Summary:
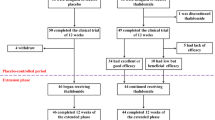
In total, 94 patients with homozygous beta thalassemia were randomized to two different conditioning regimens: busulfan 600 mg/m2+cyclophosphamide 200 mg/kg or busulfan 16 mg/kg+cyclophosphamide 200 mg/kg and antilymphocyte globulin (47 in each group), for bone marrow transplantation, to see whether increased myeloablation or increased immunosuppression would reduce rejection. Busulfan pharmacokinetics in determining outcome was evaluated. There was no significant difference in engraftment, graft-versus-host disease, rejection, and overall and disease-free survival in the two groups. Systemic exposure to busulfan was significantly higher in the 600 mg/m2 group, but in both groups there was a wide interindividual variation in the busulfan kinetics. Six patients rejected the graft, two in the busulfan 600 mg group and four in busulfan 16 mg group (P=0.677 CI −0.17, 0.07), but in five patients (pharmacokinetic data not available in one patient) who rejected the graft busulfan first dose trough level (Cmin-1) was below 150 ng/ml while it was above this level in the 66 of 68 patients with successful engraftment (P⩽0.001). This randomized trial shows that rejection is influenced by busulfan levels and suggests that monitoring of busulfan levels and dose adjustment based on first-dose kinetics may reduce the risk of rejection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Galimberti M, Angelucci E, Baroncianni D et al. Bone marrow transplantation in thalasemia. The experience of Pesaro. Bone Marrow Transplant 1997; 19 (Suppl. 2): 45–47.
Schrier SL, Angelucci E . New strategies in the treatment of the thalassemias. Ann Rev Med 2005; 56: 157–171.
Ball LM, Lankester AC, Giordano PC et al. Paediatric allogeneic bone marrow transplantation for homozygous beta-thalassaemia, the Dutch experience. Bone Marrow Transplant 2003; 31: 1081–1087.
Shing MM, Yuen PM, Li CK et al. A more immunosuppressive pre-transplant conditioning may be required for Chinese patients with thalassaemia. Bone Marrow Transplant 1996; 17: 907–910.
Poonkuzhali B, Srivastava A, Quernin MH et al. Pharmacokinetics of oral busulfan in children with beta thalassemia major undergoing allogeneic bone marrow transplantation. Bone marrow Transplant 1999; 24: 5–11.
Hassan M, Fasth A, Gerritsen B et al. Busulfan kinetics and limited sampling model in children with leukemia and inherited disorders. Bone Marrow Transplant 1996; 18: 843–850.
Vassal G, Deroussent A, Challine D et al. Is 600 mg/m2 the appropriate dosage of busulfan in children undergoing bone marrow transplantation? Blood 1992; 79: 2475–2479.
Storb R, Blume KG, O’Donnell MR et al. Cyclophosphamide and antithymocyte globulin to condition patients with aplastic anemia for allogeneic marrow transplantations: the experience in four centers. Blood Marrow Transplant 2001; 7: 39–44.
Chandy M, Srivastava A, Dennison D et al. Allogeneic bone marrow transplantation in the developing world: experience from a center in India. Bone Marrow Transplant 2001; 27: 785–790.
Glucksberg H, Storb R, Fefer A et al. Clinical manifestations of graft versus host disease in human recipients of marrow from HLA matched sibling donors. Transplantation 1974; 18: 295–304.
Bearman SI, Appelbaum FR, Buckner CD et al. Regimen-related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol 1988; 6: 1562–1568.
Jones RJ, Lee KSK, Beschorner WE et al. Venocclusive disease of the liver following bone marrow transplantation. Transplantation 1987; 44: 778–783.
Quernin MH, Poonkuzhali B, Medard Y et al. High-performance liquid chromatographic method for quantification of busulfan in plasma after derivatization by tetrafluorothiophenol. J Chromatogr 1999; 721: 147–152.
Quernin MH, Poonkuzhali B, Montes C et al. Quantification of busulfan in plasma by Gas chromatography-mass spectrometry. J Chromatogr 1998; 709: 47–56.
Heinzel G, Woloszezak R, Thoman P . Pharmacokinetic Pharmacodynamic Data Analysis System for the PC. Gustav Fischer: Sturtgart, Germany, 1993.
Thomas ED, Buckner CD, Sanders JE et al. Marrow transplantation for thalassaemia. Lancet 1982; 2: 227.
Lucarelli G, Andreani M, Angelucci E . The cure of thalassemia by bone marrow transplantation. Blood Rev 2002; 16: 81–85.
Chen YC, Lin KH, Huang WS, Tang JL . Bone marrow transplantation in Taiwan: an overview. Bone Marrow Transplant 1994; 13: 907–910.
Lawson SE, Roberts IA, Amrolia P et al. Bone marrow transplantation for beta-thalassaemia major: the UK experience in two paediatric centres. Br J Haematol 2003; 120: 289–295.
Iannone R, Casella JF, Fuchs EJ et al. Results of minimally toxic nonmyeloablative transplantation in patients with sickle cell anemia and beta-thalassemia. Biol Blood Marrow Transplant 2003; 9: 519–528.
Sodani P, Gaziev D, Polchi P et al. A new approach for bone marrow transplantation in class 3 thalassemic patients aged less than 17 years. Blood 2004; 104: 1201–1203.
Brachet C, Azzi N, Demulder A et al. Hydroxyurea treatment for sickle cell disease: impact on hematopoietic stem cell transplantation outcome. Bone Marrow Transplant 2004; 33: 799–803.
Storb R, Etzioni R, Anasetti C et al. Cyclophosphamide combined with antithymocyte globulin in preparation for allogeneic marrow transplants in patients with aplastic anemia. Blood 1994; 84: 941–949.
Vassal G, Fischer G, Challine D et al. Busulfan disposition below the age of three: alteration in children with lysosomal storage disease. Blood 1993; 82: 1030–1034.
Hassan M, Ljungman P, Bolme P et al. Busulfan bioavailability. Blood 1994; 84: 2144–2150.
Shaw PJ, Scharping CE, Brian RJ, Earl JW . Busulfan pharmacokinetics using a single daily high-dose regimen in children with acute leukemia. Blood 1994; 84: 2357–2362.
Yeager AM, Wagner JE, Graham ML et al. Optimization of busulfan dosage in children undergoing bone marrow transplantation: a pharmacokinetic study of dose escalation. Blood 1992; 80: 2425–2428.
Slattery JT, Sanders JE, Buckner CD et al. Graft rejection and toxicity following bone marrow transplantation in relation to busulfan pharmacokinetics. Bone Marrow Transplant 1995; 16: 31–42.
Slattery JT, Clift RA, Buckner CD et al. Marrow transplantation for chronic myeloid leukemia: the influence of plasma busulfan levels on the outcome of transplantation. Blood 1997; 89: 3055–3060.
McCune JS, Gooley T, Gibbs JP et al. Busulfan concentration and graft rejection in pediatric patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 2002; 30: 167–173.
Pawlowska AB, Blazar BR, Angelucci E et al. Relationship of plasma pharmacokinetics of high-dose oral busulfan to the outcome of allogeneic bone marrow transplantation in children with thalassemia. Bone Marrow Transplant 1997; 20: 915–920.
Grochow LB, Jones RJ, Brundrett RB et al. Pharmacokinetics of busulfan: correlation with veno-occlusive disease in patients undergoing bone marrow transplantation. Cancer Chemother Pharmacol 1989; 25: 55–61.
Dix SP, Wingard JR, Mullins RE et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant 1996; 17: 225–230.
Anderson BS, Thall PF, Madden T et al. Busulfan systemic exposure relative to regimen related toxicity and acute graft versus host disease: defining a therapeutic window for IV BuCy2 in chronic myelogenous leukemia. Biol Blood Marrow Transplant 2002; 8: 477–485.
Ljungman P, Hassan M, Bekassy AN et al. Busulfan concentration in relation to permanent alopecia in recipients of bone marrow transplants. Bone Marrow Transplantation 1995; 15: 869–871.
Srivastava A, Poonkuzhali B, Shaji RV et al. Glutathione S-transferase M1 polymorphism – a risk factor for hepatic venoocclusive disease in bone marrow transplantation. Blood 2004; 104: 1574–1577.
Bleyazac N, Souillet G, Magron P et al. Reduced transplant related mortality by Bayesian individualization of busulfan dosage in children. Bone Marrow Transplant 2001; 28: 743–751.
Demirer T, Buckner CD, Appelbaum FR et al. Busulfan, cyclophosphamide and fractionated total body irradiation for allogeneic marrow transplantation in advanced acute and chronic myelogenous leukemia: phase I dose escalation of busulfan based on targeted plasma levels. Bone Marrow Transplant 1996; 17: 341–346.
Dupuis LL, Najdova M, Saunders EF . Retrospective appraisal of busulfan dose adjustment in children. Bone Marrow Transplant 2000; 26: 1143–1147.
McCune JS, Gibbs JP, Slattery JT . Plasma concentration monitoring of busulfan: does it improve clinical outcome? Clin Pharmacokinet 2000; 39: 155–165.
Acknowledgements
This study was supported in part by the Indian Council of Medical Research grant towards the project ‘Advanced Center for Bone Marrow Transplantation for Thalassaemia in India’ (56/2/93-BMS II).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chandy, M., Balasubramanian, P., Ramachandran, S. et al. Randomized trial of two different conditioning regimens for bone marrow transplantation in thalassemia – the role of busulfan pharmacokinetics in determining outcome. Bone Marrow Transplant 36, 839–845 (2005). https://doi.org/10.1038/sj.bmt.1705151
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705151
Keywords
This article is cited by
-
Is Old (Fludrabine/Busulfan/Cyclophosphamide/rAntiThymocyteGlobulin) Conditioning Still Gold for Allogeneic Transplants in Transfusion Dependent Beta-Thalassemia of All Risk Categories in 21st Century?
Indian Journal of Hematology and Blood Transfusion (2023)
-
UPLC-Tandem Mass Spectrometry for Quantification of Busulfan in Human Plasma: Application to Therapeutic Drug Monitoring
Scientific Reports (2020)
-
Long-term outcome of mixed chimerism after stem cell transplantation for thalassemia major conditioned with busulfan and cyclophosphamide
Bone Marrow Transplantation (2018)
-
Hematopoietic stem cell transplantation for homozygous β-thalassemia and β-thalassemia/hemoglobin E patients from haploidentical donors
Bone Marrow Transplantation (2016)
-
Second BMT for thalassemia major using CY post transplant
Bone Marrow Transplantation (2014)