Abstract
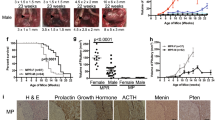
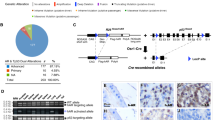
To identify possible genetic interactions between the mechanisms of tumor suppression of menin and pRb, we intercrossed mice with targeted deletions of Men1 and Rb1, and compared tumor development in cohorts of animals carrying single or dual mutations of these tumor-suppressor genes. In mice lacking one copy of Men1, pancreatic islet and anterior pituitary adenomas are common. In animals lacking one copy of Rb1, intermediate pituitary and thyroid tumors occur at high frequency, with less frequent development of pancreatic islet hyperplasia and parathyroid lesions. In mice heterozygous for both Men1 and Rb1, pancreatic hyperplasia and tumors of the intermediate pituitary and thyroid occurred at high frequency. Serum measurements of calcium and glucose did not vary significantly between genotypic groups. Loss of heterozygosity at the Rb1 locus was common in pituitary and thyroid tumors, whereas loss of menin was observed in pancreatic and parathyroid lesions. The tumor spectrum in the double heterozygotes was a combination of pathologies seen in each of the individual heterozygotes, without decrease in age of onset, indicating independent, non-additive effects of the two mutations. Together with the lack of increased tumor spectrum, this suggests that menin and pRb function in a common pathway of tumor suppression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Agarwal SK, Novotny EA, Crabtree JS, Weitzman JB, Yaniv M, Burns AL et al. (2003). Transcription factor JunD, deprived of menin, switches from growth suppressor to growth promoter. Proc Natl Acad Sci USA 100: 10770–10775.
Bertolino P, Tong WM, Galendo D, Wang ZQ, Zhang CX . (2003). Heterozygous Men1 mutant mice develop a range of endocrine tumors minicking multiple endocrine neoplasia type 1. Mol Endocrinol 17: 1880–1892.
Biondi C, Gartside MG, Tonks I, Paterson C, Hayward N, Kay GF . (2002). Targeting and conditional inactivation of the murine Men1 locus using the Cre recombinase:loxP system. Genesis 32: 150–151.
Biondi CA, Gartside MG, Waring P, Loffler KA, Stark MS, Magnuson M et al. (2004). Conditional inactivation of the Men1 gene leads to pancreatic and pituitary tumorigenesis but does not affect normal development of these tissues. Mol Cell Biol 24: 3125–3131.
Busygina V, Suphapeetiporn K, Marek LR, Stowers RS, Xu T, Bale AE . (2004). Hypermutability in a Drosophila model for multiple endocrine neoplasia type 1. Hum Mol Genet 13: 2399–2408.
Casanovas O, Hager JH, Gee Hong Chun M, Hanahan D . (2005). Incomplete inhibition of the Rb tumor suppressor pathway in the context of inactivated p53 is sufficient for pancreatic islet tumorigenesis. Oncogene 24: 6597–6604.
Chandrasekharappa S, Teh BT . (2003). Functional studies of the MEN1 gene. J Intern Med 253: 599–605.
Chandrasekharappa SC, Guru SC, Manickam P, Olufemi SE, Collins FS, Emmert-Buck MR et al. (1997). Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 276: 404–407.
Crabtree JS, Scacheri PC, Ward JM, Garrett-Beal L, Emmert-Buck MR, Edgemon KA et al. (2001). A mouse model of multiple endocrine neoplasia, type 1, develops multiple endocrine tumors. Proc Natl Acad Sci USA 98: 1118–1123.
Cryns VL, Thor A, Xu HJ, Hu SX, Wierman ME, Vickery Jr AL et al. (1994). Loss of the retinoblastoma tumor-suppressor gene in parathyroid carcinoma. N Engl J Med 330: 757–761.
Dunn JM, Phillips RA, Becker AJ, Gallie BL . (1988). Identification of germline and somatic mutations affecting the retinoblastoma gene. Science 241: 1797–1800.
Gobl AE, Berg M, Lopez-Egido JR, Oberg K, Skogseid B, Westin G . (1999). Menin represses JunD-activated transcription by a histone deacetylase-dependent mechanism. Bioch Biophys Acta 1447: 51–56.
Goodrich DW, Wang NP, Qian YW, Lee EY, Lee WH . (1991). The retinoblastoma gene product regulates progression through the G1 phase of the cell cycle. Cell 67: 293–302.
Heppner C, Bilimoria KY, Agarwal SK, Kester M, Whitty LJ, Guru SC et al. (2001). The tumor suppressor protein menin interacts with NF-kappaB proteins and inhibits NF-kappaB-mediated transactivation. Oncogene 20: 4917–4925.
Hernando E, Nahle Z, Juan G, Diaz-Rodriguez E, Alaminos M, Hemann M et al. (2004). Rb inactivation promotes genomic instability by uncoupling cell cycle progression from mitotic control. Nature 430: 797–802.
Herrera RE, Sah VP, Williams BO, Makela TP, Weinberg RA, Jacks T . (1996). Altered cell cycle kinetics, gene expression, and G1 restriction point regulation in Rb-deficient fibroblasts. Mol Cell Biol 16: 2402–2407.
Jacks T, Fazeli A, Schmitt EM, Bronson RT, Goodell MA, Weinberg RA . (1992). Effects of an Rb mutation in the mouse. Nature 359: 295–300.
Jin S, Mao H, Schnepp RW, Sykes SM, Silva AC, D'Andrea AD et al. (2003). Menin associates with FANCD2, a protein involved in repair of DNA damage. Cancer Res 63: 4204–4210.
Kaji H, Canaff L, Lebrun JJ, Goltzman D, Hendy GN . (2001). Inactivation of menin, a Smad3-interacting protein, blocks transforming growth factor type beta signaling. Proc Natl Acad Sci USA 98: 3837–3842.
Karnik SK, Hughes CM, Gu X, Rozenblatt-Rosen O, McLean GW, Xiong Y et al. (2005). Menin regulates pancreatic islet growth by promoting histone methylation and expression of genes encoding p27Kip1 and pl8INK4c. Proc Natl Acad Sci USA 102: 14659–14664.
Kim H, Lee JE, Cho EJ, Liu JO, Youn HD . (2003). Menin, a tumor suppressor, represses JunD-mediated transcriptional activity by association with an mSin3A-histone deacetylase complex. Cancer Res 63: 6135–6139.
Lemmens I, Van de Ven WJ, Kas K, Zhang CX, Giraud S, Wautot V et al. (1997). Identification of the multiple endocrine neoplasia type 1 (MEN1) gene. The European Consortium on MEN1. Hum Mol Genet 6: 1177–1183.
Loffler KA, Biondi C, Gartside MG, Waring P, Stark MS, Serewko-Auret MM et al. (2006). Broad tumour spectrum in a mouse model of multiple endocrine neoplasia type 1. Int J Cancer Oct 16; [Epub ahead of print].
Park MS, Rosai J, Nguyen HT, Capodieci P, Cordon-Cardo C, Koff A . (1999). p27 and Rb are on overlapping pathways suppressing tumorigenesis in mice. Proc Natl Acad Sci USA 96: 6382–6387.
Poisson A, Zablewska B, Gaudray P . (2003). Menin interacting proteins as clues towards the understanding of multiple endocrine neoplasia type 1. Cancer Lett 189: 1–10.
Ratineau C, Bernard C, Poncet G, Blanc M, Josso C, Fontaniere S et al. (2004). Reduction of menin expression enhances cell proliferation and is tumorigenic in intestinal epithelial cells. J Biol Chem 279: 24477–24484.
Schnepp RW, Mao H, Sykes SM, Zong WX, Silva AC, La P et al. (2004). Menin induces apoptosis in murine embryonic fibroblasts. J Biol Chem 279: 10685–10691.
Sowa H, Kaji H, Canaff L, Hendy GN, Tsukamoto T, Yamaguchi T et al. (2003). Inactivation of menin, the product of the multiple endocrine neoplasia type 1 gene, inhibits the commitment of multipotential mesenchymal stem cells into the osteoblast lineage. J Biol Chem 278: 21058–21069.
Sowa H, Kaji H, Hendy GN, Canaff L, Komori T, Sugimoto T et al. (2004). Menin is required for bone morphogenetic protein 2-and transforming growth factor beta- regulated osteoblastic differentiation through interaction with Smads and Runx2. J Biol Chem 279: 40267–40275.
Tonks ID, Hacker E, Irwin N, Muller HK, Keith P, Mould A et al. (2005). Melanocytes in conditional Rb−/− mice are normal in vivo but exhibit proliferation and pigmentation defects in vitro. Pigment Cell Res 18: 252–264.
Williams BO, Remington L, Albert DM, Mukai S, Bronson RT, Jacks T . (1994). Cooperative tumorigenic effects of germline mutations in Rb and p53. Nat Genet 7: 480–484.
Acknowledgements
We acknowledge the advice and assistance of N Pandeya, N Irwin, E Hacker, G Walker, S Cassidy, M Trivett, F Feleppa, the QIMR/University of Queensland Histotechnology Facility, and the Chemical Pathology and Anatomical Pathology sections of Queensland Health Pathology and Scientific Services at the Royal Brisbane and Women's Hospital. The work described here has been supported by grants from the Queensland Cancer Fund and the National Health and Medical Research Council of Australia. GFK is a Queensland Cancer Fund Fellow and NKH is a Principal Research Fellow of the National Health and Medical Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Loffler, K., Biondi, C., Gartside, M. et al. Lack of augmentation of tumor spectrum or severity in dual heterozygous Men1 and Rb1 knockout mice. Oncogene 26, 4009–4017 (2007). https://doi.org/10.1038/sj.onc.1210163
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210163
Keywords
This article is cited by
-
Recent progress of experimental model in pancreatic neuroendocrine tumors: drawbacks and challenges
Endocrine (2023)
-
Mice deleted for cell division cycle 73 gene develop parathyroid and uterine tumours: model for the hyperparathyroidism-jaw tumour syndrome
Oncogene (2017)
-
Menin and p53 have non-synergistic effects on tumorigenesis in mice
BMC Cancer (2012)
-
Menin and bone metabolism
Journal of Bone and Mineral Metabolism (2012)
-
Clinical and molecular genetics of acromegaly: MEN1, Carney complex, McCune–Albright syndrome, familial acromegaly and genetic defects in sporadic tumors
Reviews in Endocrine and Metabolic Disorders (2008)