Abstract
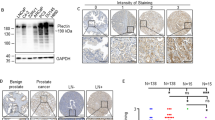
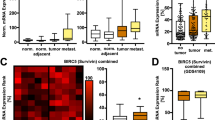
We have identified a novel gene, six transmembrane protein of prostate 2 (STAMP2), named for its high sequence similarity to the recently identified STAMP1 gene. STAMP2 displays a tissue-restricted expression with highest expression levels in placenta, lung, heart, and prostate and is predicted to code for a 459-amino acid six transmembrane protein. Using a form of STAMP2 labeled with green flourescent protein (GFP) in quantitative time-lapse and immunofluorescence confocal microscopy, we show that STAMP2 is primarily localized to the Golgi complex, trans-Golgi network, and the plasma membrane. STAMP2 also localizes to vesicular-tubular structures in the cytosol and colocalizes with the Early Endosome Antigen1 (EEA1) suggesting that it may be involved in the secretory/endocytic pathways. STAMP2 expression is exquisitely androgen regulated in the androgen-sensitive, androgen receptor-positive prostate cancer cell line LNCaP, but not in androgen receptor-negative prostate cancer cell lines PC-3 and DU145. Analysis of STAMP2 expression in matched normal and tumor samples microdissected from prostate cancer specimens indicates that STAMP2 is overexpressed in prostate cancer cells compared with normal prostate epithelial cells. Furthermore, ectopic expression of STAMP2 in prostate cancer cells significantly increases cell growth and colony formation suggesting that STAMP2 may have a role in cell proliferation. Taken together, these data suggest that STAMP2 may contribute to the normal biology of the prostate cell, as well as prostate cancer progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Accession codes
References
Ausebel FM, Brent R, Kingston R, Moore DD, Seidman JG, Smith JA and Struhl K . (1997). Curr Protocols in Molecular Biology. John Wiley and Sons: New York.
Bieberich CJ, Fujita K, He WW and Jay G . (1996). J. Biol. Chem., 271, 31779–31782.
Bonner RF, Emmert-Buck M, Cole K, Pohida T, Chuaqui R, Goldstein S and Liotta LA . (1997). Science, 278, 1481–1483.
Coffey DS . (1993). Cancer, 71, 880–886.
Crawford ED, Rosenblum M, Ziada AM and Lange PH . (1999). Urology, 54, 1–7.
Cunha GR, Donjacour AA, Cooke PS, Mee S, Bigsby RM, Higgins SJ and Sugimura Y . (1987). Endocr. Rev., 8, 338–362.
Deppenmeier U . (2002). Cell Mol. Life Sci., 59, 1513–1533.
Donaldson JG and Lippincott-Schwartz J . (2000). Cell, 101, 693–696.
Haass C . (1996). Curr. Opin. Neurol., 9, 254–259.
Haigh NG and Johnson AE . (2002). Protein Targeting, Transport and Translocation. In: Dalbey RE and von Heijne G (eds). Academic Press: London, UK, pp. 74–106.
He WW, Sciavolino PJ, Wing J, Augustus M, Hudson P, Meissner PS, Curtis RT, Shell BK, Bostwick DG, Tindall DJ, Gelmann EP, Abate-Shen C and Carter KC . (1997). Genomics., 43, 69–77.
Horoszewicz JS, Leong SS, Kawinski E, Karr JP, Rosenthal H, Chu TM, Mirand EA and Murphy GP . (1983). Cancer Res., 43, 1809–1818.
Hubert RS, Vivanco I, Chen E, Rastegar S, Leong K, Mitchell SC, Madraswala R, Zhou Y, Kuo J, Raitano AB, Jakobovits A, Saffran DC and Afar DE . (1999). Proc. Natl. Acad. Sci. USA, 96, 14523–14528.
Huggins C, Steven RE and Hodges CV . (1941). Arch. Surg., 43, 209–223.
Jenster G . (1999). Semin. Oncol., 26, 407–421.
Kleene R and Berger EG . (1993). Biochim. Biophys. Acta, 1154, 283–325.
Kokontis JM and Liao S . (1999). Vitam. Horm., 55, 219–307.
Korkmaz KS, Elbi C, Korkmaz CG, Loda M, Hager GL and Saatcioglu F . (2002). J. Biol. Chem., 277, 36689–36696.
Korkmaz KS, Korkmaz CG, Pretlow TG and Saatcioglu F . (2001). DNA Cell Biol., 20, 435–445.
Korkmaz KS, Korkmaz CG, Ragnhildstveit E, Pretlow TG and Saatcioglu F . (2000). DNA Cell Biol., 19, 499–506.
Lemmon SK and Traub LM . (2000). Curr. Opin. Cell Biol., 12, 457–466.
Linstedt AD and Hauri HP . (1993). Mol. Biol. Cell, 4, 679–693.
Lippincott-Schwartz J, Cole N and Presley J . (1998). Trends Cell Biol., 8, 16–20.
Mellman I . (2000). J. Cell Biol., 149, 529–530.
Misteli T and Spector DL . (1999). Mol. Cell., 3, 697–705.
Moldes M, Lasnier F, Gauthereau X, Klein C, Pairault J, Feve B and Chambaut-Guerin AM . (2001). J. Biol. Chem., 276, 33938–33946.
Natsuka S, Gersten KM, Zenita K, Kannagi R and Lowe JB . (1994). J. Biol. Chem., 269, 16789–16794.
Nelson PS, Clegg N, Arnold H, Ferguson C, Bonham M, White J, Hood L and Lin B . (2002). Proc. Natl. Acad. Sci. USA, 99, 11890–11895.
Nelson PS, Gan L, Ferguson C, Moss P, Gelinas R, Hood L and Wang K . (1999). Proc. Natl. Acad. Sci. USA, 96, 3114–3119.
Ordovas JM . (2000). Nutr. Rev., 58, 76–79.
Passer BJ, Nancy-Portebois V, Amzallag N, Prieur S, Cans C, Roborel de Climens A, Fiucci G, Bouvard V, Tuynder M, Susini L, Morchoisne S, Crible V, Lespagnol A, Dausset J, Oren M, Amson R and Telerman A . (2003). Proc. Natl. Acad. Sci. USA, 100, 2284–2289.
Pepperkok R, Scheel J, Horstmann H, Hauri HP, Griffiths G and Kreis TE . (1993). Cell, 74, 71–82.
Phang JM . (1985). Curr. Top. Cell. Regul., 25, 91–132.
Ponnambalam S and Banting G . (1996). Curr. Biol., 6, 1076–1078.
Rittenhouse HG, Finlay JA, Mikolajczyk SD and Partin AW . (1998). Crit. Rev. Clin. Lab. Sci., 35, 275–368.
Roy AK, Lavrovsky Y, Song CS, Chen S, Jung MH, Velu NK, Bi BY and Chatterjee B . (1999). Vitam. Horm., 55, 309–352.
Srikantan V, Zou Z, Petrovics G, Xu L, Augustus M, Davis L, Livezey JR, Connell T, Sesterhenn IA, Yoshino K, Buzard GS, Mostofi FK, McLeod DG, Moul JW and Srivastava S . (2000). Proc. Natl. Acad. Sci. USA, 97, 12216–12221.
Steiner MS, Zhang X, Wang Y and Lu Y . (2000). Cancer Res., 60, 4419–4425.
Stenmark H, Aasland R, Toh BH and D'Arrigo A . (1996). J. Biol. Chem., 271, 24048–24054.
Stephan C, Jung K, Diamandis EP, Rittenhouse HG, Lein M and Loening SA . (2002). Urology, 59, 2–8.
Stephenson SA, Verity K, Ashworth LK and Clements JA . (1999). J. Biol. Chem., 274, 23210–23214.
Tsien RY . (1998). Annu. Rev. Biochem., 67, 509–544.
Warkentin E, Mamat B, Sordel-Klippert M, Wicke M, Thauer RK, Iwata M, Iwata S, Ermler U and Shima S . (2001). EMBO J., 20, 6561–6569.
Xu LL, Su YP, Labiche R, Segawa T, Shanmugam N, McLeod DG, Moul JW and Srivastava S . (2001). Int. J. Cancer, 92, 322–328.
Yousef GM, Obiezu CV, Luo LY, Black MH and Diamandis EP . (1999). Cancer Res., 59, 4252–4256.
Acknowledgements
This work was supported by grants from the Norwegian Research Council, Norwegian Cancer Society, and University of Oslo to FS. We thank Dr Erik Snapp and members of the FS lab for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Korkmaz, C., Korkmaz, K., Kurys, P. et al. Molecular cloning and characterization of STAMP2, an androgen-regulated six transmembrane protein that is overexpressed in prostate cancer. Oncogene 24, 4934–4945 (2005). https://doi.org/10.1038/sj.onc.1208677
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208677
Keywords
This article is cited by
-
A novel prognostic signature and therapy guidance for hepatocellular carcinoma based on STEAP family
BMC Medical Genomics (2024)
-
STEAP4 inhibits cisplatin-induced chemotherapy resistance through suppressing PI3K/AKT in hepatocellular carcinoma
Cancer & Metabolism (2023)
-
MicroRNA-301a-3p promotes diabetic retinopathy via regulation of six-transmembrane epithelial antigen of prostate 4
Inflammation Research (2021)
-
Near-infrared fluorescent probe for fast track of cyclooxygenase-2 in Golgi apparatus in cancer cells
Frontiers of Chemical Science and Engineering (2020)
-
Cryo-EM structures of human STEAP4 reveal mechanism of iron(III) reduction
Nature Communications (2018)