Abstract
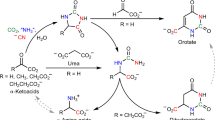
THE similarity of major biochemical pathways throughout the animal and plant kingdoms1 presumably reflects the ancient formation of the pathways in a common ancestral counterpart of modern cell types. The pathway previously known for L-tyrosine biosynthesis in both prokaryotie and eukaryotic microorganisms (Fig. 1a) involves a dehydrogenation-decarboxylation conversion of prephenate to 4-hydroxyphenylpyruvate followed by transamination in the presence of a suitable amino donor. Our data (Fig. 1b) demonstrate a reversed order of these two reactions in several species of blue–green algae (more appropriately designated bacteria (order Chrooeocales2)).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Florkin, M., in Unity and Diversity in Biochemistry (Pergamon, London and New York, 1960).
Stanier, R. Y., Kunisawa, R., Mandel, M., and Cohen-Bazire, G., Bact. Rev., 35, 171 (1971).
Van Baalen, C., Bot. Mar., 4, 129 (1962).
Gibson, F., Biochem. J., 190, 256 (1964).
Young, I. G., Gibson, F., and MacDonald, C. G., Biochim. biophys. Acta, 192, 62 (1969).
Walker, F. P., and Udenfriend, S., J. Lab. clin. Med., 50, 733 (1957).
Jensen, R. A., and Rebello, J. L., Devl. Indust. Microbiol., 11, 105 (1970).
Jensen, R. A., J. biol. Chem., 244, 2816 (1969).
Bower, J. W., and Skerman, V. B. D., J. gen. Microbiol., 54, 299 (1968).
Van Baalen, C., Hoare, D. S., and Brandt, E., J. Bact., 105, 685 (1971).
Barghoorn, E. S., Sci. Am., 244, 30 (1971).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
STENMARK, S., PIERSON, D., JENSEN, R. et al. Blue-Green Bacteria synthesise L-Tyrosine by the Pretyrosine Pathway. Nature 247, 290–292 (1974). https://doi.org/10.1038/247290a0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1038/247290a0
This article is cited by
-
Chimeric natural products derived from medermycin and the nature-inspired construction of their polycyclic skeletons
Nature Communications (2022)
-
De-regulated assimilation and over-production of amino acids in analogue-resistant mutants of a cyanobacterium, Phormidium uncinatum
World Journal of Microbiology & Biotechnology (1995)
-
Phylogenetic distribution of components of the overflow pathway tol-phenylalanine within the enteric lineage of bacteria
Current Microbiology (1988)
-
New prospects for deducing the evolutionary history of metabolic pathways in prokaryotes: Aromatic biosynthesis as a case-in-point
Origins of life and evolution of the biosphere (1988)
-
Production of L-phenylalanine from phenylpyruvate using resting cells of Escherichia coli
Biotechnology Letters (1987)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.