Abstract
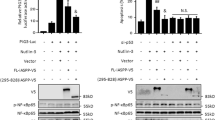
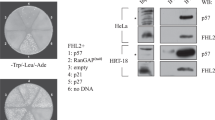
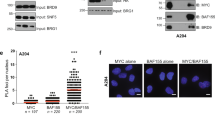
HIPK2 shows overlapping localization with p53 in promyelocytic leukemia (PML) nuclear bodies (PML-NBs) and functionally interacts with p53 to increase gene expression. Here we demonstrate that HIPK2 and the PML-NB resident protein Sp100 synergize for the activation of p53-dependent gene expression. Sp100 and HIPK2 interact and partially colocalize in PML-NBs. The cooperation of HIPK2 and Sp100 for the induction of p21Waf1 is completely dependent on the presence of p53 and the kinase function of HIPK2. Downregulation of Sp100 levels by expression of siRNA does not interfere with p53-mediated transcription, but obviates the enhancing effect of HIPK2. In summary, these experiments reveal a novel function for Sp100 as a coactivator for HIPK2-mediated p53 activation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bloch DB, Nakajima A, Gulick T, Chiche JD, Orth D, de La Monte SM and Bloch KD . (2000). Mol. Cell. Biol., 20, 6138–6146.
Borden KL . (2002). Mol. Cell. Biol., 22, 5259–5269.
Brummelkamp TR, Bernards R and Agami R . (2002). Science, 296, 550–553.
Choi CY, Kim YH, Kwon HJ and Kim Y . (1999). J. Biol. Chem., 274, 33194–33197.
D'Orazi G, Cecchinelli B, Bruno T, Manni I, Higashimoto Y, Saito S, Gostissa M, Coen S, Marchetti A, Del Sal G, Piaggio G, Fanciulli M, Appella E and Soddu S . (2002). Nat. Cell Biol., 4, 11–19.
Ecsedy JA, Michaelson JS and Leder P . (2003). Mol. Cell. Biol., 23, 950–960.
el Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW and Vogelstein B . (1993). Cell, 75, 817–825.
Engelhardt OG, Boutell C, Orr A, Ullrich E, Haller O and Everett RD . (2003). Exp. Cell Res., 283, 36–50.
Eskiw CH and Bazett-Jones DP . (2002). Biochem. Cell Biol., 80, 301–310.
Greenwood E . (2002). Nat. Rev. Cancer, 2, 5.
Guldner HH, Szostecki C, Grotzinger T and Will H . (1992). J. Immunol., 149, 4067–4073.
Hofmann TG, Moller A, Sirma H, Zentgraf H, Taya Y, Droge W, Will H and Schmitz ML . (2002). Nat. Cell Biol., 4, 1–10.
Kern SE, Pietenpol JA, Thiagalingam S, Seymour A, Kinzler KW and Vogelstein B . (1992). Science, 256, 827–830.
Kim EJ, Park JS and Um SJ . (2002). J. Biol. Chem., 277, 32020–32028.
Kondo S, Lu Y, Debbas M, Lin AW, Sarosi I, Itie A, Wakeham A, Tuan J, Saris C, Elliott G, Ma W, Benchimol S, Lowe SW, Mak TW and Thukral SK . (2003). Proc. Natl. Acad. Sci. USA, 100, 5431–5436.
Lehming N, Le Saux A, Schuller J and Ptashne M . (1998). Proc. Natl. Acad. Sci. USA, 95, 7322–7326.
Möller A, Sirma H, Hofmann TG, Rueffer S, Klimczak E, Dröge W, Will H and Schmitz ML . (2003). Cancer Res., 63, 4310–4314.
Negorev D, Ishov AM and Maul GG . (2001). J. Cell Sci., 114, 59–68.
Negorev D and Maul GG . (2001). Oncogene, 20, 7234–7242.
Rogalla P, Kazmierczak B, Flohr AM, Hauke S and Bullerdiek J . (2000). Genomics, 63, 117–122.
Salomoni P and Pandolfi PP . (2002). Cell, 108, 165–170.
Seeler JS and Dejean A . (2001). Oncogene, 20, 7243–7249.
Seeler JS, Marchio A, Sitterlin D, Transy C and Dejean A . (1998). Proc. Natl. Acad. Sci. USA, 95, 7316–7321.
Sourvinos G and Everett RD . (2002). EMBO J., 21, 4989–4997.
Sternsdorf T, Jensen K, Reich B and Will H . (1999). J. Biol. Chem., 274, 12555–12566.
Szostecki C, Guldner HH, Netter HJ and Will H . (1990). J. Immunol., 145, 4338–4347.
Vogel A, Strassburg CP, Obermayer-Straub P, Brabant G and Manns MP . (2002). J. Mol. Med., 80, 201–211.
Wasylyk C, Schlumberger SE, Criqui-Filipe P and Wasylyk B . (2002). Mol. Cell. Biol., 22, 2687–2702.
Weis K, Rambaud S, Lavau C, Jansen J, Carvalho T, Carmo-Fonseca M, Lamond A and Dejean A . (1994). Cell, 76, 345–356.
Zong RT, Das C and Tucker PW . (2000). EMBO J., 19, 4123–4133.
Acknowledgements
Our work was supported by grants from the Deutsche Forschungsgemeinschaft (Schm 1417/3-1), Fonds der chemischen Industrie, EU project (QLK3-CT-2000-00463) sponsored by the Schweizerisches Bundesamt für Bildung und Wissenschaft, Oncosuisse, Schweizerischer Nationalfonds, Association for International Cancer Research, ‘Stiftung zur Förderung der wissenschaftlichen Forschung an der Universität Bern' and a grant from the Roche Research Foundation awarded to AM and MLS. The work of HS, TGH, HS and HW was supported by grants from the ‘Stiftung zur Bekämpfung Neuroviraler Erkrankungen', the Deutsche Krebshilfe (Wi2-10-1624) and the Deutsche Forschungsgemeinschaft (HO 2438/2-1; WI664/6-2). The Heinrich-Pette-Institut is supported by the Freie und Hansestadt Hamburg and the Bundesministerium für Gesundheit.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Möller, A., Sirma, H., Hofmann, T. et al. Sp100 is important for the stimulatory effect of homeodomain-interacting protein kinase-2 on p53-dependent gene expression. Oncogene 22, 8731–8737 (2003). https://doi.org/10.1038/sj.onc.1207079
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207079
Keywords
This article is cited by
-
Proteome profiling of phosphatidylinositol-5-phosphate 4-kinase type 2A and 2B knockdown cells identify modifications in key regulators involved in cell homeostasis and genome integrity
Genome Instability & Disease (2022)
-
Functional domains of SP110 that modulate its transcriptional regulatory function and cellular translocation
Journal of Biomedical Science (2018)
-
The role of cullin 5-containing ubiquitin ligases
Cell Division (2016)
-
Sp100A is a tumor suppressor that activates p53-dependent transcription and counteracts E1A/E1B-55K-mediated transformation
Oncogene (2016)
-
The DNA damage-induced cell death response: a roadmap to kill cancer cells
Cellular and Molecular Life Sciences (2016)