Abstract
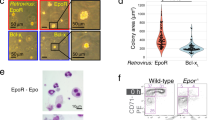
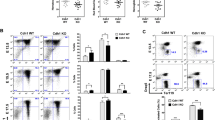
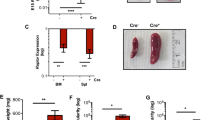
Primary erythroid progenitors can be expanded by the synergistic action of erythropoietin (Epo), stem cell factor (SCF) and glucocorticoids. While Epo is required for erythropoiesis in general, glucocorticoids and SCF mainly contribute to stress erythropoiesis in hypoxic mice. This ability of normal erythroid progenitors to undergo expansion under stress conditions is targeted by the avian erythroblastosis virus (AEV), harboring the oncogenes v-ErbB and v-ErbA. We investigated the signaling pathways required for progenitor expansion under stress conditions and in leukemic transformation. Immortal strains of erythroid progenitors, able to undergo normal, terminal differentiation under appropriate conditions, were established from fetal livers of p53−/− mice. Expression and activation of the EGF-receptor (HER-1/c-ErbB) or its mutated oncogenic version (v-ErbB) in these cells abrogated the requirement for Epo and SCF in expansion of these progenitors and blocked terminal differentiation. Upon inhibition of ErbB function, differentiation into erythrocytes occurred. Signal transducing molecules important for renewal induction, i.e. Stat5- and phosphoinositide 3-kinase (PI3K), are utilized by both EpoR/c-Kit and v/c-ErbB. However, while v-ErbB transformed cells and normal progenitors depended on PI3K signaling for renewal, c-ErbB also induces progenitor expansion by PI3K-independent mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bauer A, Tronche F, Wessely O, Kellendonk C, Reichardt HM, Steinlein P, Schütz G, Beug H . 1999 Genes Dev. 13: 2996–3002
Bauer A, Gandrillon O, Samarut J, Beug H . 2001 Hematopoiesis: a developmental approach. In Zon, L. (ed. Oxford University Press, Oxford in press
Beug H, Palmieri S, Freudenstein C, Zentgraf H, Graf T . 1982 Cell 28: 907–919
Beug H, Kahn P, Doederlein G, Hayman MJ, Graf T . 1985 Modern Trends in Human Leukaemia VI. Neth R, Gallo R, Greaves M and Janka K . (eds).. Springer Verlag, Berlin, Heidelberg 29: pp. 284–289
Beug H, Bartunek P, Steinlein P, Hayman MJ . 1995 Oncogene Techniques. Vogt, P.K. and Verma, I.M. (eds) Academic Press Inc., New York and London 254: pp. 41–76
Beug H, Bauer A, Dolznig H, von Lindern M, Lobmayer L, Mellitzer G, Steinlein P, Wessely O, Mullner E . 1996 Biochim. Biophys. Acta. 1288: M35–M47
Broudy VC, Lin NL, Priestley GV, Nocka K, Wolf NS . 1996 Blood 88: 75–81
Broudy VC, Lin NL, Buhring HJ, Komatsu N, Kavanagh TJ . 1998 Blood 91: 898–906
Dolznig H, Bartunek P, Nasmyth K, Müllner E, Beug H . 1995 Cell Growth Differ. 6: 1341–1352
Dolznig H, Boulmé F, Stangl K, Deiner EM, Mikulits W, Beug H, Müllner E . 2001 FASEB J. in press
Downward J, Yarden Y, Mayes E, Scrace G, Totty N, Stockwell P, Ullrich A, Schlessinger J, Waterfield MD . 1984 Nature 307: 521–527
Fry DW, Kraker AJ, McMichael A, Ambroso LA, Nelson JE, Leopold WR, Connors RW, Bridges AJ . 1994 Science 265: 1093–1095
Gandrillon O, Schmidt U, Beug H, Samarut J . 1999 EMBO J. 18: 2764–2781
Hayman MJ, Meyer S, Martin F, Steinlein P, Beug H . 1993 Cell 74: 157–169
Hinds PW, Weinberg RA . 1994 Curr. Opin. Genet. Dev. 4: 135–141
Keller G . 1992 Curr. Opin. Immunol. 4: 133–139
Khazaie K, Dull TJ, Graf T, Schlessinger J, Ullrich A, Beug H, Vennstrom B . 1988 EMBO J. 7: 3061–3071
Kieslinger M, Woldman I, Moriggl R, Hofmann J, Marine JC, Ihle JN, Beug H, Decker T . 2000 Genes Dev. 14: 232–244
Kops GJ, Burgering BM . 1999 J. Mol. Med. 77: 656–665
Kowenz E, Leutz A, Doderlein G, Graf T, Beug H . 1987 Modern Trends in Human Leukemia VII. Neth R, Gallo RC, Greaves MF and Kabisch H (eds.. Springer Verlag, Heidelberg 31: pp. 199–209
Lecoq-Lafon C, Verdier F, Fichelson S, Chretien S, Gisselbrecht S, Lacombe C, Mayeux P . 1999 Blood 93: 2578–2585
Lemmon MA, Ferguson KM . 2000 Biochem. J. 350: 1–18
Levy DE, Gilliland DG . 2000 Oncogene 19: 2505–2510
Mellitzer G, Wessely O, Decker T, Eilers A, Hayman MJ, Beug H . 1996 Proc. Natl. Acad. Sci. USA 93: 9600–9605
Metz T, Harris AW, Adams JM . 1995 Cell 82: 29–36
Meyer S, LaBudda K, McGlade I, Hayman MJ . 1994 Mol. Cell Biol. 14: 3253–3262
Muta K, Krantz SB, Bondurant MC, Dai CH . 1995 Blood 86: 572–580
Nishigaki K, Hanson C, Ohashi T, Thompson D, Muszynski K, Ruscetti S . 2000 J. Virol. 74: 3037–3045
Nutt SL, Heavey B, Rolink AG, Busslinger M . 1999 Nature 401: 556–563
Ozbun MA, Medina D, Butel JS . 1993 Growth Differ. 4: 811–819
Pain B, Melet F, Saez J, Flickinger T, Raines M, Peyrol S, Moscovici C, Moscovici MG, Kung HJ, Jurdic P, Lazarides F, Samarut J . 1991 Cell 65: 37–46
Reichardt HM, Kaestner KH, Tuckermann J, Kretz O, Wessely O, Bock R, Gass P, Schmid W, Herrlich P, Angel P, Schütz G . 1998 Cell 93: 531–541
Rolink A, Kudo A, Karasyama H, Kikuchi Y . 1991 EMBO J. 10: 327–336
Steinlein P, Wessely O, Meyer S, Deiner EM, Hayman MJ, Beug H . 1995 Curr. Biol. 5: 191–204
Sui X, Krantz SB, You M, Zhao Z . 1998 Blood 92: 1142–1149
Tamir A, Howard J, Higgins RR, Li YJ, Berger L, Zacksenhaus E, Reis M, Ben-David Y . 1999 Mol. Cell. Biol. 19: 4452–4464
Teglund S, McKay C, Schuetz E, van Deursen JM, Stravopodis D, Wang D, Brown M, Bodner S, Grosveld G, and Ihle JN . 1998 Cell 93: 841–850
Till JE, McCulloch EA . 1980 Biochim. Biophys. Acta. 605: 431–459
van Dijk T, Van den Akker E, Parren-van Amelsvoort M, Mano H, Lowenberg B, von Lindern M . 2000 Blood 96: 3406–3413
von Lindern M, Zauner W, Steinlein P, Mellitzer G, Fritsch G, Huber K, Löwenberg B, Beug H . 1999 Blood 94: 550–559
von Lindern M, Amelsvoort MP, van Dijk T, Deiner E, van Den Akker E, van Emst-De Vries S, Willems P, Beug H, Lowenberg B . 2000 J. Biol. Chem. 275: 34719–34727
von Rueden T, Kandels S, Radaszkiewicz T, Ullrich A, Wagner EF . 1992 Blood 79: 3145–3158
Wessely O, Deiner EM, Beug H, von Lindern M . 1997a EMBO J. 16: 267–280
Wessely O, von Lindern M, Levitzki A, Gazit A, Ischenko I, Mellitzer G, Hayman MJ, Beug H . 1997b Cell. Growth Diff. 8: 481–493
Wessely O, Bauer A, Quang CT, Deiner EM, von Lindern M, Mellitzer G, Steinlein P, Ghysdael J, Beug H . 1999 A novel way to induce erythroid progenitor self renewal: cooperation of c-Kit with the erythropoietin receptor Biol. Chem. 380: 187–202
Wong KS, Li YJ, Howard J, Ben-David Y . 1999 Loss of p53 in F-MuLV induced-erythroleukemias accelerates the acquisition of mutational events that confers immortality and growth factor independence Oncogene 18: 5525–5534
Wu H, Klingmuller U, Besmer P, Lodish HF . 1995a nteraction of the erythropoietin and stem-cell-factor receptors Nature 377: 242–246
Wu H, Liu X, Jaenisch R, Lodish HF . 1995b Cell 83: 59–67
Zochodne B, Truong AH, Stetler K, Higgins RR, Howard J, Dumont D, Berger SA, Ben-David Y . 2000 Oncogene 19: 2296–2304
Acknowledgements
We thank Gabi Litos and Gabi Stengl for expert technical assistance. This work was supported by grants from the Austrian Forschungsförderungs fonds (FFF, Project No. 802447, HB), the Austrian Science Foundation (FWF, SFB006, HB, EM), the Dutch Cancer Society (EUR 95-1021), the European Community (HPRN-CT-2000-00083, HB, MvL and ERBFMRX-CT-98-0197, HB, EM) and by a fellowship of the Dutch Academy for Arts and Sciences (KNAW) to MvL.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
von Lindern, M., Deiner, E., Dolznig, H. et al. Leukemic transformation of normal murine erythroid progenitors: v- and c-ErbB act through signaling pathways activated by the EpoR and c-Kit in stress erythropoiesis. Oncogene 20, 3651–3664 (2001). https://doi.org/10.1038/sj.onc.1204494
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204494
Keywords
This article is cited by
-
Decreased PGC1β expression results in disrupted human erythroid differentiation, impaired hemoglobinization and cell cycle exit
Scientific Reports (2021)
-
A tissue-specific self-interacting chromatin domain forms independently of enhancer-promoter interactions
Nature Communications (2018)
-
Macrophage migration inhibitory factor is an endogenous regulator of stress-induced extramedullary erythropoiesis
Histochemistry and Cell Biology (2016)
-
Kit transduced signals counteract erythroid maturation by MAPK-dependent modulation of erythropoietin signaling and apoptosis induction in mouse fetal liver
Cell Death & Differentiation (2015)
-
p38α suppresses normal and cancer cell proliferation by antagonizing the JNK–c-Jun pathway
Nature Genetics (2007)