Abstract
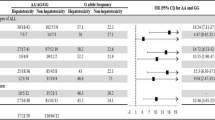
A missense variant (c.1637C>T, T546M) in ABCC11 encoding the MRP8 (multidrug resistance protein 8), a transporter of 5-fluorodeoxyuridine monophosphate, has been associated with an increased risk of 5-fluorouracil-related severe leukopenia. To validate this association, we investigated the impact of the ABCC11 variants c.1637C>T, c.538G>A and c.395+1087C>T on the risk of early-onset fluoropyrimidine-related toxicity in 514 cancer patients. The ABCC11 variant c.1637C>T was strongly associated with severe leukopenia in patients carrying risk variants in DPYD, encoding the key fluoropyrimidine-metabolizing enzyme dihydropyrimidine dehydrogenase (odds ratio (OR): 71.0; 95% confidence interval (CI): 2.5–2004.8; Pc.1637C>T*DPYD=0.013). In contrast, in patients without DPYD risk variants, no association with leukopenia (OR: 0.95; 95% CI: 0.34–2.6) or overall fluoropyrimidine-related toxicity (OR: 1.02; 95% CI: 0.5–2.1) was observed. Our study thus suggests that c.1637C>T affects fluoropyrimidine toxicity to leukocytes particularly in patients with high drug exposure, for example, because of reduced fluoropyrimidine catabolism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Amstutz U, Froehlich TK, Largiadèr CR . Dihydropyrimidine dehydrogenase gene as a major predictor of severe 5-fluorouracil toxicity. Pharmacogenomics 2011; 12: 1321–1336.
Meulendijks D, Henricks LM, Sonke GS, Deenen MJ, Froehlich TK, Amstutz U et al. Clinical relevance of DPYD variants c.1679T>G, c.1236G>A/HapB3, and c.1601G>A as predictors of severe fluoropyrimidine-associated toxicity: a systematic review and meta-analysis of individual patient data. Lancet Oncol 2015; 16: 1639–1650.
Rosmarin D, Palles C, Church D, Domingo E, Jones A, Johnstone E et al. Genetic markers of toxicity from capecitabine and other fluorouracil-based regimens: investigation in the QUASAR2 study, systematic review, and meta-analysis. J Clin Oncol 2014; 32: 1031–1039.
Lee AM, Shi Q, Pavey E, Alberts SR, Sargent DJ, Sinicrope FA et al. DPYD variants as predictors of 5-fluorouracil toxicity in adjuvant colon cancer treatment (NCCTG N0147). J Natl Cancer Inst 2014; 106doi:10.1093/jnci/dju298.
Longley DB, Harkin DP, Johnston PG . 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer 2003; 3: 330–338.
Harstrick A, Gonzales A, Schleucher N, Vanhoefer U, Lu K, Formento J-L et al. Comparison between short or long exposure to 5-fluorouracil in human gastric and colon cancer cell lines: biochemical mechanism of resistance. Anticancer Drugs 1998; 9: 625–634.
Nies AT, Magdy T, Schwab M, Zanger UM . Role of ABC transporters in fluoropyrimidine-based chemotherapy response. Adv Cancer Res 2015; 125: 217–243.
Oguri T, Bessho Y, Achiwa H, Ozasa H, Maeno K, Maeda H et al. MRP8/ABCC11 directly confers resistance to 5-fluorouracil. Mol Cancer Ther 2007; 6: 122–127.
Pratt S, Shepard RL, Kandasamy RA, Johnston PA, Perry W, Dantzig AH . The multidrug resistance protein 5 (ABCC5) confers resistance to 5-fluorouracil and transports its monophosphorylated metabolites. Mol Cancer Ther 2005; 4: 855–863.
Guo Y, Kotova E, Chen Z-S, Lee K, Hopper-Borge E, Belinsky MG et al. MRP8, ATP-binding cassette C11 (ABCC11), is a cyclic nucleotide efflux pump and a resistance factor for fluoropyrimidines 2',3'-dideoxycytidine and 9‘-(2’-phosphonylmethoxyethyl)adenine. J Biol Chem 2003; 278: 29509–29514.
Magdy T, Arlanov R, Winter S, Lang T, Klein K, Toyoda Y et al. ABCC11/MRP8 polymorphisms affect 5-fluorouracil-induced severe toxicity and hepatic expression. Pharmacogenomics 2013; 14: 1433–1448.
Arlanov R, Lang T, Jedlitschky G, Schaeffeler E, Ishikawa T, Schwab M et al. Functional characterization of common protein variants in the efflux transporter ABCC11 and identification of T546M as functionally damaging variant. Pharmacogenomics J 2015. doi:10.1038/tpj.2015.27; e-pub ahead of print.
The 1000 Genomes Project Consortium. An integrated map of genetic variation from 1,092 human genomes. Nature 2012; 491: 56–65.
Yoshiura K, Kinoshita A, Ishida T, Ninokata A, Ishikawa T, Kaname T et al. A SNP in the ABCC11 gene is the determinant of human earwax type. Nat Genet 2006; 38: 324–330.
Schröder A, Klein K, Winter S, Schwab M, Bonin M, Zell A et al. Genomics of ADME gene expression: mapping expression quantitative trait loci relevant for absorption, distribution, metabolism and excretion of drugs in human liver. Pharmacogenomics J 2013; 13: 12–20.
National Institutes of Health, National Cancer Institute. Common Terminology Criteria for Adverse Events v3.0 (CTCAE). 2006; 1–72; http://ctep.cancer.gov/protocolDevelopment/.
Froehlich TK, Amstutz U, Aebi S, Joerger M, Largiadèr CR . Clinical importance of risk variants in the dihydropyrimidine dehydrogenase gene for the prediction of early-onset fluoropyrimidine toxicity. Int J Cancer 2015; 136: 730–739.
Amstutz U, Offer SM, Sistonen J, Joerger M, Diasio RB, Largiadèr CR . Polymorphisms in MIR27A associated with early-onset toxicity in fluoropyrimidine-based chemotherapy. Clin Cancer Res 2015; 21: 2038–2044.
Raymond M, Rousset F . GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Heredity 1995; 86: 248–249.
Harrell FE . rms: Regression Modeling Strategies. R package version 4.1-1 2009. https://cran.r-project.org/web/packages/rms/index.html.
R Development Core Team. R: A Language and Environment for Statistical Computing. The R Foundation for Statistical Computing: Vienna, Austria; http://www.R-project.org/, 2013.
Caudle KE, Thorn CF, Klein TE, Swen JJ, McLeod HL, Diasio RB et al. Clinical Pharmacogenetics Implementation Consortium guidelines for dihydropyrimidine dehydrogenase genotype and fluoropyrimidine dosing. Clin Pharmacol Ther 2013; 94: 640–645.
Faul F, Erdfelder E, Lang A-G, Buchner A . G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 2007; 39: 175–191.
Kummer D, Froehlich TK, Joerger M, Aebi S, Sistonen J, Amstutz U et al. Dihydropyrimidinase and β-ureidopropionase gene variation and severe fluoropyrimidine-related toxicity. Pharmacogenomics 2015; 16: 1367–1377.
Amstutz U, Farese S, Aebi S, Largiadèr CR . Dihydropyrimidine dehydrogenase gene variation and severe 5-fluorouracil toxicity: a haplotype assessment. Pharmacogenomics 2009; 10: 931–944.
Schwab M, Zanger UM, Marx C, Schaeffeler E, Klein K, Dippon J et al. Role of genetic and nongenetic factors for fluorouracil treatment-related severe toxicity: a prospective clinical trial by the German 5-FU Toxicity Study Group. J Clin Oncol 2008; 26: 2131–2138.
Stein TA, Bailey B, Burns GP . Comparison of 5-fluorouracil anabolite levels after intravenous bolus and continuous infusion. Cancer Chemother Pharmacol 1994; 34: 293–296.
van Kuilenburg ABP, Häusler P, Schalhorn A, Tanck MWT, Proost JH, Terborg C et al. Evaluation of 5-fluorouracil pharmacokinetics in cancer patients with a c.1905+1G>A mutation in DPYD by means of a Bayesian limited sampling strategy. Clin Pharmacokinet 2012; 51: 163–174.
Thrall MM, Wood P, King V, Rivera W, Hrushesky W . Investigation of the comparative toxicity of 5-FU bolus versus 5-FU continuous infusion circadian chemotherapy with concurrent radiation therapy in locally advanced rectal cancer. Int J Radiat Oncol Biol Phys 2000; 46: 873–881.
Meta-analysis Group In Cancer, Piedbois P, Rougier P, Buyse M, Pignon J, Ryan L et al. Efficacy of intravenous continuous infusion of fluorouracil compared with bolus administration in advanced colorectal cancer. J Clin Oncol 1998; 16: 301–308.
Tsuchiya T, Arai J, Matsumoto K, Miyazaki T, Honda S, Tagawa T et al. Prognostic impact of the ABCC11/MRP8 polymorphism in adjuvant oral chemotherapy with S-1 for non-small cell lung cancer. Chemotherapy 2015: 77–86.
Toyoda Y, Sakurai A, Mitani Y, Nakashima M, Yoshiura K-I, Nakagawa H et al. Earwax, osmidrosis, and breast cancer: why does one SNP (538G>A) in the human ABC transporter ABCC11 gene determine earwax type? FASEB J 2009; 23: 2001–2013.
Acknowledgements
We thank Gisela Andrey, Marion Ernst, Gabriela Mäder and Daniela Sommer for their laboratory work and Johanna Sistonen for helpful discussions. We also thank Simone Farese and all those involved from participating oncology centers for their contributions in patient recruitment and clinical data collection. We also thank the Swiss National Science Foundation for their financial support (Grants 119839 and 138285).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Hamzic, S., Wenger, N., Froehlich, T. et al. The impact of ABCC11 polymorphisms on the risk of early-onset fluoropyrimidine toxicity. Pharmacogenomics J 17, 319–324 (2017). https://doi.org/10.1038/tpj.2016.23
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2016.23