Abstract
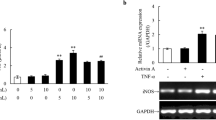
We have previously demonstrated that transforming growth factor-β (TGF-β) and pro-inflammatory cytokines, such as tumor necrosis factor-α (TNF-α) or interleukin-1β, synergistically enhance the expression of type VII collagen gene (COL7A1) in human dermal fibroblasts in culture (Mauviel et al., 1994). Recently, we identified a SMAD-containing complex, rapidly induced by TGF-β and binding the region [−496/−444] of the COL7A1 promoter, responsible for COL7A1 gene transactivation (Vindevoghel et al., 1998a). In this report, we demonstrate that TGF-β and TNF-α response elements are distinct entities within the COL7A1 promoter. In particular, we demonstrate that the TNF-α effect is mediated by NF-κB1/RelA (p50/p65) and RelA/RelA (p65/p65) NF-κB complexes binding the TNF-α response element (TaRE) located in the region [−252/−230], with RelA acting as the transcriptional activator. Finally, we provide definitive evidence for the role of both TGF-β and TNF-α response elements as enhancer sequences, functioning in the context of a heterologous promoter in an additive manner in response to TGF-β and TNF-α. This study provides the first identification of a functional interaction between the two immediate-early transcription factors, SMAD and NF-κB, to activate the expression of an extracellular matrix-related gene, COL7A1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Alevizopoulos A and Mermod N. . 1996 J. Biol. Chem. 271: 29672–29678.
Andrews NC and Faller DV. . 1991 Nucl. Acids Res. 19: 2499.
Baker JC and Harland RM. . 1997 Curr. Opin. Genet. Dev. 7: 467–473.
Burgeson RE. . 1993 J. Invest. Dermatol. 10: 252–255.
Chen YQ, Mauviel A and Uitto J. . 1994a J. Geriatric Derm. 2: 163–169.
Chen YQ, Mauviel A, Ryynänen J, Sollberg S and Uitto J. . 1994b J. Invest. Dermatol. 102: 205–209.
Christiano AM and Uitto J. . 1996 Curr. Opin. Dermatol. 3: 225–232.
Chung KY, Agarwal A, Uitto J and Mauviel A. . 1996 J. Biol. Chem. 271: 3272–3278.
Duckett CS, Perkins ND, Kowalik TF, Schmid RM, Huang ES, Baldwin Jr AS and Nabel GJ. . 1993 Mol. Cell. Biol. 13: 1315–1322.
Gorman CM, Moffat LF and Howard BH. . 1982 Mol. Cell. Biol. 2: 1044–1051.
Graham F and Van der Eb A. . 1973 Virology 54: 536–539.
Heldin CH, Miyazono K and ten Dijke P. . 1997 Nature 390: 465–471.
Helenius M, Hänninen M, Lehtinen SK and Salminen A. . 1996 Biochem. J. 318: 603–608.
Inagaki Y, Truter S, Tanaka S, Di Liberto M and Ramirez F. . 1995 J. Biol. Chem. 270: 3353–3358.
Iwasaki T, Lapière JC, Uitto J, Kim YH and Woodley DT. . 1993 Biochem. Biophys. Res. Commun. 193: 604–610.
Kouba DJ, Chung KY, Vindevoghel L, Uitto J and Mauviel A. . 1997 J. Invest. Dermatol. 108: 586, abstr. 292.
Lanoix J, Lacoste J, Pèpin N, Rice NR and Hiscott J. . 1994 Oncogene 9: 841–852.
Lyakh L, Ghosh P and Rice NR. . 1997 Mol. Cell. Biol. 17: 2475–2484.
Massaguè J. . 1996 Cell 85: 947–950.
Mauviel A and Uitto J. . 1993 Wounds 5: 137–152.
Mauviel A, Chen YQ, Dong W, Evans CH and Uitto J. . 1993 Curr. Biol. 3: 822–831.
Mauviel A, Lapière JC, Halcin C, Evans CH and Uitto J. . 1994 J. Biol. Chem. 269: 25–28.
Rice NR, MacKichan ML and Israel A. . 1992 Cell 71: 243–253.
Sambrook J, Fritsch E and Maniatis T. . 1989 Molecular cloning: a laboratory manual. 2nd edition Cold Spring Harbor Laboratory Press: Cold Spring Harbor, New York.
Siebenlist U, Franzoso G and Brown K. . 1994 Ann. Rev. Cell Biol. 10: 405–455.
Tan TH, Huang GP, Sica A, Ghosh P, Young HA, Longo DL and Rice NR. . 1992 Mol. Cell. Biol. 12: 4067–4075.
Trebilcock GU and Ponnanppan U. . 1996 Gerontology 42: 137–146.
Uitto J and Christiano AM. . 1998 In: Principles of molecular medicine. Jameson JL, (ed.). Humana Press: Totowa, NJ. pp.729–734.
Varga J, Rosenbloom J and Jimenez SA. . 1987 Biochem. J. 247: 597–604.
Vindevoghel L, Chung KY, Davis A, Kouba DJ, Kivirikko S, Alder H, Uitto J and Mauviel A. . 1997 J. Biol. Chem. 272: 10196–10204.
Vindevoghel L, Kon A, Lechleider RJ, Uitto J, Roberts AB and Mauviel A. . 1998a J. Biol. Chem. 273: 13053–13057.
Vindevoghel L, Lechleider RJ, Kon A, de Caestecker MP, Uitto J, Roberts AB and Mauviel A. . 1998b Proc. Natl. Acad. Sci. USA in press.
Woodley DT, Zelickson AS, Briggaman RA, Hamilton TA, Weiss JS, Ellis CN and Voorhees JJ. . 1990 JAMA 263: 3057–3059.
Acknowledgements
We thank Dr Nancy Rice (National Cancer Institute, Frederick, MD) for all Rel and NF-AT antibodies. The expert technical assistance of Ying-Jie Song is greatly acknowledged. This work was supported in part by National Institutes of Health grants R29-AR43751 (to AM), RO1-AR41439 and T32-AR07651 (to JU), and by a Dermatology Foudnation Research Fellowship (LV).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kon, A., Vindevoghel, L., Kouba, D. et al. Cooperation between SMAD and NF-κB in growth factor regulated type VII collagen gene expression. Oncogene 18, 1837–1844 (1999). https://doi.org/10.1038/sj.onc.1202495
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202495
Keywords
This article is cited by
-
The pathogenesis of renal injury and treatment in light chain deposition disease
Journal of Translational Medicine (2019)
-
XIAP gene expression and function is regulated by autocrine and paracrine TGF-β signaling
Molecular Cancer (2010)
-
Signaling cross-talk between TGF-β/BMP and other pathways
Cell Research (2009)
-
Smads mediate signaling of the TGFβ superfamily in normal keratinocytes but are lost during skin chemical carcinogenesis
Oncogene (2001)
-
Monoclonal Light Chain–Mesangial Cell Interactions: Early Signaling Events and Subsequent Pathologic Effects
Laboratory Investigation (2001)