrhea associated with clindamycin and ampicillin therapy. Am J Gastroenterol 1980; 73: 244–248.
Simmons N A, Ball A P, Cawson R A et al. Antibiotic prophylaxis and infective endocarditis. Lancet 1992; 339: 1292–1293.
Acknowledgements
I would like to thank Dr Mike Martin, Consultant Microbiologist, for his help, encouragement and expertise in the preparation of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed paper
Rights and permissions
About this article
Cite this article
Ellison, S. An outcome audit of three day antimicrobial prescribing for the acute dentoalveolar abscess. Br Dent J 211, 591–594 (2011). https://doi.org/10.1038/sj.bdj.2011.1051
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2011.1051
This article is cited by
-
Optimum length of treatment with systemic antibiotics in adults with dental infections: a systematic review
Evidence-Based Dentistry (2022)
-
'Antibiotics advice is nothing new' says FGDP (UK)
British Dental Journal (2017)
-
Antimicrobial prescribing by dentists in Wales, UK: findings of the first cycle of a clinical audit
British Dental Journal (2016)
-
Countersignature code
British Dental Journal (2012)