Abstract
Objective:
To evaluate early changes in glycemia, insulin physiology and gut hormone responses to an easily tolerated and slowly ingested solid, low-carbohydrate mixed meal test (MMT) following laparoscopic adjustable gastric banding (LAGB) or Roux-en-Y gastric bypass (RYGB) surgery.
Subjects/Methods:
This was a prospective non-randomized study. Plasma glucose, insulin and c-peptide (to estimate hepatic insulin extraction; %HIE), incretins (GIP, aGLP-1) and pancreatic polypeptide (PP) responses to the MMT were measured at 4–8 weeks before and after surgery in obese, metabolically healthy patients (RYGB=10F or LAGB =7F/1M). Supplementary clamp data on basal endogenous glucose production (EGP) and peripheral insulin action (Rd=rate of glucose disposal) and metabolic clearance rates of insulin (MCR-INS) were available in five of the RYGB patients. Repeated measures were appropriately accounted for in the analyses.
Results:
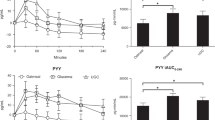
Following LAGB surgery, C-peptide and insulin MMT profiles (P=0.004 and P=0.0005, respectively) were lower with no change in %HIE (P=0.98). In contrast, in RYGB subjects, both fasting glucose and insulin (Δ=−0.66 mmol l–1, P⩽0.05 and Δ=−44.4 pmol l–1, P⩽0.05, respectively) decreased, and MMT glucose (P<0.0001) and insulin (P=0.001) but not c-peptide (P= 0.69) decreased. Estimated %HIE increased at fasting (Δ=8.4%, P⩽0.05) and during MMT (P=0.0005). Early (0–20 min) prandial glucose (0.27±0.26 versus 0.006±0.21 mmol l–1, P⩽0.05) and insulin (63(48, 66) versus 18(12, 24) pmol l–1, P⩽0.05) responses increased after RYGB. RYGB altered the trajectory of prandial aGLP-1 responses (treatment × trajectory P=0.02), and PP was lower (P<0.0001). Clamp data in a subset of RYGB patients showed early improvement in basal EGP (P=0.001), and MCR-INS (P=0.015).
Conclusion:
RYGB results in distinctly different changes in plasma glucose, insulin and gut hormone response patterns to a solid, slowly ingested low-carbohydrate MMT versus LAGB. Altered nutrient delivery, along with indirect evidence for changes in hepatic and peripheral insulin physiology, are consistent with the greater early improvement in glycemia observed after RYGB versus LAGB surgery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cummings DE . Endocrine mechanisms mediating remission of diabetes after gastric bypass surgery. Int J Obes (Lond) 2009; 33: S33–S40.
Camastra S, Gastaldelli A, Mari A, Bonuccelli S, Scartabelli G, Frascerra S et al. Early and longer term effects of gastric bypass surgery on tissue-specific insulin sensitivity and beta cell function in morbidly obese patients with and without type 2 diabetes. Diabetologia 2011; 54: 2093–2102.
Pories WJ, Caro JF, Flickinger EG, Meelheim HD, Swanson MS . The control of diabetes mellitus (NIDDM) in the morbidly obese with the Greenville Gastric Bypass. Ann Surg 1987; 206: 316–323.
Rubino F, Marescaux J . Effect of duodenal-jejunal exclusion in a non-obese animal model of type 2 diabetes: a new perspective for an old disease. Ann Surg 2004; 239: 1–11.
Strader AD, Vahl TP, Jandacek RJ, Woods SC, D'Alessio DA, Seeley RJ . Weight loss through ileal transposition is accompanied by increased ileal hormone secretion and synthesis in rats. Am J Physiol Endocrinol Metab 2005; 288: E447–E453.
Wang PY, Caspi L, Lam CK, Chari M, Li X, Light PE et al. Upper intestinal lipids trigger a gut-brain-liver axis to regulate glucose production. Nature 2008; 452: 1012–1016.
Breen DM, Rasmussen BA, Kokorovic A, Wang R, Cheung GW, Lam TK . Jejunal nutrient sensing is required for duodenal-jejunal bypass surgery to rapidly lower glucose concentrations in uncontrolled diabetes. Nat Med 2012; 18: 950–955.
Lam TK . Neuronal regulation of homeostasis by nutrient sensing. Nat Med 2010; 16: 392–395.
le Roux CW, Aylwin SJ, Batterham RL, Borg CM, Coyle F, Prasad V et al. Gut hormone profiles following bariatric surgery favor an anorectic state, facilitate weight loss, and improve metabolic parameters. Ann Surg 2006; 243: 108–114.
Korner J, Bessler M, Inabnet W, Taveras C, Holst JJ . Exaggerated glucagon-like peptide-1 and blunted glucose-dependent insulinotropic peptide secretion are associated with Roux-en-Y gastric bypass but not adjustable gastric banding. Surg Obes Relat Dis 2007; 3: 597–601.
Rodieux F, Giusti V, D'Alessio DA, Suter M, Tappy L . Effects of gastric bypass and gastric banding on glucose kinetics and gut hormone release. Obesity (Silver Spring) 2008; 16: 298–305.
Bose M, Machineni S, Olivan B, Teixeira J, McGinty JJ, Bawa B et al. Superior appetite hormone profile after equivalent weight loss by gastric bypass compared to gastric banding. Obesity (Silver Spring) 2010; 18: 1085–1091.
Bradley D, Conte C, Mittendorfer B, Eagon JC, Varela JE, Fabbrini E et al. Gastric bypass and banding equally improve insulin sensitivity and beta cell function. J Clin Invest 2012; 122: 4667–4674.
Kashyap SR, Daud S, Kelly KR, Gastaldelli A, Win H, Brethauer S et al. Acute effects of gastric bypass versus gastric restrictive surgery on beta-cell function and insulinotropic hormones in severely obese patients with type 2 diabetes. Int J Obes (Lond) 2010; 34: 462–471.
Salehi M, Prigeon RL, D'Alessio DA . Gastric bypass surgery enhances glucagon-like peptide 1-stimulated postprandial insulin secretion in humans. Diabetes 2011; 60: 2308–2314.
Laferrere B, Heshka S, Wang K, Khan Y, McGinty J, Teixeira J et al. Incretin levels and effect are markedly enhanced 1 month after Roux-en-Y gastric bypass surgery in obese patients with type 2 diabetes. Diabetes Care 2007; 30: 1709–1716.
Falken Y, Hellstrom PM, Holst JJ, Naslund E . Changes in glucose homeostasis after Roux-en-Y gastric bypass surgery for obesity at day three, two months, and one year after surgery: role of gut peptides. J Clin Endocrinol Metab 2011; 96: 2227–2235.
Laferrere B, Teixeira J, McGinty J, Tran H, Egger JR, Colarusso A et al. Effect of weight loss by gastric bypass surgery versus hypocaloric diet on glucose and incretin levels in patients with type 2 diabetes. J Clin Endocrinol Metab 2008; 93: 2479–2485.
Magkos F, Bradley D, Eagon JC, Patterson BW, Klein S . Effect of Roux-en-Y gastric bypass and laparoscopic adjustable gastric banding on gastrointestinal metabolism of ingested glucose. Am J Clin Nutr 2016; 103: 61–65.
Groop PH, Melander A, Groop LC . The relationship between early insulin release and glucose tolerance in healthy subjects. Scand J Clin Lab Invest 1993; 53: 405–409.
Pratley RE, Weyer C . The role of impaired early insulin secretion in the pathogenesis of Type II diabetes mellitus. Diabetologia 2001; 44: 929–945.
Jacobsen SH, Olesen SC, Dirksen C, Jorgensen NB, Bojsen-Moller KN, Kielgast U et al. Changes in gastrointestinal hormone responses, insulin sensitivity, and beta-cell function within 2 weeks after gastric bypass in non-diabetic subjects. Obes Surg 2012; 22: 1084–1096.
Polonsky KS, Rubenstein AH . C-peptide as a measure of the secretion and hepatic extraction of insulin. Pitfalls and limitations. Diabetes 1984; 33: 486–494.
Campioni M, Toffolo G, Basu R, Rizza RA, Cobelli C . Minimal model assessment of hepatic insulin extraction during an oral test from standard insulin kinetic parameters. Am J Physiol Endocrinol Metab 2009; 297: E941–E948.
Bojsen-Moller KN, Dirksen C, Jorgensen NB, Jacobsen SH, Serup AK, Albers PH et al. Early enhancements of hepatic and later of peripheral insulin sensitivity combined with increased postprandial insulin secretion contribute to improved glycemic control after Roux-en-Y gastric bypass. Diabetes 2014; 63: 1725–1737.
Expert Committee on the D, Classification of Diabetes M. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 2003; 26: S5–S20.
Lillioja S, Mott DM, Howard BV, Bennett PH, Yki-Jarvinen H, Freymond D et al. Impaired glucose tolerance as a disorder of insulin action. Longitudinal and cross-sectional studies in Pima Indians. N Engl J Med 1988; 318: 1217–1225.
Steele R . Influences of glucose loading and of injected insulin on hepatic glucose output. Ann N Y Acad Sci 1959; 82: 420–430.
Lillioja S, Bogardus C . Obesity and insulin resistance: lessons learned from the Pima Indians. Diabetes Metab Rev 1988; 4: 517–540.
Best JD, Taborsky GJ Jr., Halter JB, Porte D Jr . Glucose disposal is not proportional to plasma glucose level in man. Diabetes 1981; 30: 847–850.
Wittgrove AC, Clark GW . Laparoscopic gastric bypass, Roux-en-Y- 500 patients: technique and results, with 3-60 month follow-up. Obes Surg 2000; 10: 233–239.
Varela JE, Hinojosa MW, Nguyen NT . Perioperative outcomes of bariatric surgery in adolescents compared with adults at academic medical centers. Surg Obes Relat Dis 2007; 3: 537–540.
Hansen EN, Tamboli RA, Isbell JM, Saliba J, Dunn JP, Marks-Shulman PA et al. Role of the foregut in the early improvement in glucose tolerance and insulin sensitivity following Roux-en-Y gastric bypass surgery. Am J Physiol Gastrointest Liver Physiol 2011; 300: G795–G802.
Zalesin KC, Franklin BA, Lillystone MA, Shamoun T, Krause KR, Chengelis DL et al. Differential loss of fat and lean mass in the morbidly obese after bariatric surgery. Metab Syndr Relat Disord 2010; 8: 15–20.
Levitt DG, Beckman LM, Mager JR, Valentine B, Sibley SD, Beckman TR et al. Comparison of DXA and water measurements of body fat following gastric bypass surgery and a physiological model of body water, fat, and muscle composition. J Appl Physiol (1985) 2010; 109: 786–795.
Pattou F, Beraud G, Arnalsteen L, Seguy D, Pigny P, Fermont C et al. Restoration of beta cell function after bariatric surgery in type 2 diabetic patients: a prospective controlled study comparing gastric banding and gastric bypass. Obes Surg 2007; 17: 1041–1043.
Lee W, Lee Y-C, Chen J-C, Ser K-H, Chen S-C, Lin C-M . PL-03: A randomized trial comparing laparoscopic sleeve gastrectomy versus gastric bypass for the treatment of type 2 diabetes mellitus: Preliminary report. Surg Obes Relat Dis 2008; 4: 290.
Plum L, Ahmed L, Febres G, Bessler M, Inabnet W, Kunreuther E et al. Comparison of glucostatic parameters after hypocaloric diet or bariatric surgery and equivalent weight loss. Obesity (Silver Spring) 2011; 19: 2149–2157.
Bojsen-Moller KN, Dirksen C, Jorgensen NB, Jacobsen SH, Hansen DL, Worm D et al. Increased hepatic insulin clearance after Roux-en-Y gastric bypass. J Clin Endocrinol Metab 2013; 98: E1066–E1071.
Chandarana K, Batterham RL . Shedding pounds after going under the knife: metabolic insights from cutting the gut. Nat Med 2012; 18: 668–669.
Sarruf DA, Bonner-Weir S, Schwartz MW . New clues to bariatric surgery's benefits. Nat Med 2012; 18: 860–861.
Salinari S, Carr RD, Guidone C, Bertuzzi A, Cercone S, Riccioni ME et al. Nutrient infusion bypassing duodenum-jejunum improves insulin sensitivity in glucose-tolerant and diabetic obese subjects. Am J Physiol Endocrinol Metab 2013; 305: E59–E66.
Schwartz TW . Pancreatic polypeptide: a hormone under vagal control. Gastroenterology 1983; 85: 1411–1425.
Campos GM, Rabl C, Havel PJ, Rao M, Schwarz JM, Schambelan M et al. Changes in post-prandial glucose and pancreatic hormones, and steady-state insulin and free fatty acids after gastric bypass surgery. Surg Obes Relat Dis 2014; 10: 1–8.
Schmidt PT, Naslund E, Gryback P, Jacobsson H, Holst JJ, Hilsted L et al. A role for pancreatic polypeptide in the regulation of gastric emptying and short-term metabolic control. J Clin Endocrinol Metab 2005; 90: 5241–5246.
Kahleova H, Mari A, Nofrate V, Matoulek M, Kazdova L, Hill M et al. Improvement in beta-cell function after diet-induced weight loss is associated with decrease in pancreatic polypeptide in subjects with type 2 diabetes. J Diabetes Complications 2012; 26: 442–449.
Promintzer-Schifferl M, Prager G, Anderwald C, Mandl M, Esterbauer H, Shakeri-Leidenmuhler S et al. Effects of gastric bypass surgery on insulin resistance and insulin secretion in nondiabetic obese patients. Obesity (Silver Spring) 2011; 19: 1420–1426.
Dunn JP, Abumrad NN, Breitman I, Marks-Shulman PA, Flynn CR, Jabbour K et al. Hepatic and peripheral insulin sensitivity and diabetes remission at 1 month after Roux-en-Y gastric bypass surgery in patients randomized to omentectomy. Diabetes Care 2012; 35: 137–142.
Immonen H, Hannukainen JC, Iozzo P, Soinio M, Salminen P, Saunavaara V et al. Effect of bariatric surgery on liver glucose metabolism in morbidly obese diabetic and non-diabetic patients. J Hepatol 2014; 60: 377–383.
de Weijer BA, Aarts E, Janssen IM, Berends FJ, van de Laar A, Kaasjager K et al. Hepatic and peripheral insulin sensitivity do not improve 2 weeks after bariatric surgery. Obesity (Silver Spring) 2013; 21: 1143–1147.
Campos GM, Rabl C, Peeva S, Ciovica R, Rao M, Schwarz JM et al. Improvement in peripheral glucose uptake after gastric bypass surgery is observed only after substantial weight loss has occurred and correlates with the magnitude of weight lost. J Gastrointest Surg 2010; 14: 15–23.
Lima MM, Pareja JC, Alegre SM, Geloneze SR, Kahn SE, Astiarraga BD et al. Acute effect of roux-en-y gastric bypass on whole-body insulin sensitivity: a study with the euglycemic-hyperinsulinemic clamp. J Clin Endocrinol Metab 2010; 95: 3871–3875.
Viljanen AP, Iozzo P, Borra R, Kankaanpaa M, Karmi A, Lautamaki R et al. Effect of weight loss on liver free fatty acid uptake and hepatic insulin resistance. J Clin Endocrinol Metab 2009; 94: 50–55.
Wallace TM, Matthews DR . The assessment of insulin resistance in man. Diabetes Med 2002; 19: 527–534.
Acknowledgements
The authors acknowledge the contributions of the clinical and dietary staffs of the Scottsdale Bariatric Center and Phoenix NIDDK Obesity and Diabetes Clinical Research Unit. We are indebted to the bariatric surgery patients who participated as research volunteers. The study was funded by Intramural Research funding of NIDDK.
Author contributions
JB wrote the study design, collected metabolic data, did statistical analysis and wrote the manuscript. RB wrote the study design, performed the surgeries and wrote/edited the manuscript. MST assisted on statistical analysis, and reviewed/edited the manuscript. KLV wrote/edited the manuscript. SV designed and supervised meal administration, wrote/edited the manuscript. JK wrote the study design, assisted with statistical analysis and reviewed/edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Bunt, J., Blackstone, R., Thearle, M. et al. Changes in glycemia, insulin and gut hormone responses to a slowly ingested solid low-carbohydrate mixed meal after laparoscopic gastric bypass or band surgery. Int J Obes 41, 706–713 (2017). https://doi.org/10.1038/ijo.2017.22
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.22
This article is cited by
-
Laparoscopic adjustable gastric banding with liraglutide in adults with obesity and type 2 diabetes (GLIDE): a pilot randomised placebo controlled trial
International Journal of Obesity (2023)
-
Length of biliopancreatic limb in Roux-en-Y gastric bypass and its impact on post-operative outcomes in metabolic and obesity surgery—systematic review and meta-analysis
International Journal of Obesity (2022)
-
Impact of Vagotomy on Postoperative Weight Loss, Alimentary Intake, and Enterohormone Secretion After Bariatric Surgery in Experimental Translational Models
Obesity Surgery (2022)
-
Potential Hormone Mechanisms of Bariatric Surgery
Current Obesity Reports (2017)