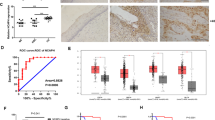
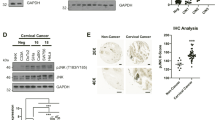
Abstract
Human epithelial cells can be infected by more than 200 types of human papilloma viruses (HPVs), and persistent HPV infections lead to cervical cancer or other deadly cancers. It has been established that mitotic progression is critical for HPV16 infection, but the underlying mechanism remains unknown. Here, we report that oncoprotein E7 of HPV16 but not HPV18 retards mitotic progression in host cell by direct binding to the C terminus of Microtubule-Associated Protein 4 (MAP4). MAP4 is a novel bona fide target of HPV16E7 protein which binds and recruits the latter to spindle microtubule in mitosis. HPV16E7 protein promotes MAP4 stability by inhibiting MAP4 phosphorylation- mediated degradation to increase the stability of microtubule polymerization and cause an extend mitotic progression. We further uncovered that Mps1 is a kinase of MAP4, and E7-MAP4 binding blocks Mps1 phosphorylation of MAP4, thereby interrupting phosphorylation-dependent MAP4 degradation. Mutations of MAP4 at T927ES928E partially abolished E7-binding capacity and rescued mitotic progression in host cells. In conclusion, our study reveals a molecular mechanism by which HPV16E7 perturbs host mitotic progression by interfering Mps1-MAP4 signaling cascade, which results in an extended infection window and may facilitate the persistent HPV16 infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Muñoz N, Bosch FX, de Sanjosé S, Herrero R, Castellsagué X, Shah KV, et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med. 2003;348:518–27.
Frisch M, Biggar RJ. Aetiological parallel between tonsillar and anogenital squamous-cell carcinomas. Lancet. 1999;354:1442–3.
Marur S, D’Souza G, Westra WH, Forastiere AA. HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol. 2010;11:781–9.
Shaw R, Robinson M. The increasing clinical relevance of human papillomavirus type 16 (HPV-16) infection in oropharyngeal cancer. Br J Oral Maxillofac Surg. 2011;49:423–9.
Bosch FX, Burchell AN, Schiffman M, Giuliano AR, de Sanjose S, Bruni L, et al. Epidemiology and natural history of human papillomavirus infections and type-specific implications in cervical neoplasia. Vaccine. 2008;26(Suppl 10):K1–16.
Koshiol J, Lindsay L, Pimenta JM, Poole C, Jenkins D, Smith JS. Persistent human papillomavirus infection and cervical neoplasia: a systematic review and meta-analysis. Am J Epidemiol. 2008;168:123–37.
Bosch FX, Muñoz N. The viral etiology of cervical cancer. Virus Res. 2002;89:180–90.
Mirabello L, Yeager M, Yu K, Clifford GM, Xiao Y, Zhu B, et al. HPV16 E7 genetic conservation is critical to carcinogenesis. Cell. 2017;170:1164–74.
Pyeon D, Pearce SM, Lank SM, Ahlquist P, Lambert PF. Establishment of human papillomavirus infection requires cell cycle progression. PLoS Pathog. 2009;5:e1000318.
DiGiuseppe S, Luszczek W, Keiffer TR, Bienkowska-Haba M, Guion LGM, Sapp MJ. Incoming human papillomavirus type 16 genome resides in a vesicular compartment throughout mitosis. Proc Natl Acad Sci USA. 2016;113:6289–94.
Burger MP, van Leeuwen AM, Hollema H, Quint WG, Pieters WJ. Human papillomavirus type influences the extent of chromosomal lag during mitosis in cervical intraepithelial neoplasia grade III. Int J Gynecol Pathol. 1997;16:10–4.
Thomas LK, Bermejo JL, Vinokurova S, Jensen K, Bierkens M, Steenbergen R, et al. Chromosomal gains and losses in human papillomavirus-associated neoplasia of the lower genital tract—a systematic review and meta-analysis. Eur J Cancer. 2014;50:85–98.
Espinosa AM, Alfaro A, Roman-Basaure E, Guardado-Estrada M, Palma Í, Serralde C, et al. Mitosis is a source of potential markers for screening and survival and therapeutic targets in cervical cancer. PLoS ONE. 2013;8:e55975.
Steinmann KE, Pei XF, Stoppler H, Schlegel R, Schlegel R. Elevated expression and activity of mitotic regulatory proteins in human papillomavirus-immortalized keratinocytes. Oncogene. 1994;9:387–94.
Kremer BE, Haystead T, Macara IG. Mammalian septins regulate microtubule stability through interaction with the microtubule-binding protein MAP4. Mol Biol Cell. 2005;16:4648–59.
Takahashi M, Shiraishi H, Ishibashi Y, Blade KL, McDermott PJ, Menick DR, et al. Phenotypic consequences of beta1-tubulin expression and MAP4 decoration of microtubules in adult cardiocytes. Am J Physiol Heart Circ Physiol. 2003;285:H2072–83.
Holmfeldt P, Brattsand G, Gullberg M. MAP4 counteracts microtubule catastrophe promotion but not tubulin-sequestering activity in intact cells. Curr Biol. 2002;12:1034–9.
Zahnleiter D, Hauer NN, Kessler K, Uebe S, Sugano Y, Neuhauss SC, et al. MAP4-dependent regulation of microtubule formation affects centrosome, cilia, and Golgi architecture as a central mechanism in growth regulation. Hum Mutat. 2015;36:87–97.
Jiang YY, Shang L, Shi ZZ, Zhang TT, Ma S, Lu CC, et al. Microtubule-associated protein 4 is an important regulator of cell invasion/migration and a potential therapeutic target in esophageal squamous cell carcinoma. Oncogene. 2016;35:4846–56.
Ou Y, Zheng X, Gao Y, Shu M, Leng T, Li Y, et al. Activation of cyclic AMP/PKA pathway inhibits bladder cancer cell invasion by targeting MAP4-dependent microtubule dynamics. Urol Oncol. 2014;32:21–8.
Gallo DE, Hope TJ. Knockdown of MAP4 and DNAL1 produces a post-fusion and pre-nuclear translocation impairment in HIV-1 replication. Virology. 2012;422:13–21.
Abrieu A, Magnaghi-Jaulin L, Kahana JA, Peter M, Castro A, Vigneron S, et al. Mps1 is a kinetochore-associated kinase essential for the vertebrate mitotic checkpoint. Cell. 2001;106:83–93.
Lauzé E, Stoelcker B, Luca FC, Weiss E, Schutz AR, Winey M. Yeast spindle pole body duplication gene MPS1 encodes an essential dual specificity protein kinase. EMBO J. 1995;14:1655–63.
Maciejowski J, Drechsler H, Grundner-Culemann K, Ballister ER, Rodriguez-Rodriguez JA, Rodriguez-Bravo V, et al. Mps1 regulates kinetochore-microtubule attachment stability via the ska complex to ensure error-free chromosome segregation. Dev Cell. 2017;41:143–56.
Hiruma Y, Sacristan C, Pachis ST, Adamopoulos A, Kuijt T, Ubbink M, et al. Competition between MPS1 and microtubules at kinetochores regulates spindle checkpoint signaling. Science. 2015;348:1264–7.
West RR, Tenbarge KM, Olmsted JB. A model for microtubule-associated protein 4 structure. Domains defined by comparisons of human, mouse, and bovine sequences. J Biol Chem. 1991;266:21886–96.
Piperno G, LeDizet M, Chang XJ. Microtubules containing acetylated alpha-tubulin in mammalian cells in culture. J Cell Biol. 1987;104:289–302.
Mandelkow E, Mandelkow EM. Microtubules and microtubule-associated proteins. Curr Opin Cell Biol. 1995;7:72–81.
Andersen SS. Spindle assembly and the art of regulating microtubule dynamics by MAPs and Stathmin/Op18. Trends Cell Biol. 2000;10:261–7.
Dou Z, Sawagechi A, Zhang J, Luo H, Brako L, Yao XB. Dynamic distribution of TTK in HeLa cells: insights from an ultrastructural study. Cell Res. 2003;13:443–9.
Xu Q, Zhu S, Wang W, Zhang X, Old W, Ahn N, et al. Regulation of kinetochore recruitment of two essential mitotic spindle checkpoint proteins by Mps1 phosphorylation. Mol Biol Cell. 2009;20:10–20.
Holmfeldt P, Brattsand G, Gullberg M. Interphase and monoastral-mitotic phenotypes of overexpressed MAP4 are modulated by free tubulin concentrations. J Cell Sci. 2003;116:3701–11.
Acknowledgements
This work was supported in part by grants from National Basic Research Program of China (2014CB138505) and National Natural Science Foundation of China (81871130).
Author information
Authors and Affiliations
Contributions
Y.G. and X.Z. performed the majority of experiments, analyzed the data, and prepared the figures. Q.X. performed the live-cell imaging experiment. F.N., W.Z., and J.L. assisted in the experiments and data analysis. F.G. and X.S. performed histological analysis. C.L., R.H., and J.T. constructed the plasmids. R.L., L.Z., and L.C. performed manuscript proofreading. R.Z.M. directed the project. Y.G., X.Z., and R.Z.M wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, Y., Zhang, X., Xu, Q. et al. Human Papillomavirus 16 oncoprotein E7 retards mitotic progression by blocking Mps1-MAP4 signaling cascade. Oncogene 38, 5959–5970 (2019). https://doi.org/10.1038/s41388-019-0851-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-019-0851-1