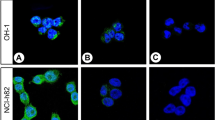
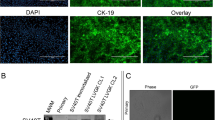
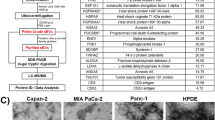
Abstract
A wide variety of virally and spontaneously transformed fibroblasts secrete a major transformation-related phosphoprotein with a molecular weight (MW), depending on the species of origin, of about 62,000 (62K)1–13. Markedly elevated extracellular levels of this major 32P-labelled protein are not simply linked to exponential growth1 but instead are associated directly with transformation. The phosphoprotein is not antigenically related to p60src, p60c-src or simian virus 40 (SV40) non-viral T antigen2, and it is further distinguishable from SV40 non-viral T antigen (pp 53) on the basis of its electrophoretic mobility. In this study we have compared a variety of normal and transformed epithelial cells for secretion of this 32P-labelled protein and have found that this marker distinguishes neoplastic from preneoplastic and normal mouse mammary epithelium and also identifies highly tumorigenic cells derived from guinea pig bile duct epithelium and rat liver epithelium. Because the classical phenotypic properties commonly associated with transformation of fibroblasts cannot be generally used to discriminate tumorigenic from non-tumorigenic epithelial cells4–9, this phosphoprotein, which identifies tumorigenic cells of both fibroblastic and epithelial origin, is likely to be of particular importance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Senger, D. R., Wirth, D. F. & Hynes, R. O. Cell 16, 885–893 (1979).
Senger, D. R., Wirth, D. F., Bryant, C. & Hynes, R. O. Cold Spring Harb. Symp. quant. Biol. 44, 651–657 (1980).
Senger, D. R., Wirth, D. F. & Hynes, R. O. Nature 286, 619–621 (1980).
Butel, J. S., Dudley, J. P. & Noonan, C. P. in Cell Biology of Breast Cancer (eds McGrath, C., Brennan, M. S. & Rich, M. A.) 317–345 (Academic, New York, 1980).
Hosick, H. L., Anderson, L. W., Angello, J. C. & Danielson, K. G. in Cell Biology of Breast Cancer (eds McGrath, C., Brennan, M. S. & Rich, M. A.) 393–412 (Academic, New York, 1980).
Miller, F. R., Dexter, D. L. & Heppner, G. H. in Cell Biology of Breast Cancer (eds McGrath, C., Brennan, M. S. & Rich, M. A.) 413–422 (Academic, New York, 1980).
Marshall, C. J., Franks, L. M. & Carbonell, A. W. J. natn. Cancer Inst. 58, 1743–1751 (1977).
San, R. H. C. et al. Cancer Res. 39, 1026–1034 (1979).
Yuspa, S. H., Hahley-Nelson, P., Koehler, B. & Stanley, J. R. Cancer Res. 40, 4694–4703 (1980).
Asch, B. B. & Medina, D. J. natn. Cancer Inst. 61, 1423–1430 (1978).
Medina, D. & DeOme, K. B. J. natn. Cancer Inst. 45, 353–363 (1970).
Asch, B. B., Medina, D. & Brinkley, B. R. Cancer Res. 39, 893–907 (1979).
Laemmli, U. K. Nature 227, 680–685 (1970).
Kessler, S. W. J. Immun. 115, 1617–1624 (1975).
Zbar, B., Wepsic, H. T., Rapp, H. J., Whang-Peng, J. & Borsos, T. J. natn. Cancer Inst. 43, 821–831 (1969).
Zbar, B., Bernstein, I., Tanaka, T. & Rapp, H. J. Science 170, 1217–1218 (1970).
Dvorak, H. F., Dvorak, A. M., Manseau, E. J., Wiberg, L. & Churchill, W. H. J. natn. Cancer Inst. 62, 1459–1472 (1979).
Hunter, J. T., Ashley, M. P., Rapp, H. J., Sukumar, S. & Zbar, B. Cancer Immun. Immunother. 11, 39–44 (1981).
Weinstein, I. B., Orenstein, J. M., Gebart, R., Kaighn, M.E. & Stadier, U. C. Cancer Res. 35, 253–263 (1975).
Yamaguchi, N. & Weinstein, I. B. Proc. natn. Acad. Sci. U.S.A. 72, 214–218 (1975).
Cooper, J. A. & Hunter, T. Molec. Cell. Biol. 1, 165–178 (1981).
Otto, G., Baltimore, D. & Hynes, R. O. J. Virol. (submitted).
Hunter, T. & Sefton, B. Proc. natn. Acad. Sci. U.S.A. 77, 1311–1315 (1980).
Witte, O. N., Dasgupta, A. & Baltimore, D. Nature 283, 826–831 (1980).
Feldman, R. A., Hanafusa, T. & Hanafusa, H. Cell 22, 757–766 (1980).
Das, N. K., Hosick, H. L. & Nandi, S. J. natn. Cancer Inst. 52, 849–861 (1974).
Pickett, P. B., Pitelka, D. R., Hamamoto, S. T. & Misfeldt, D. S. J. Cell Biol. 66, 316–332 (1975).
Asch, B. B., Medina, D., Kretzer, F., Connolly, J. L. & Brinkley, B. R. Cancer Res. 40, 2383–2389 (1980).
McGrath, C. M., Maloney, T. & Soule, H. D. in Cell Biology of Breast Cancer (eds McGrath, C., Brennan, M. S. & Rich, M. A.) 373–391 (Academic, New York, 1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Senger, D., Asch, B., Smith, B. et al. A secreted phosphoprotein marker for neoplastic transformation of both epithelial and fibroblastic cells. Nature 302, 714–715 (1983). https://doi.org/10.1038/302714a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/302714a0
This article is cited by
-
Suppression of tumour growth by orally administered osteopontin is accompanied by alterations in tumour blood vessels
British Journal of Cancer (2014)
-
The epigenetic landscape of oral squamous cell carcinoma
British Journal of Cancer (2013)
-
Osteopontin: Participation in Inflammation or Mucosal Protection in Inflammatory Bowel Diseases?
Digestive Diseases and Sciences (2013)
-
Transcriptional regulation of human osteopontin promoter by histone deacetylase inhibitor, trichostatin A in cervical cancer cells
Molecular Cancer (2010)
-
Small integrin-binding ligand N-linked glycoproteins (SIBLINGs): multifunctional proteins in cancer
Nature Reviews Cancer (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.