Featured
-
-
Article
| Open Access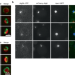 A mechanism that ensures non-selective cytoplasm degradation by autophagy
A mechanism that ensures non-selective cytoplasm degradation by autophagy
How membrane morphology is regulated during autophagosome formation remains elusive. Here, authors reveal a mechanism by which the forming autophagosomal membrane expands with a large opening for non-selective sequestration of the cytoplasm.
- Tetsuya Kotani
- , Yuji Sakai
- & Hitoshi Nakatogawa
-
Article
| Open Access Membrane compression by synaptic vesicle exocytosis triggers ultrafast endocytosis
Membrane compression by synaptic vesicle exocytosis triggers ultrafast endocytosis
The authors describe the mechanism of exo-endocytosis coupling at synapses. They find that actin forms a ring around the region of exocytosis. This ring conserves membrane area, allowing induction of inward membrane buckling following exocytosis.
- Tyler H. Ogunmowo
- , Haoyuan Jing
- & Jian Liu
-
Article
| Open Access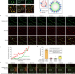 Tetraspanin 4 stabilizes membrane swellings and facilitates their maturation into migrasomes
Tetraspanin 4 stabilizes membrane swellings and facilitates their maturation into migrasomes
Migrasomes are signaling organelles that form in retraction fibers of migrating cells. Here authors show that migrasomes form by a two-stage process: an initial local membrane swelling matures and is stabilized by recruited tetraspanin proteins.
- Raviv Dharan
- , Yuwei Huang
- & Raya Sorkin
-
Article
| Open Access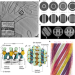 Cryo-electron tomography reveals structural insights into the membrane remodeling mode of dynamin-like EHD filaments
Cryo-electron tomography reveals structural insights into the membrane remodeling mode of dynamin-like EHD filaments
Eps15-homology domain containing proteins comprise a family of dynamin-related ATPases. Here, Melo et al. use cryo-electron tomography to determine the membrane-bound EHD4 structure, therefore clarifying the membrane binding and oligomerization mode.
- Arthur A. Melo
- , Thiemo Sprink
- & Oliver Daumke
-
Article
| Open Access The cholesterol transport protein GRAMD1C regulates autophagy initiation and mitochondrial bioenergetics
The cholesterol transport protein GRAMD1C regulates autophagy initiation and mitochondrial bioenergetics
The functions of specific lipids in autophagosome biogenesis are not entirely clear. Here, the authors show that the ER protein GRAMD1C, a cholesterol transport protein, suppresses autophagy initiation and has roles in mitochondrial cholesterol homeostasis and respiration.
- Matthew Yoke Wui Ng
- , Chara Charsou
- & Anne Simonsen
-
Article
| Open Access Actin polymerization promotes invagination of flat clathrin-coated lattices in mammalian cells by pushing at lattice edges
Actin polymerization promotes invagination of flat clathrin-coated lattices in mammalian cells by pushing at lattice edges
The role of actin filaments in clathrin-mediated endocytosis (CME) is unclear. Here, Yang et al. show that branched actin filaments promote CME by pushing on clathrin coat edges in an epsin-dependent manner, dividing large flat clathrin plaques into sizes that facilitate invagination.
- Changsong Yang
- , Patricia Colosi
- & Tatyana Svitkina
-
Article
| Open Access Imaging vesicle formation dynamics supports the flexible model of clathrin-mediated endocytosis
Imaging vesicle formation dynamics supports the flexible model of clathrin-mediated endocytosis
Despite decades of research, the dynamics of clathrin-coated vesicle formation is ambiguous. Here, authors use STAR microscopy to quantify the nanoscale dynamics of vesicle formation, supporting the flexible model of clathrin-mediated endocytosis.
- Tomasz J. Nawara
- , Yancey D. Williams II
- & Alexa L. Mattheyses
-
Article
| Open Access Lysine acetylation regulates the interaction between proteins and membranes
Lysine acetylation regulates the interaction between proteins and membranes
Lysine acetylation regulates the function of soluble proteins in vivo, yet it remains largely unexplored whether lysine acetylation regulates the function of membrane proteins. Here, the authors map lysine acetylation predominantly in membrane-interaction regions in peripheral membrane proteins and show with three candidate proteins how lysine acetylation is a regulator of membrane protein function.
- Alan K. Okada
- , Kazuki Teranishi
- & Ralf Langen
-
Article
| Open Access Subtomogram averaging of COPII assemblies reveals how coat organization dictates membrane shape
Subtomogram averaging of COPII assemblies reveals how coat organization dictates membrane shape
The COPII coat assembles in two concentric layers and mediates protein export from the endoplasmic reticulum. Here the authors present the 4.9 Å resolution cryo-tomography and subtomogram averaging structure of the membrane bound COPII inner coat that was obtained by in vitro reconstitution and discuss mechanistic implications.
- Joshua Hutchings
- , Viktoriya Stancheva
- & Giulia Zanetti
-
Article
| Open Access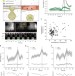 Small GTPases and BAR domain proteins regulate branched actin polymerisation for clathrin and dynamin-independent endocytosis
Small GTPases and BAR domain proteins regulate branched actin polymerisation for clathrin and dynamin-independent endocytosis
Several endocytic pathways operate simultaneously at the cell surface, including the clathrin and dynamin-independent CLIC/GEEC (CG) pathway. Here the authors show that small GTPases and BAR domain proteins regulate branched actin to make clathrin and dynamin-independent endocytic vesicles.
- Mugdha Sathe
- , Gayatri Muthukrishnan
- & Satyajit Mayor
-
Article
| Open Access Clathrin-adaptor ratio and membrane tension regulate the flat-to-curved transition of the clathrin coat during endocytosis
Clathrin-adaptor ratio and membrane tension regulate the flat-to-curved transition of the clathrin coat during endocytosis
The sequence of structural and molecular events during clathrin-mediated endocytosis is unclear. Here the authors combine correlative microscopy and simple mathematical growth laws to demonstrate that the flat patch starts to curve when around 70% of the final clathrin content is reached.
- Delia Bucher
- , Felix Frey
- & Steeve Boulant
-
Article
| Open Access Annexin A4 and A6 induce membrane curvature and constriction during cell membrane repair
Annexin A4 and A6 induce membrane curvature and constriction during cell membrane repair
The role of annexins in cell membrane repair is largely undefined. Here the authors use a model lipid bilayer to show that annexin A4 induces curvature at the membrane free edge and annexin A6 induces constriction force, and find that both annexins are recruited to wound edges in cells and are required for repair.
- Theresa Louise Boye
- , Kenji Maeda
- & Jesper Nylandsted
-
Article
| Open Access Regulated membrane remodeling by Mic60 controls formation of mitochondrial crista junctions
Regulated membrane remodeling by Mic60 controls formation of mitochondrial crista junctions
The MICOS complex has an essential role in crista junction formation and mitochondrial inner membrane morphology. Here, the authors show that one of its components, Mic60, known to form contact sites between inner and outer membranes, also displays membrane-shaping activity.
- Manuel Hessenberger
- , Ralf M. Zerbes
- & Oliver Daumke
-
Article
| Open Access High cell-surface density of HER2 deforms cell membranes
High cell-surface density of HER2 deforms cell membranes
HER2 is frequently overexpressed in breast cancer in association with increased metastatic potential. Here, the authors show that HER2 overexpression causes deformation of cell membranes in a signalling-independent manner that contributes to the disease phenotype by disrupting epithelial features.
- Inhee Chung
- , Mike Reichelt
- & Mark X. Sliwkowski
-
Article
| Open Access IRSp53 senses negative membrane curvature and phase separates along membrane tubules
IRSp53 senses negative membrane curvature and phase separates along membrane tubules
The inverted-BAR domain protein IRSp53 associates with the inner leaflet of tubular membranes such as filopodia. Here, Prévostet al. demonstrate that the I-BAR domain of IRSp53 senses negative membrane curvature, and undergoes phase separation which may aid its clustering upon filopodia generation.
- Coline Prévost
- , Hongxia Zhao
- & Patricia Bassereau
-
Article
| Open Access Intrinsically disordered proteins drive membrane curvature
Intrinsically disordered proteins drive membrane curvature
Proteins that bend membranes often contain curvature-promoting structural motifs such as wedges or crescent-shaped domains. Busch et al.report that intrinsically disordered domains can also drive membrane curvature and provide evidence that steric pressure driven by protein crowding mediates this effect.
- David J. Busch
- , Justin R. Houser
- & Jeanne C. Stachowiak
-
Article
| Open Access Membrane tension controls the assembly of curvature-generating proteins
Membrane tension controls the assembly of curvature-generating proteins
BAR domain proteins are known to reshape cell membranes. Using coarse-grained molecular dynamics simulations, Simunovic and Voth demonstrate that membrane tension strongly affects the association of BAR proteins, in turn controlling their recruitment to membrane-remodelling sites.
- Mijo Simunovic
- & Gregory A. Voth
-
Article |
 Membrane curvature sensing by the C-terminal domain of complexin
Membrane curvature sensing by the C-terminal domain of complexin
Complexin binds to synaptic vesicles and inhibits spontaneous exocytosis. Here Snead et al. show that the high curvature of the vesicle membrane is important for membrane binding, and induces a conformational change that is important for complexin’s inhibitory function.
- David Snead
- , Rachel T. Wragg
- & David Eliezer
-
Article |
 A sub-nanometre view of how membrane curvature and composition modulate lipid packing and protein recruitment
A sub-nanometre view of how membrane curvature and composition modulate lipid packing and protein recruitment
Membrane curvature and lipid composition direct the binding of many peripheral membrane proteins. Here, Vanni et al. use a combination of in vitroand molecular dynamics approaches to quantify how lipid-packing defects in membranes of various composition and curvature dictate the membrane adsorption of a model lipid-binding protein.
- Stefano Vanni
- , Hisaaki Hirose
- & Romain Gautier

 Structural insight into an Arl1–ArfGEF complex involved in Golgi recruitment of a GRIP-domain golgin
Structural insight into an Arl1–ArfGEF complex involved in Golgi recruitment of a GRIP-domain golgin