Key Points
-
Current evidence suggests a number of associations between oral diseases (periodontitis, tooth loss, oral cancer) and diabetes mellitus (diabetes). Whilst diabetic patients are at increased risk of post-surgical complications generally, there is little evidence that they are at increased risk following surgery in the oral cavity.
-
There is evidence that oral healthcare/management of oral disease, most notably periodontal care, has a short term beneficial influence on diabetes outcomes but there is no evidence that this is sustained over the long-term and/or reduces the development of the complications of diabetes.
-
Regular periodontal/oral care may benefit patients with periodontitis and diabetes.
-
A diagnosis of diabetes should be considered and excluded in those who have severe periodontitis and vice versa.
Abstract
Introduction This paper is the third of four rapid reviews undertaken to explore the relationships between oral health and general medical conditions in order to support teams within Public Health England, health practitioners and policymakers.
Aims This review aimed to explore the nature of the association between poor oral health and diabetes when found in the same individuals or populations, having reviewed the most contemporary evidence in the field.
Methods The reviews were undertaken by four groups each comprising consultant clinicians from medicine and dentistry, trainees, public health and academics. The methodology involved a streamlined rapid review process and synthesis of the findings.
Results The results identified a number of systematic reviews of low to high quality suggesting that diabetes is associated with periodontal disease, tooth loss, and oral cancer in particular, and that the management of oral diseases, most notably periodontal care, has a short-term beneficial influence on metabolic outcomes related to diabetes; however, there is no evidence that this is sustained over the long-term and reduces the prevalence of the long-term complications.
Conclusion Current evidence, of mixed quality, suggests a number of associations between oral diseases and diabetes mellitus (diabetes). Further high quality research is required in this field.
Similar content being viewed by others
Background
In England almost three million people have diabetes and many more are at risk of developing the condition. The estimated national spend by the NHS on diabetes is approximately £10 billion per year.1
Diabetes mellitus is a long-term condition characterised by an inability to control glucose levels in the blood due to an absolute or relative lack of the hormone insulin (International Classification of Diseases ICD-10 [E08E13]).2
The majority of people with diabetes can be classified as having type 1 or type 2. Type 1 diabetes is an autoimmune disease, whereby the body's own immune system attacks the body's own host tissues rather than infections. It tends to occur at a younger age and it is essential to treat with insulin. Type 2 diabetes is 8-9 times more common than type 1. It occurs as a result of a combination of lack of response to insulin (insulin resistance), and a lack of insulin production. It tends to occur in later life and in the early stages may be treated with oral medication.
Elevated glucose levels are associated with a number of complications including blindness, kidney failure, and minor and major leg amputation, which can be prevented by maintaining low glucose levels through a combination of both lifestyle change and medication. Diabetic foot disease occurs as a result of peripheral neuropathy; high glucose levels are associated with a wide range of both chronic and acute neuropathies that affect the full anatomic breadth of the peripheral nervous system from the nerve root to the skin, mirroring the spectrum of peripheral nerve diseases in general.3 In addition, diabetes is associated with an increased risk of vascular complications such as myocardial infarction, stroke and peripheral vascular disease.4 Obesity is a strong risk factor for the development and progression of diabetes; indeed, the increased incidence of overweight and obesity worldwide explains the ever increasing number of people affected by type 2 diabetes.5
It is preferable to view type 1 and 2 diabetes as separate conditions. For the purpose of this review we have used the term diabetes alone when the population studied is mixed, undifferentiated or not classified in the original article. In general, the majority of patients in the studies reviewed would be expected to have type 2 diabetes as it is more common.
The two most common diseases affecting oral health are dental caries and periodontitis. Dental caries (caries) is the localised destruction of susceptible dental hard tissues by acidic by-products from bacterial fermentation of dietary carbohydrates.6 Periodontitis is a chronic inflammatory disease caused by a dysbiosis of the sub-gingival microflora, resulting in the loss of the tissues surrounding the teeth.7 Approximately half of all adults in the UK are affected by some level of irreversible periodontitis, which increases with age, and almost a third of the same population have obvious dental decay.8
Both type 1 and 2 diabetes have been associated with many oral diseases in the past. Observational studies have suggested diabetes links with periodontal diseases (including peri-implant disease), caries (with its risks of tooth loss), oral mucosal disease (including oral infections), oral cancer, salivary dysfunction and oral dysaesthesias, including taste disturbances. Further clinical studies focused on the role of oral diseases and in particular periodontal status or therapy and their effect on hyperglycaemic control as well as the role of anti-diabetic medication in oral tissues.
However, with all the studies investigating these associations there has been little consensus with respect to the overall effect of diabetes mellitus on oral health and vice versa. This has made it difficult for both dental and medical clinicians alike to advise patients on these issues with great confidence. By pooling together the facts from evidence-based research in one place, this paper should provide a source for clinicians to give contemporary advice confidently.
Review methods
A rapid review of articles published between 2005 and 2015 investigating the relationship between diabetes and oral health was performed. A rapid review is a synthesis of the most current and best evidence to inform decision-makers.9 It combines elements of systematic reviews with a streamlined approach to summarise available evidence in a timely manner.
Search syntax was developed based on subject knowledge, MeSH terms and task group agreements (Appendix 1, included in online supplementary material), followed by duplicate systematic title and abstract searches of three electronic databases: Cochrane, PubMed, OVID (Embase, MEDLINE [R] and PsycINFO). Inclusion criteria were systematic reviews/meta-analyses involving diabetes and oral health, published in the ten-year period, involving human research and the full text being available in English. Exclusion criteria included animal studies and only systematic reviews and/or meta-analyses of observational and experimental studies were included in the final results.
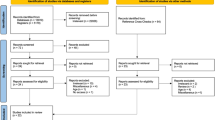
Two independent searches were undertaken, screening papers by title and abstract for relevance and duplication. Each researcher reviewed their search results and excluded papers that were not systematic reviews/meta-analyses, were not related to diabetes and any aspect of oral health, and were not available in English despite contacting the authors. After the final selection the searches were combined and duplicates removed. Both researchers discussed included and excluded papers before final agreement on the papers to be included for the rapid review. A flow diagram of the process is shown in Figure 1.
The following information was extracted from each paper: author, year, title, journal, population studied, oral disease/intervention, definitions used, methods, comparison/intervention and controls, outcomes, results, authors' conclusions, quality and quality justification; all shown in the data extraction table. From a total of 2,406 papers initially identified, there were 30 articles identified by the review for inclusion (Appendix 2, included in online supplementary material). Key findings from the rapid review extracted from the information above are shown below, with examples of the papers that provide evidence for the results synthesis. Each of the relevant oral health conditions, and relevant therapy, are considered in turn below starting with periodontitis and its management.
Quality assessment was undertaken for each systematic review using the PRISMA and AMSTAR tools to ascertain risk of bias. An AMSTAR assessment was carried out on all papers with the methodological quality of the review being rated as 'High' with a score between 11 and eight, 'Moderate' between seven and four, and 'Low' between four and zero. The quality of all papers was confirmed by group discussion.
The quality of the systematic reviews identified varied. Of the 30 systematic reviews, 12 were deemed to be of high quality, 14 of moderate quality and four were of low quality (Appendix 2, included in online supplementary material). Common AMSTAR quality issues were a) lack of a priori design or question, b) no quality appraisal, c) missing tables, and d) no duplicate study selection.
Within the theme identified by this review, reviews related to investigation of i] glycaemic control and periodontal therapy and ii] the risk of oral cancer in patients with diabetes were of high quality, while articles on the association between dental implant therapy,10,11osseo-integration12 and diabetic neuropathy,13 with oral diseases or therapy were of low quality.
Results: evidence synthesis
The results are reported in nine sections, each relating to an oral disease or condition investigated. Each section contains evidence of any association between the oral disorder and diabetes and also on the impact of oral therapy on diabetes and diabetes management on oral health.
I] Periodontitis and diabetes
There is high quality evidence that diabetes (type 2) is a risk factor for periodontitis.14 There is some moderate quality evidence that while the extent of periodontitis, that is, percentage of surfaces/sites affected is similar between patients with or without diabetes, measures of severity including greater clinical attachment loss and pocket probing depths are greater in patients with diabetes.15 Patients with diabetic complications, most specifically diabetic neuropathy, appear to have poorer oral health, including periodontitis, than those without neuropathy.13 However, there is scant evidence from two reviews of variable quality that diagnosis of periodontitis: a) promotes development of type 2 diabetes and b) adversely affects glycaemic control and diabetes complications in patients with diabetes.13,16
In summary, while there is strong evidence that diabetes is a recognised risk factor for periodontitis, more research is needed to ascertain the impact of periodontitis on the development and progression of diabetes.
II] Periodontal therapy and glycaemic control
One high quality systematic review provides evidence that in patients with type 2 diabetes, intensive periodontal therapy involving scaling and root planning reduced HbA1c (a marker of glycaemic control) by 0.29% [3-4 mmol/l] for up to three months; however, after six months there was no evidence that this reduction was sustained.17 Modest improvements in glycaemic control, as demonstrated by a reduction in Hb1Ac, are supported by seven other moderate quality systematic reviews,16,18,19,20,21,22,23 while one was equivocal.24 In one of the reviews by Wang et al. 2014, there was evidence that after three months follow-up, periodontal treatment substantially lowered Haemoglobin A1c (HbA1c), compared with no treatment (–0.36%, 95%CI, 0.52% to 0.19%, P <0.0001). These findings were accompanied by substantial and statistically significant reductions in pocket probing depth (PPD) and clinical attachment loss (CAL) between study groups (PPD 0.42 mm, 95%CI: 0.60 to 0.23, P <0.00001; CAL 0.34 mm, 95%CI: 0.52 to 0.16, P = 0.0002). Consistent with the 2015 Cochrane review, there was no substantial change of HbA1c levels after six months.22
In summary, there is merit in performing periodontal therapy in patients with type 2 diabetes with regards to a short term improvement in metabolic control. Further research is needed to demonstrate that this benefit is sustained over longer follow-up and translates into reduction of diabetes complications.
III] Periodontal therapy and systemic/surrogate markers
One high quality review suggests that periodontal treatment reduced markers of systemic inflammation in patients with diabetes: serum levels of TNF- α and CRP.25 Sgolastra et al., 201320 reported no significant improvements in lipid fractions (total cholesterol, triglycerides and high and low density lipoprotein cholesterol [TC, TG, HDL, or LDL]) in patients with diabetes and chronic periodontitis who received scaling and root planing (SRP).20
Lastly, five different reviews suggest that different types of periodontal treatment: surgical or non-surgical, with or without the use of adjunctive antibiotics, antiseptics, or oral hygiene instructions do not appear to produce different effects on glycaemic control in patients with diabetes.17,18,21,22,26
IV] Tooth loss and diabetes
Patients with diabetic complications, most specifically diabetic neuropathy, appear to have more tooth loss than those without neuropathy.13 This finding is consistent with the view that diabetes and its complications are associated with poorer oral health.
V] Caries and diabetes
Conflicting evidence on the association between diabetes and increased prevalence of caries was found. Greater levels of dental plaque were noted in patients with diabetes and this may have implications for future risk of caries.27 Furthermore, children with type 1 diabetes are at increased risk of periodontal diseases as evidenced by greater dental plaque levels, gingival inflammation and bleeding.
VI] Dental implants and diabetes
There is limited evidence that poor metabolic control is associated with peri-implant disease28,29and weak evidence of higher marginal bone loss around dental implants in patients with diabetes with unspecified metabolic control.10 There is equivocal evidence that patients with diabetes have higher failure rates of dental implants and no evidence that diabetes is a contraindication to dental implant placement;11,12,30 however, some evidence for delay in implant osseo-integration based on glycaemic control was identified.10
VII] Oral surgery and diabetes
While there is evidence that people with diabetes are more likely to suffer complications of surgery in other areas, specific evidence that diabetes is associated with post-operative complications in the oral cavity is lacking.31
VIII] Oral squamous cell carcinoma and diabetes
A high quality systematic review suggests that type 2 diabetes is associated with an elevated risk of oral cancer and precancerous lesions.32 Patients with type 2 diabetes have a higher case mortality on diagnosis of oral cancer, independent of tobacco, alcohol and obesity factors with a relative risk of 1.41(95%CI: 1.16–1.72) when comparing patients with diabetes to people without diabetes, with no evident heterogeneity among studies.32
IX] Saliva and diabetes
Patients with diabetes complications, most specifically diabetic neuropathy, appear to have greater mouth dryness than controls.13 There is limited evidence on the role of diabetes in increasing the risk of salivary dysfunction.33 There is some recent, limited and weak evidence that salivary protein markers may be used to monitor glycaemic control accurately as a less painful alternative to capillary blood glucose measurements.34
Summary
The evidence reviewed to form the conclusions for this rapid review demonstrates that there are oral manifestations of diabetes, including effects on oral health, notably periodontitis, oral cancer risk, and that diabetes may affect dental implants success. More evidence is required before advising clinicians of any contraindications to perform implants in diabetic patients. There is strong evidence of type 2 diabetes being a risk factor for periodontal diseases and weak evidence in relation to type 1. There is weak evidence in relation to dental caries experience in children. Limited evidence exists of periodontitis being a risk factor for diabetes; however, there is a growing body of evidence that professional periodontal treatment (that is, scaling and root planning as a minimum) results in modest improvement in glycaemic control in the short-term but this is not sustained beyond three months. Furthermore, the average reduction in glycated haemoglobin observed following periodontal therapy (0.3%/3-4 mmol/l) could translate into long-term clinical benefits but further research is needed. The evidence from phase 3 trials should make further contributions to this issue.35
Overall, there is some evidence of a bidirectional relationship between diabetes and oral health and the management of conditions, most notably for periodontitis on diabetes. This is a very important area for further interdisciplinary patient care and research, particularly as health professionals care for an ageing population with multiple co-morbidities. As dental professionals we need to keep abreast of the emerging evidence of associations between non-communicable diseases. While there is no evidence to support a major change in patient management we should be aware that regular periodontal care may benefit patients with periodontitis and diabetes. However, patients with periodontitis need help with regular self-care and professional periodontal treatment.
The rapid nature of the review is a potential limitation especially with the large scope of the topic reviewed. However, the methodology used has been proven successful previously in work by Khangura et al.9 This rapid review was limited by the quality of the systematic reviews and meta-analyses appraised. These limitations are well summarised in the Cochrane review by Simpson et al.17 and highlighted in Appendix 2 (included in online supplementary material). Overall the quality of the reviews in this field was mixed while the research on the topic was generally judged to be of low quality.
It is important to note that the publications do not use standard definitions and classification of periodontitis as used by the American guidelines;7 these should be standardised in future to facilitate comparability between studies.
There is evidence that management of oral disease, most notably periodontal care, has a short term beneficial influence on diabetes but no evidence that this is sustained over the long-term and reduces diabetes complications. The vast majority of the articles described define diabetes only by its presence or absence and many do not even classify which type of diabetes was present. There was no information regarding the effect of different levels of glucose control on either the absence or presence of an association or whether the association has a glucose exposure 'dose effect'. Testing the association of HbA1c and dental outcomes would therefore be useful for future research. Ideally, type 1 and 2 diabetes should be studied separately given the above. More high quality multidisciplinary research is needed to investigate the relationships between the non-communicable diseases and their management as shown in Box 1.
In summary, the current evidence suggests a number of associations between oral diseases and diabetes. There is limited evidence to support major changes to medical or dental therapy; however, regular periodontal/oral care may benefit glycaemic control in patients with periodontitis and diabetes. At present, diagnosis of diabetes should not require a change to dental therapy, such as restricting use of dental implants. Greater awareness of the impact of diabetes on oral health and vice versa is needed among medical and dental health professionals.
References
National Health Service. Five year forward view 2014 11/11/2014. Available at: http://www.england.nhs.uk/wp-content/uploads/2014/10/5yfv-web.pdf (accessed May 2017).
World Health Organization. International Classification of Diseases ICD-10. Geneva: WHO, 2017. Available at: http://www.who.int/classifications/icd/en/ (accessed May 2017).
Bullon P, Newman H N, Battino M . Obesity, diabetes mellitus, atherosclerosis and chronic periodontitis: a shared pathology via oxidative stress and mitochondrial dysfunction? Periodontol 2000 2014; 64: 139–153.
King P, Peacock I, Donnelly R . The UK prospective diabetes study (UKPDS): clinical and therapeutic implications for type 2 diabetes. Br J Clin Pharmacol 1999; 48: 643–648.
Eckel R H, Kahn S E, Ferrannini E et al. Obesity and type 2 diabetes: what can be unified and what needs to be individualized? Diabetes Care 2011; 34: 1424–1430.
Fejerskov O, Kidd E . Dental caries: the disease and its clinical management. John Wiley & Sons, 2009.
Eke P I, Page R C, Wei L, Thornton-Evans G, Genco R J . Update of the case definitions for population-based surveillance of periodontitis. J Periodontol 2012; 83: 1449–1454.
Steele J, O'Sullivan I . Adult Dental Health Survey 2011 21/05/2011. Available at: http://www.hscic.gov.uk/catalogue/PUB01061/adul-dent-heal-surv-firs-rele-2009-rep.pdf (accessed May 2017).
Khangura S, Konnyu K, Cushman R, Grimshaw J, Moher D . Evidence summaries: the evolution of a rapid review approach. Syst Rev 2012; 1: 10 10.1186/2046-4053-1-10.
Oates T W, Huynh-Ba G, Vargas A, Alexander P, Feine J . A critical review of diabetes, glycaemic control, and dental implant therapy. Clin Oral Implants Res 2013; 24: 117–27.
Bornstein M M, Cionca N, Mombelli A . Systemic conditions and treatments as risks for implant therapy. Int J Oral Maxillofac Implants 2009; 24 Suppl: 12–27.
Mombelli A, Cionca N . Systemic diseases affecting osseointegration therapy. Clin Oral Implants Res 2006; 17: 97–103.
Borgnakke W S, Anderson P F, Shannon C, Jivanescu A . Is there a relationship between oral health and diabetic neuropathy? Curr Diab Rep 2015; 15: 93.
Chavarry N G, Vettore M V, Sansone C, Sheiham A . The relationship between diabetes mellitus and destructive periodontal disease: a meta-analysis. Oral Health Prev Dent 2009; 7: 107–127.
Khader Y S, Dauod A S, El-Qaderi S S, Alkafajei A, Batayha W Q . Periodontal status of diabetics compared with nondiabetics: a meta-analysis. J Diabetes Complications 2006; 20: 59–68.
Borgnakke W S, Ylostalo P V, Taylor G W, Genco R J . Effect of periodontal disease on diabetes: systematic review of epidemiologic observational evidence. J Periodontol 2013; 84 (4 Suppl): S135–152.
Simpson T C, Weldon J C, Worthington H V et al. Treatment of periodontal disease for glycaemic control in people with diabetes mellitus. Cochrane Database Syst Rev 2015; 11: Cd004714.
Engebretson S, Kocher T . Evidence that periodontal treatment improves diabetes outcomes: a systematic review and meta-analysis. J Periodontol 2013; 84 (4 Suppl): S153–169.
Li Q, Hao S, Fang J, Xie J, Kong X H, Yang J X . Effect of non-surgical periodontal treatment on glycaemic control of patients with diabetes: a meta-analysis of randomized controlled trials. Trials 2015; 16: 291.
Sgolastra F, Severino M, Pietropaoli D, Gatto R, Monaco A . Effectiveness of periodontal treatment to improve metabolic control in patients with chronic periodontitis and type 2 diabetes: a meta-analysis of randomized clinical trials. J Periodontol 2013; 84: 958–973.
Teeuw W J, Gerdes V E, Loos B G . Effect of periodontal treatment on glycaemic control of diabetic patients: a systematic review and meta-analysis. Diabetes Care 2010; 33: 421–427.
Wang T F, Jen I A, Chou C, Lei Y P . Effects of periodontal therapy on metabolic control in patients with type 2 diabetes mellitus and periodontal disease: a meta-analysis. Medicine (Baltimore) 2014; 93: e292.
Sun Q, Feng M, Zhang M et al. Effects of periodontal treatment on glycaemic control in type 2 diabetes patients; a meta-analysis of randomised controlled trials. Chinese J Physiol 2014; 57: 305–314.
Mauri-Obradors E, Jane-Salas E, Sabater-Recolons Mdel M, Vinas M, Lopez-Lopez J. Effect of nonsurgical periodontal treatment on glycosylated haemoglobin in diabetic patients: a systematic review. Odontology 2015; 103: 301–313.
Artese H P C, Foz A M, Rabelo M D et al. Periodontal therapy and systemic inflammation in type 2 diabetes mellitus: a meta-analysis. PLoS One 2015; 10: 14.
Corbella S, Francetti L, Taschieri S, De Siena F, Fabbro M D . Effect of periodontal treatment on glycaemic control of patients with diabetes: A systematic review and meta-analysis. J Diabetes Investig 2013; 4: 502–509.
Ismail A F, McGrath C P, Yiu C K Y . Oral health of children with type 1 diabetes mellitus: a systematic review. Diabetes Res Clin Pract 2015; 108: 369–381.
Chrcanovic BR, Albrektsson T, Wennerberg A . Diabetes and oral implant failure: a systematic review. J Dent Res. 2014; 93 (9): 859–67.
Javed F, Romanos G E . Impact of diabetes mellitus and glycaemic control on the osseointegration of dental implants: a systematic literature review. J Periodontol 2009; 80: 1719–1730.
Chen H, Liu N, Xu X, Qu X, Lu E . Smoking, radiotherapy, diabetes and osteoporosis as risk factors for dental implant failure: a meta-analysis. PLoS One 2013; 8: e71955.
Barasch A, Safford M M, Litaker M S, Gilbert G H . Risk factors for oral postoperative infection in patients with diabetes. Spec Care Dentist 2008; 28: 159–166.
Gong Y, Wei B, Yu L, Pan W . Type 2 diabetes mellitus and risk of oral cancer and precancerous lesions: a meta-analysis of observational studies. Oral Oncol 2015; 51: 332–340.
de Morais E F, de Paiva Macedo R A, da Silva Lira J A, de Lima K C, Borges B C D . Factors related to dry mouth and low salivary flow rates in diabetic elderly: a systematic literature review. Revista Brasileira de Geriatria e Gerontologia 2014; 17: 417–423.
Mascarenhas P, Fatela B, Barahona I . Effect of Diabetes Mellitus Type 2 on Salivary Glucose–A Systematic Review and Meta-Analysis of Observational Studies. PLoS One 2014; 9: e101706.
Suvan J . Periodontitis and type 2 diabetes mellitus: Phase 3 trial. BMC ISRCTN registry. 2017; ISRCTN83229304.
Carramolino-Cuellar E, Tomas I, Jimenez-Soriano Y. Relationship between the oral cavity and cardiovascular diseases and metabolic syndrome. Med Oral Patol Oral Cir Bucal. 2014; 19: e289–e294.
Darre L, Vergnes J N, Gourdy P, Sixou M . Efficacy of periodontal treatment on glycaemic control in diabetic patients: A meta-analysis of interventional studies. Diabetes Metab. 2008; 34: 497–506.
Janket S J, Wightman A, Baird A E, Van Dyke T E, Jones J A . Does Periodontal Treatment Improve Glycemic Control in Diabetic Patients? A Meta-analysis of Intervention Studies. J Dent Res 2005; 84: 1154–1159.
Kudiyirickal M G, Pappachan J M . Diabetes mellitus and oral health. Endocrine 2015; 49: 27–34.
Renvert S, Roos-Jansaker AM, Claffey N . Non-surgical treatment of peri-implant mucositis and peri-implantitis: a literature review. J Clin Periodontol 2008; 35 (8 Suppl): 305–315.
Acknowledgements
We would like to acknowledge the support of Public Health England, the Royal College of Surgeons and the British Dental Association.
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed Paper
Supplementary information
Appendix 1
Search terms (PDF 79 kb)
Appendix 2
Included papers (PDF 245 kb)
Rights and permissions
About this article
Cite this article
D'Aiuto, F., Gable, D., Syed, Z. et al. Evidence summary: The relationship between oral diseases and diabetes. Br Dent J 222, 944–948 (2017). https://doi.org/10.1038/sj.bdj.2017.544
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2017.544
This article is cited by
-
Opportunistic health screening for cardiovascular and diabetes risk factors in primary care dental practices: experiences from a service evaluation and a call to action
British Dental Journal (2023)
-
Feasibility of a nursing guide for the oral management of outpatients with diabetes mellitus
Diabetology International (2023)
-
Nurses’ knowledge, attitude, and practice regarding oral management for outpatients with type 2 diabetes: a national survey on certified diabetes educators
Diabetology International (2022)
-
The prevalence of potentially undiagnosed type II diabetes in patients with chronic periodontitis attending a general dental practice in London - a feasibility study
British Dental Journal (2021)
-
Massive data analyses show negative impact of type 1 and 2 diabetes on the outcome of periodontal treatment
Clinical Oral Investigations (2021)