Abstract
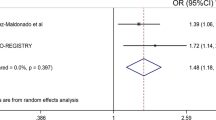
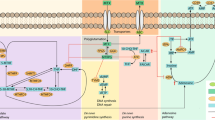
Several genetic variants in Toll-like receptor (TLR) and nuclear factor (NF)-κB signalling pathways have been reported associated with responsiveness to tumour necrosis factor inhibitor (anti-TNF) treatment in rheumatoid arthritis (RA). The present study was undertaken to replicate these findings. In a retrospective case–case study including 1007 Danish anti-TNF-treated RA patients, we genotyped 7 previously reported associated single-nucleotide polymorphisms (SNPs) in these pathways. Furthermore, 5 SNPs previously reported by our group were genotyped in a subcohort (N=469). Primary analyses validated the IRAK3 rs11541076 variant as associated (odds ratio (OR)=1.33, 95% confidence interval (CI): 1.00–1.77, P-value=0.047) with a positive treatment response (EULAR (European League Against Rheumatism) good/moderate vs none response at 4±2 months), and found the NLRP3 rs461266 variant associated (OR=0.75, 95% CI: 0.60–0.94, P=0.014) with a negative treatment response. Meta-analyses combining data from previous studies suggested smaller effect sizes of associations between variant alleles of CHUK rs11591741, NFKBIB rs3136645 and rs9403 and a negative treatment response. In conclusion, this study validates rs11541076 in IRAK3, a negative regulator of TLR signalling, as a predictor of anti-TNF treatment response, and suggests true positive associations of previously reported SNPs within genes encoding activators/inhibitors of NF-κB (CHUK, MYD88, NFKBIB, and NLRP3).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hetland ML, Christensen IJ, Tarp U, Dreyer L, Hansen A, Hansen IT et al. Direct comparison of treatment responses, remission rates, and drug adherence in patients with rheumatoid arthritis treated with adalimumab, etanercept, or infliximab: results from eight years of surveillance of clinical practice in the nationwide Danish DANBIO registry. Arthritis Rheum 2010; 62: 22–32.
Dennis G Jr, Holweg CT, Kummerfeld SK, Choy DF, Setiadi AF, Hackney JA et al. Synovial phenotypes in rheumatoid arthritis correlate with response to biologic therapeutics. Arthritis Res Ther 2014; 16: R90.
Plant D, Prajapati R, Hyrich KL, Morgan AW, Wilson AG, Isaacs JD et al. Replication of association of the PTPRC gene with response to anti-tumor necrosis factor therapy in a large UK cohort. Arthritis Rheum 2012; 64: 665–670.
Cui J, Stahl EA, Saevarsdottir S, Miceli C, Diogo D, Trynka G et al. Genome-wide association study and gene expression analysis identifies CD84 as a predictor of response to etanercept therapy in rheumatoid arthritis. PLoS Genet 2013; 9: e1003394.
Acosta-Colman I, Palau NF, Tornero JF, Fernandez-Nebro AF, Blanco FF, Gonzalez-Alvaro IF et al. GWAS replication study confirms the association of PDE3A-SLCO1C1 with anti-TNF therapy response in rheumatoid arthritis. Pharmacogenomics 2013; 14: 727–734.
Sode J, Vogel U, Bank S, Andersen PS, Thomsen MK, Hetland ML et al. Anti-TNF treatment response in rheumatoid arthritis patients is associated with genetic variation in the NLRP3-inflammasome. PLoS One 2014; 9: e100361.
Sode J, Vogel U, Bank S, Andersen PS, Hetland ML, Locht H et al. Genetic variations in pattern recognition receptor loci are associated with anti-TNF response in patients with rheumatoid arthritis. PLoS One 2015; 10: e0139781.
Potter C, Cordell HJ, Barton A, Daly AK, Hyrich KL, Mann DA et al. Association between anti-tumour necrosis factor treatment response and genetic variants within the TLR and NF{kappa}B signalling pathways. Ann Rheum Dis 2010; 69: 1315–1320.
Hetland ML . DANBIO—powerful research database and electronic patient record. Rheumatology (Oxford) 2011; 50: 69–77.
Gauderman WJ . Sample size requirements for matched case-control studies of gene-environment interaction. Stat Med 2002; 21: 35–50.
Ferreiro-Iglesias A, Montes A, Perez-Pampin E, Canete JD, Raya E, Magro-Checa C et al. Replication of PTPRC as genetic biomarker of response to TNF inhibitors in patients with rheumatoid arthritis. Pharmacogenomics J 2015; 16: 137–140.
Lawrence T . The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol 2009; 1: a001651.
Kinoshita T, Imamura R, Kushiyama H, Suda T . NLRP3 mediates NF-kappaB activation and cytokine induction in microbially induced and sterile inflammation. PLoS One 2015; 10: e0119179.
Kobayashi K, Hernandez LD, Galan JE, Janeway CA Jr, Medzhitov R, Flavell RA . IRAK-M is a negative regulator of Toll-like receptor signaling. Cell 2002; 110: 191–202.
O'neill LA, Sheedy FJ, McCoy CE . MicroRNAs: the fine-tuners of Toll-like receptor signalling. Nat Rev Immunol 2011; 11: 163–175.
del Fresno C, Soler-Rangel L, Soares-Schanoski A, Gomez-Pina V, Gonzalez-Leon MC, Gomez-Garcia L et al. Inflammatory responses associated with acute coronary syndrome up-regulate IRAK-M and induce endotoxin tolerance in circulating monocytes. J Endotoxin Res 2007; 13: 39–52.
Acknowledgements
We thank Salomea Hirschorn, Department of Rheumatology, Frederiksberg Hospital, Vibeke Østergaard Thomsen, International Reference Laboratory of Mycobacteriology, Statens Serum Institut; Marianne Kragh Thomsen, Department of Clinical Microbiology, Aarhus University Hospital, Aarhus, Denmark; and Hans Jürgen Hoffmann, Department of Respiratory Diseases, Aarhus University Hospital, Aarhus, Denmark, for collecting samples; Ewa Kogutowska and Mette Errebo Rønne, Department of Autoimmunology and Biomarkers, Statens Serum Institut, Copenhagen, Denmark, for laboratory support; and Niels Steen Krogh, Zitelab Aps, Copenhagen, Denmark for database management. This study was supported by the Danish Rheumatism Association, R95-A1913/R99-A1923/R122-A3037 (www.gigtforeningen.dk) and Region of Southern Denmark’s PhD Fund, 12/7725 (www.regionsyddanmark.dk). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
V Andersen received compensation as a consultant for MSD (Merck) and Janssen, and as a member of the advisory board for MSD (Merck). The other authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the The Pharmacogenomics Journal website
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Sode, J., Vogel, U., Bank, S. et al. Confirmation of an IRAK3 polymorphism as a genetic marker predicting response to anti-TNF treatment in rheumatoid arthritis. Pharmacogenomics J 18, 81–86 (2018). https://doi.org/10.1038/tpj.2016.66
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2016.66
This article is cited by
-
Association of P2X7 receptor genetic polymorphisms and expression with rheumatoid arthritis susceptibility in a sample of the Iranian population: a case-control study
Clinical Rheumatology (2021)
-
NFKB2 polymorphisms associate with the risk of developing rheumatoid arthritis and response to TNF inhibitors: Results from the REPAIR consortium
Scientific Reports (2020)
-
Genetically determined high activities of the TNF-alpha, IL23/IL17, and NFkB pathways were associated with increased risk of ankylosing spondylitis
BMC Medical Genetics (2018)
-
Peripheral-blood gene expression profiling studies for coronary artery disease and its severity in Xinjiang population in China
Lipids in Health and Disease (2018)
-
Associations between functional polymorphisms and response to biological treatment in Danish patients with psoriasis
The Pharmacogenomics Journal (2018)