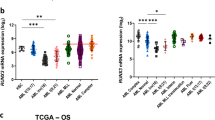
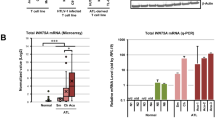
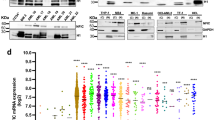
Abstract
The LYL1 gene encodes a basic helix–loop–helix transcription factor involved in T-cell acute lymphoblastic leukemia. Using real-time quantitative RT-PCR assay, we found that the expression of LYL1 was at higher levels in the majority cases of acute myeloblastic leukemia (AML) or myelodysplastic syndrome when compared to normal bone marrow. Our study also showed that LYL1 was highly expressed in most AML cell lines and in CD34+ AML cells. To determine whether LYL1 had an affect on the phenotype and behavior of myeloid cells, we introduced full-length LYL1 cDNA into K562 cells using electroporation and U937 cells with retroviral infection. Both of the derivative cell lines with overexpression of LYL1 had an increased growth rate and clonogenecity. Forced expression of LYL1 in K562 cells enhanced spontaneous and hemin-induced erythroid differentiation but blocked spontaneous as well as PMA-induced megakaryocytic differentiation. Overexpression of LYL1 in U937 cells blocked all-trans retinoic acid-induced monocytic differentiation. The LYL1-transfected U937 cells were also more resistant to the cytotoxic drug cytarabine. These results demonstrate that LYL1 may play a role in early hematopoiesis and may be a potential oncogenic factor in AML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cleary ML, Mellentin JD, Spies J, Smith SD . Chromosomal translocation involving the beta T cell receptor gene in acute leukemia. J Exp Med 1988; 167: 682–687.
Mellentin JD, Smith SD, Cleary ML . lyl-1, a novel gene altered by chromosomal translocation in T cell leukemia, codes for a protein with a helix–loop–helix DNA binding motif. Cell 1989; 58: 77–83.
Ferrando AA, Neuberg DS, Staunton J, Loh ML, Huard C, Raimondi SC et al. Gene expression signatures define novel oncogenic pathways in T cell acute lymphoblastic leukemia. Cancer Cell 2002; 1: 75–87.
Ferrier R, Nougarede R, Doucet S, Kahn-Perles B, Imbert J, Mathieu-Mahul D . Physical interaction of the bHLH LYL1 protein and NF-kappaB1 p105. Oncogene 1999; 18: 995–1005.
Bruckner R, Jentsch-Ullrich K, Franke A, Wieacker P, Stumm M . A novel translocation (17;19)(p13;p13) in a patient with acute myelomonocytic leukemia. Cancer Genet Cytogenet 2000; 119: 77–79.
Vardiman JW, Harris NL, Brunning RD . The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 2002; 100: 2292–2302.
McCulloch EA, Minden MD, Curtis JE . The influence of growth regulation on treatment strategy for acute myeloblastic leukaemia. Cancer Surv 1990; 9: 169–198.
Han Y, San Marina S, Liu J, Minden MD . Transcriptional activation of c-myc proto-oncogene by WT1 protein. Oncogene 2004; 23: 6933–6941.
Asnafi V, Beldjord K, Libura M, Villarese P, Millien C, Ballerini P et al. Age-related phenotypic and oncogenic differences in T-cell acute lymphoblastic leukemias may reflect thymic atrophy. Blood 2004; 104: 4173–4180.
Miyamoto A, Cui X, Naumovski L, Cleary ML . Helix–loop–helix proteins LYL1 and E2a form heterodimeric complexes with distinctive DNA-binding properties in hematolymphoid cells. Mol Cell Biol 1996; 16: 2394–2401.
Keating A, Horsfall W, Hawley RG, Toneguzzo F . Effect of different promoters on expression of genes introduced into hematopoietic and marrow stromal cells by electroporation. Exp Hematol 1990; 18: 99–102.
Hu ZB, Minden MD, McCulloch EA, Stahl J . Regulation of drug sensitivity by ribosomal protein S3a. Blood 2000; 95: 1047–1055.
Bonnet D, Dick JE . Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730–737.
Visvader J, Begley CG, Adams JM . Differential expression of the LYL, SCL and E2A helix–loop–helix genes within the hemopoietic system. Oncogene 1991; 6: 187–194.
Ballerini P, Blaise A, Busson-Le Coniat M, Su XY, Zucman-Rossi J, Adam M et al. HOX11L2 expression defines a clinical subtype of pediatric T-ALL associated with poor prognosis. Blood 2002; 100: 991–997.
Bernard M, Delabesse E, Novault S, Hermine O, Macintyre EA . Antiapoptotic effect of ectopic TAL1/SCL expression in a human leukemic T-cell line. Cancer Res 1998; 58: 2680–2687.
Acknowledgements
We thank Dr Claude Cantin and Rakesh Nayyar in the lab of flow cytometry for sorting the GFP-expressing cells. We thank all our colleagues in Dr MD Minden's lab for invaluable help and critical discussions. This study was supported by grants from the Leukemia Lymphoma Society and the National Cancer Institute of Canada.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meng, YS., Khoury, H., Dick, J. et al. Oncogenic potential of the transcription factor LYL1 in acute myeloblastic leukemia. Leukemia 19, 1941–1947 (2005). https://doi.org/10.1038/sj.leu.2403836
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403836
Keywords
This article is cited by
-
Super-enhancer profiling identifies novel critical and targetable cancer survival gene LYL1 in pediatric acute myeloid leukemia
Journal of Experimental & Clinical Cancer Research (2022)
-
LYL1 gene amplification predicts poor survival of patients with uterine corpus endometrial carcinoma: analysis of the Cancer genome atlas data
BMC Cancer (2018)
-
Annexin A2-S100A10 heterotetramer is upregulated by PML/RARα fusion protein and promotes plasminogen-dependent fibrinolysis and matrix invasion in acute promyelocytic leukemia
Frontiers of Medicine (2017)
-
Regulation of p21 by TWIST2 contributes to its tumor-suppressor function in human acute myeloid leukemia
Oncogene (2015)
-
Conformational flexibility of the oncogenic protein LMO2 primes the formation of the multi-protein transcription complex
Scientific Reports (2014)