Key Points
-
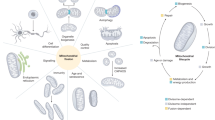
Like the Golgi apparatus, the population of peroxisomes in a cell consists of structurally and functionally distinct subcompartments (subforms) that are related through the ordered conversion of one subform to another.
-
The import of a limited subset of peroxisomal membrane proteins commits a specialized preperoxisomal endomembrane to become the earliest peroxisomal precursor (nascent peroxisome). Nascent peroxisomes can import another subset of membrane proteins and the bulk of matrix proteins, and they eventually mature into functional peroxisomes in a multistep process. In yeast and plant cells, the endoplasmic reticulum, might serve as the preperoxisomal endomembrane. The nature of the preperoxisomal endomembrane is under debate. In human cells, a preperoxisomal endomembrane might exist as an autonomous vesicular structure.
-
Many soluble and membrane-associated components of the peroxisomal machineries specific for the import of membrane and matrix proteins have been identified. These import machineries probably assemble in a temporally and spatially ordered manner in distinct intermediates along the peroxisome assembly pathway.
-
Whereas only unfolded, monomeric proteins can be translocated across the endoplasmic reticulum and mitochondrial membranes, the machinery for the import of matrix proteins can translocate completely folded polypeptides and oligomeric proteins across the peroxisomal membrane.
-
Peroxisomes undergo both constitutive (independent of extracellular stimuli) division during mitosis and regulated division induced in response to some external signal. A specific subset of peroxins coordinates the growth and regulated division of peroxisomes.
Abstract
Peroxisomes are highly adaptable organelles that carry out oxidative reactions. Distinct cellular machineries act together to coordinate peroxisome formation, growth, division, inheritance, turnover, movement and function. Soluble and membrane-associated components of these machineries form complex networks of physical and functional interactions that provide supramolecular control of the precise dynamics of peroxisome biogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lazarow, P. B. & Fujiki, Y. Biogenesis of peroxisomes. Annu. Rev. Cell Biol. 1, 489–530 (1985).
Subramani, S. Protein import into peroxisomes and biogenesis of the organelle. Annu. Rev. Cell Biol. 9, 445–478 (1993).
Bodnar, A. G. & Rachubinski, R. A. Characterization of the integral membrane polypeptides of rat liver peroxisomes isolated from untreated and clofibrate-treated rats. Biochem. Cell Biol. 69, 499–508 (1991).
Wanders, R. J. A. & Tager, J. M. Lipid metabolism in peroxisomes in relation to human disease. Mol. Aspects Med. 19, 69–154 ( 1998).
Geraghty, M. T. et al. Detecting patterns of protein distribution and gene expression in silico. Proc. Natl Acad. Sci. USA 96, 2937–2942 (1999).
Titorenko, V. I. & Rachubinski, R. A. Dynamics of peroxisome assembly and function. Trends Cell Biol. 11, 22–29 (2001).
Subramani, S., Koller, A. & Snyder, W. B. Import of peroxisomal matrix and membrane proteins . Annu. Rev. Biochem. 69, 399– 418 (2000).
Fujiki, Y. Peroxisome biogenesis and peroxisome biogenesis disorders. FEBS Lett. 476, 42–46 ( 2000).
Gould, S. J. & Valle, D. Peroxisome biogenesis disorders: genetics and cell biology. Trends Genet. 16, 340– 345 (2000).
Suzuki, Y. et al. Biosynthesis of membrane polypeptides of rat liver peroxisomes . J. Biochem. 101, 491– 496 (1987).
Imanaka, T., Shiina, Y., Takano, T., Hashimoto, T. & Osumi, T. Insertion of the 70 kDa peroxisomal membrane protein into peroxisomal membranes in vivo and in vitro. J. Biol. Chem. 271, 3706–3713 (1996).
Diestelkötter, P. & Just, W. W. In vitro insertion of the 22-kD peroxisomal membrane protein into isolated rat liver peroxisomes. J. Cell Biol. 123, 1717– 1725 (1993).
Just, W. W. & Diestelkötter, P. Protein insertion into the peroxisomal membrane. Ann. NY Acad. Sci. 804, 60–75 (1996).
Pause, B., Diestelkötter, P., Heid, H. & Just, W. W. Cytosolic factors mediate protein insertion into the peroxisomal membrane . FEBS Lett. 414, 95–98 (1997).
Dyer, J. M., McNew, J. A. & Goodman, J. M. The sorting sequence of the peroxisomal integral membrane protein PMP47 is contained within a short hydrophilic loop. J. Cell Biol. 133, 269–280 (1996).A detailed analysis of the first identified peroxisomal membrane targeting signal.
Koller, A. et al. Pex22p of Pichia pastoris, essential for peroxisomal matrix protein import, anchors the ubiquitin-conjugating enzyme, Pex4p, on the peroxisomal membrane. J. Cell Biol. 146, 99–112 (1999).
Pause, B., Saffrich, R., Hunziker, A., Ansorge, W. & Just, W. W. Targeting of the 22 kDa integral peroxisomal membrane protein. FEBS Lett. 471, 23–28 (2000).
Götte, K. et al. Pex19p, a farnesylated protein essential for peroxisome biogenesis . Mol. Cell. Biol. 18, 616– 628 (1998).
James, G. L., Goldstein, J. L., Pathak, R. K., Anderson, R. G. & Brown, M. S. PxF, a prenylated protein of peroxisomes. J. Biol. Chem. 269, 14182–14190 (1994).
Kammerer, S. et al. Genomic organization and molecular characterization of a gene encoding HsPXF, a human peroxisomal farnesylated protein. Genomics 45, 200–210 ( 1997).
Snyder, W. B. et al. Pex19p interacts with Pex3p and Pex10p and is essential for peroxisome biogenesis in Pichia pastoris. Mol. Biol. Cell 10, 1745–1761 ( 1999).
Matsuzono, Y. et al. Human PEX19: cDNA cloning by functional complementation, mutational analysis in a patient with Zellweger syndrome and potential role in peroxisomal membrane assembly. Proc. Natl Acad. Sci. USA 96, 2116–2121 (1999).
Sacksteder, K. A. et al. PEX19 binds multiple peroxisomal membrane proteins, is predominantly cytoplasmic, and is required for peroxisome membrane synthesis. J. Cell Biol. 148, 931–944 (2000).
Snyder, W. B., Koller, A., Choy, A. J. & Subramani, S. The peroxin Pex19p interacts with multiple, integral membrane proteins at the peroxisomal membrane. J. Cell Biol. 149, 1171– 1177 (2000).
Hettema, E. H., Girzalsky, W., van den Berg, M., Erdmann, R. & Distel, B. Saccharomyces cerevisiae Pex3p and Pex19p are required for proper localization and stability of peroxisomal membrane proteins. EMBO J. 19, 223– 233 (2000).
Snyder, W. B. et al. Pex17p is required for import of both peroxisome membrane and lumenal proteins and interacts with Pex19p and the peroxisome targeting signal-receptor docking complex in Pichia pastoris. Mol. Biol. Cell 10, 4005–4019 (1999).Evidence for the existence of multiple subcomplexes containing separable pools of peroxins.
Hettema, E. H., Distel, B. & Tabak, H. F. Import of proteins into peroxisomes. Biochim. Biophys. Acta 1451, 17–34 (1999).
Crookes, W. J. & Olsen, L. J. The effects of chaperones and the influence of protein assembly on peroxisomal protein import . J. Biol. Chem. 273, 17236– 17242 (1998).Evidence that cotranslational binding of a peroxisomal matrix protein to Hsp70 is required for its Hsp90-mediated post-translational import into the lumen of the plant peroxisome.
Frydman, J., Nimmesgern, E., Ohtsuka, K. & Hartl, F.-U. Folding of nascent polypeptide chains in a high molecular mass assembly with molecular chaperones. Nature 370, 111– 117 (1994).
Hettema, E. H. et al. The cytosolic DnaJ-like protein Djp1p is involved specifically in peroxisomal protein import. J. Cell Biol. 142 , 421–434 (1998).
Walton, P. A., Wendland, M., Subramani, S., Rachubinski, R. A. & Welch, W. J. Involvement of 70-kD heat-shock proteins in peroxisomal import. J. Cell Biol. 125, 1037–1046 ( 1994).
Preisig-Müller, R., Muster, G. & Kindl, H. Heat shock enhances the amount of prenylated Dnaj protein at membranes of glyoxysomes. Eur. J. Biochem. 219, 57–63 (1994).
Titorenko, V. I., Smith, J. J., Szilard, R. K. & Rachubinski, R. A. Pex20p of the yeast Yarrowia lipolytica is required for the oligomerization of thiolase in the cytosol and for its targeting to the peroxisome. J. Cell Biol. 142, 403–420 (1998).
Kragler, F., Langeder, A., Raupachova, J., Binder, M. & Hartig, A. Two independent peroxisomal targeting signals in catalase-A of Saccharomyces cerevisiae. J. Cell Biol. 120, 665–673 ( 1993).
Elgersma, Y., van Roermund, C. W. T., Wanders, R. J. A. & Tabak, H. F. Peroxisomal and mitochondrial carnitine acetyltransferases of Saccharomyces cerevisiae are encoded by a single gene. EMBO J. 14, 3472–3479 (1995).
McNew, J. A. & Goodman, J. M. The targeting and assembly of peroxisomal proteins: some old rules do not apply. Trends Biochem. Sci. 21, 54–58 ( 1996).
Zhang, J. W., Luckey, C. & Lazarow, P. B. Three peroxisome protein packaging pathways suggested by selective permeabilization of yeast mutants defective in peroxisome biogenesis . Mol. Biol. Cell 4, 1351– 1359 (1993).
Braverman, N., Dodt, G., Gould, S. J. & Valle, D. An isoform of Pex5p, the human PTS1 receptor, is required for the import of PTS2 proteins into peroxisomes. Hum. Mol. Genet. 7, 1195– 1205 (1998).
Otera, H. et al. Peroxisome targeting signal type 1 (PTS1) receptor is involved in import of both PTS1 and PTS2: studies with PEX5-defective CHO cell mutants . Mol. Cell. Biol. 18, 388– 399 (1998).
Dodt, G. et al. Mutations in the PTS1 receptor gene, PXR1, define complementation group 2 of the peroxisome biogenesis disorders. Nature Genet. 9, 115–125 (1995).
Otera, H. et al. The mammalian peroxin Pex5pL, the longer isoform of the mobile peroxisome targeting signal (PTS) type 1 transporter, translocates the Pex7p·PTS2 protein complex into peroxisomes via its initial docking site, Pex14p. J. Biol. Chem. 275, 21703–21714 (2000).Evidence that the longer isoform of the mammalian PTS1 receptor, Pex5L, binds cargo-loaded PTS2 receptor, Pex7, and is required for translocation of the Pex7–PTS2 protein complex across the peroxisomal membrane.
Matsumura, T., Otera, H. & Fujiki, Y. Disruption of the interaction of the longer isoform of Pex5p, Pex5pL, with Pex7p abolishes peroxisome targeting signal type 2 protein import in mammals. Study with a novel PEX5-impaired Chinese hamster ovary cell mutant. J. Biol. Chem. 275, 21715 –21721 (2000).
Fransen, M., Terlecky, S. R. & Subramani, S. Identification of a human PTS1 receptor docking protein directly required for peroxisomal protein import. Proc. Natl Acad. Sci. USA 95, 8087–8092 ( 1998).
Chang, C.-C., Warren, D. S., Sacksteder, K. A. & Gould, S. J. PEX12 interacts with PEX5 and PEX10 and acts downstream of receptor docking in peroxisomal matrix protein import. J. Cell Biol. 147, 761–773 (1999).
McCollum, D., Monosov, E. & Subramani, S. The pas8 mutant of Pichia pastoris exhibits the peroxisomal protein import deficiencies of Zellweger syndrome cells; the PAS8 protein binds to the COOH-terminal tripeptide peroxisomal targeting signal and is a member of the TPR protein family. J. Cell Biol. 121, 761–774 (1993).
Albertini, M. et al. Pex14p, a peroxisomal membrane protein binding both receptors of the two PTS-dependent import pathways. Cell 89, 83–92 (1997).
Girzalsky, W. et al. Involvement of Pex13p in Pex14p localization and peroxisomal targeting signal 2-dependent protein import into peroxisomes. J. Cell Biol. 144, 1151–1162 (1999).
Purdue, P. E., Yang, X. & Lazarow, P. B. Pex18p and Pex21p, a novel pair of related peroxins essential for peroxisomal targeting of the PTS2 pathway. J. Cell Biol. 143, 1859–1869 ( 1998).
Elgersma, Y. et al. The SH3 domain of the Saccharomyces cerevisiae peroxisomal membrane protein Pex13p functions as a docking site for Pex5p, a mobile receptor for the import of PTS1 containing proteins. J. Cell Biol. 135, 97–109 (1996).
Huhse, B. et al. Pex17p of Saccharomyces cerevisiae is a novel peroxin and component of the peroxisomal protein translocation machinery. J. Cell Biol. 140, 49–60 (1998).
Urquhart, A. J., Kennedy, D., Gould, S. J. & Crane, D. I. Interaction of Pex5p, the type 1 peroxisome targeting signal receptor, with the peroxisomal membrane proteins Pex14p and Pex13p. J. Biol. Chem. 275, 4127–4136 ( 2000).A detailed kinetic analysis of in vitro protein–protein interactions showing that the initial binding of the Pex5–PTS1 protein complex to its initial docking site, Pex14, might result in dissociation of the complex and promote the subsequent or concomitant interaction of cargo-free Pex5 with Pex13.
Collins, C. S., Kalish, J. E., Morrell, J. C., McCaffery, J. M. & Gould, S. J. The peroxisome biogenesis factors Pex4p, Pex22p, Pex1p, and Pex6p act in the terminal steps of peroxisomal matrix protein import. Mol. Cell. Biol. 20, 7516 –7526 (2000).
Rehling, P. et al. Pex8p, an intraperoxisomal peroxin of Saccharomyces cerevisiae required for protein transport into peroxisomes binds the PTS1 receptor Pex5p. J. Biol. Chem. 275, 3593– 3602 (2000).
van der Klei, I. J. et al. The ubiquitin-conjugating enzyme Pex4p of Hansenula polymorpha is required for efficient functioning of the PTS1 import machinery. EMBO J. 17, 3608–3618 ( 1998).
Walton, P. A., Gould, S. J., Rachubinski, R. A., Subramani, S. & Feramisco, J. R. Transport of microinjected alcohol oxidase from Pichia pastoris into vesicles in mammalian cells: involvement of the peroxisomal targeting signal. J. Cell Biol. 118, 499–508 ( 1992).
Glover, J. R., Andrews, D. W. & Rachubinski, R. A. Saccharomyces cerevisiae peroxisomal thiolase is imported as a dimer. Proc. Natl Acad. Sci. USA 91 , 10541–10545 (1994).
McNew, J. A. & Goodman, J. M. An oligomeric protein is imported into peroxisomes in vivo. J. Cell Biol. 127, 1245–1257 (1994).
Walton, P. A., Hill, P. E. & Subramani, S. Import of stably folded proteins into peroxisomes. Mol. Biol. Cell 6, 675–683 (1995).The largest nondeformable structure, a colloidal gold particle, able to traverse the peroxisomal membrane is at least 9 nm in diameter.
Goodman, J. M., Scott, C. W., Donahue, P. N. & Atherton, J. P. Alcohol oxidase assembles post-translationally into the peroxisome of Candida boidinii. J. Biol. Chem. 259, 8485–8493 (1984).
Waterham, H. R., Russell, K. A., de Vries, Y. & Cregg, J. M. Peroxisomal targeting, import, and assembly of alcohol oxidase in Pichia pastoris. J. Cell. Biol. 139, 1419– 1431 (1997).
Corpas, F. J. & Trelease, R. N. The plant 73 kDa peroxisomal membrane protein (PMP73) is immunorelated to molecular chaperones. Eur. J. Cell Biol. 73, 49–57 (1997).
Wimmer, B., Lottspeich, F., van der Klei, I., Veenhuis, M. & Gietl, C. The glyoxysomal and plastid molecular chaperones (70-kDa heat shock protein) of watermelon cotyledons are encoded by a single gene. Proc. Natl Acad. Sci. USA 94, 13624–13629 (1997).
Teter, S. A. & Klionsky, D. J. How to get a folded protein across a membrane. Trends Cell Biol. 9, 428–431 (1999).
Heinemann, P. & Just, W. W. Peroxisomal protein import. In vivo evidence for a novel translocation competent compartment. FEBS Lett. 300, 179–182 (1992).The first demonstration that newly synthesized peroxisomal proteins are imported primarily into small peroxisomal vesicles of intermediate buoyant density that subsequently convert to mature peroxisomes of high density.
Lüers, G., Hashimoto, T., Fahimi, H. D. & Völkl, A. Biogenesis of peroxisomes: isolation and characterization of two distinct peroxisomal populations from normal and regenerating rat liver. J. Cell Biol. 121, 1271–1280 (1993).
Wilcke, M., Hultenby, K. & Alexson, S. E. H. Novel peroxisomal populations in subcellular fractions from rat liver. Implications for peroxisome structure and biogenesis. J. Biol. Chem. 270, 6949–6958 (1995).
Titorenko, V. I., Eitzen, G. A. & Rachubinski, R. A. Mutations in the PAY5 gene of the yeast Yarrowia lipolytica cause the accumulation of multiple subpopulations of peroxisomes. J. Biol. Chem. 271, 20307 –20314 (1996).
South, S. T. & Gould, S. J. Peroxisome synthesis in the absence of preexisting peroxisomes. J. Cell Biol. 144, 255–266 (1999).A model for two overlapping, multistep pathways of peroxisome assembly in human fibroblasts.
Titorenko, V. I., Chan, H. & Rachubinski, R. A. Fusion of small peroxisomal vesicles in vitro reconstructs an early step in the in vivo multistep peroxisome assembly pathway of Yarrowia lipolytica. J. Cell Biol. 148, 29–43 (2000). Evidence for the existence of a multistep peroxisome assembly pathway that operates by the ordered conversion of structurally distinct peroxisomal subforms to mature peroxisomes.
Titorenko, V. I. & Rachubinski, R. A. Peroxisomal membrane fusion requires two AAA family ATPases, Pex1p and Pex6p. J. Cell Biol. 150, 881–886 (2000).
Titorenko, V. I. & Rachubinski, R. A. Mutants of the yeast Yarrowia lipolytica defective in protein exit from the endoplasmic reticulum are also defective in peroxisome biogenesis. Mol. Cell. Biol. 18, 2789–2803 (1998).
Waterham, H. R., Titorenko, V. I., Swaving, G. J., Harder, W. & Veenhuis, M. Peroxisomes in the methylotrophic yeast Hansenula polymorpha do not necessarily derive from pre-existing organelles. EMBO J. 12, 4785– 4794 (1993).
Honsho, M. et al. Mutation in PEX16 is causal in the peroxisome-deficient Zellweger syndrome of complementation group D. Am. J. Hum. Genet. 63, 1622–1630 ( 1998).
Baerends, R. J. S. et al. The Hansenula polymorpha PER9 gene encodes a peroxisomal membrane protein essential for peroxisome assembly and integrity . J. Biol. Chem. 271, 8887– 8894 (1996).
South, S. T., Sacksteder, K. A., Li, X., Liu, Y. & Gould, S. J. Inhibitors of COPI and COPII do not block PEX3-mediated peroxisome synthesis. J. Cell Biol. 149, 1345–1359 ( 2000).
Titorenko, V. I. & Rachubinski, R. A. The endoplasmic reticulum plays an essential role in peroxisome biogenesis. Trends Biochem. Sci. 23, 231–233 (1998).
Mullen, R. T., Lisenbee, C. S., Miernyk, J. A. & Trelease, R. N. Peroxisomal membrane ascorbate peroxidase is sorted to a membranous network that resembles a subdomain of the endoplasmic reticulum. Plant Cell 11, 2167–2185 ( 1999).Some yeast and plant peroxisomal membrane proteins are inserted post-translationally in vitro into membranes of the endoplasmic reticulum but not into membranes of peroxisomes or of any other purified organelle.
Elgersma, Y. et al. Overexpression of Pex15p, a phosphorylated peroxisomal integral membrane protein required for peroxisome assembly in S. cerevisiae, causes proliferation of the endoplasmic reticulum membrane. EMBO J. 16, 7326–7341 ( 1997).
Mullen, R. T. & Trelease, R. N. The sorting signals for peroxisomal membrane-bound ascorbate peroxidase are within its C-terminal tail. J. Biol. Chem. 275, 16337–16344 (2000).
Marshall, P. A., Dyer, J. M., Quick, M. E. & Goodman, J. M. Redox-sensitive homodimerization of Pex11p: a proposed mechanism to regulate peroxisomal division. J. Cell Biol. 135, 123–137 (1996).A detailed analysis of the mechanism of Pex11 action in regulating division of yeast peroxisomes from within the organelle.
Tan, X., Waterham, H. R., Veenhuis, M. & Cregg, J. M. The Hansenula polymorpha PER8 gene encodes a novel peroxisomal integral membrane protein involved in proliferation. J. Cell Biol. 128, 307–319 (1995).
Veenhuis, M. & Goodman, J. M. Peroxisomal assembly: membrane proliferation precedes the induction of the abundant matrix proteins in the methylotrophic yeast Candida boidinii. J. Cell Sci. 96, 583–590 (1990).
Erdmann, R. & Blobel, G. Giant peroxisomes in oleic acid-induced Saccharomyces cerevisiae lacking the peroxisomal membrane protein Pmp27p . J. Cell Biol. 128, 509– 523 (1995).
Marshall, P. A. et al. Pmp27 promotes peroxisomal proliferation. J. Cell Biol. 129, 345–355 ( 1995).
Schrader, M. et al. Expression of PEX11β mediates peroxisome proliferation in the absence of extracellular stimuli. J. Biol. Chem. 273, 29607–29614 (1998).
van Roermund, C. W. T., Tabak, H. F., van den Berg, M., Wanders, R. J. A. & Hettema, E. H. Pex11p plays a primary role in medium-chain fatty acid oxidation, a process that affects peroxisome number and size in Saccharomyces cerevisiae. J. Cell Biol. 150, 489–497 (2000).
Lorenz, P., Maier, A. G., Baumgart, E., Erdmann, R. & Clayton, C. Elongation and clustering of glycosomes in Trypanosoma brucei overexpressing the glycosomal Pex11p. EMBO J. 17, 3542–3555 ( 1998).
Passreiter, M. et al. Peroxisome biogenesis: involvement of ARF and coatomer. J. Cell Biol. 141, 373–383 (1998).Evidence that the recruitment of ARF and coatomer by Pex11, which resides on the cytosolic face of mammalian peroxisomes, might initiate fission and/or budding of these organelles followed by their division.
Chang, C.-C. et al. Metabolic control of peroxisome abundance. J. Cell Sci. 112, 1579–1590 ( 1999).
Poll-Thé, B. T. et al. A new peroxisomal disorder with enlarged peroxisomes and a specific deficiency of acyl-CoA oxidase (pseudo-neonatal adrenoleukodystrophy) . Am. J. Hum. Genet. 42, 422– 434 (1988).
Nakagawa, T. et al. Peroxisomal membrane protein Pmp47 is essential in the metabolism of middle-chain fatty acid in yeast peroxisomes and is associated with peroxisome proliferation. J. Biol. Chem. 275, 3455– 3461 (2000).
Fan, C.-Y. et al. Steatohepatitis, spontaneous peroxisome proliferation and liver tumors in mice lacking peroxisomal fatty acyl-CoA oxidase. Implications for peroxisome proliferator-activated receptor α natural ligand metabolism . J. Biol. Chem. 273, 15639– 15645 (1998).
Smith, J. J., Brown, T. W., Eitzen, G. A. & Rachubinski, R. A. Regulation of peroxisome size and number by fatty acid β-oxidation in the yeast Yarrowia lipolytica. J. Biol. Chem. 275, 20168–20178 (2000).
Baerends, R. J. S., Salomons, F. A., Kiel, J. A. K. W., van der Klei, I. J. & Veenhuis, M. Deviant Pex3p levels affect normal peroxisome formation in Hansenula polymorpha: a sharp increase of the protein level induces the proliferation of numerous, small protein-import competent peroxisomes. Yeast 13 , 1449–1463 (1997).
Eitzen, G. A., Szilard, R. K. & Rachubinski, R. A. Enlarged peroxisomes are present in oleic acid-grown Yarrowia lipolytica overexpressing the PEX16 gene encoding an intraperoxisomal peripheral membrane peroxin. J. Cell Biol. 137, 1265–1278 (1997).
Acknowledgements
We would like to thank N.-H. Chua, I. Coppens, G. Jedd and F.R. Opperdoes for providing images for Fig. 1 and for helpful discussion. This work was supported by grants from the Canadian Institutes of Health Research (CIHR) to R.A.R. R.A.R. is a Senior Scientist of the CIHR and an International Research Scholar of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Related links
Related links
DATABASE LINKS
peroxisome biogenesis disorders
FURTHER INFORMATION
ENCYCLOPEDIA OF LIFE SCIENCES
Protein import into peroxisomes
Glossary
- PEROXINS
-
Proteins required for various aspects of peroxisome (microbody) biogenesis, including assembly of the peroxisomal membrane, import of peroxisomal matrix proteins, peroxisome proliferation and peroxisome inheritance.
- mPTS1
-
Peroxisomal membrane targeting signal 1 for post-translational targeting of peroxisomal membrane proteins directly from the cytosol. There is no clear sequence consensus for the mPTS1, but several motifs have been reported, including (K/R)(K/R)X3–7(T/S)X2(D/E) and (Y)X3(L)X3(P)X3(K/Q/N).
- mPTS2
-
Peroxisomal membrane targeting signal 2 for targeting of peroxisomal membrane proteins indirectly from the endoplasmic reticulum (ER). The mPTS2 acts in the ER lumen. Two similar consensus motifs have been reported, namely (R/K)X(K/R)X (K/R)X(L/I)X9–10(F/Y) and (L/I/V)X(R)X(K/R)X(K)X(L/I)
- PTS1
-
Carboxy-terminal peroxisomal targeting signal 1 found in most peroxisomal matrix proteins. It is a tripeptide with the consensus motif (S/A/C)(K/R/H)(L/M/I).
- PTS2
-
Peroxisomal targeting signal 2 that has been found so far at the amino termini of four peroxisomal matrix proteins. It is a nonapeptide with the consensus motif (R/K)(L/V/I)X5(H/Q)(L/A).
- CHAPERONE
-
Protein that mediates assembly of another polypeptide-containing structure, but does not form part of the completed structure, or participate in its biological function.
- PREPEROXISOMAL ENDOMEMBRANE
-
An endomembrane committed to become the earliest peroxisomal precursor (the nascent peroxisome), and not another organelle, by uptake of a limited subset of peroxisomal membrane proteins. Also called early preperoxisome.
- NASCENT PEROXISOME
-
The earliest peroxisomal precursor formed by uptake of a limited subset of peroxisomal membrane proteins into a preperoxisomal endomembrane and that can import an additional set of membrane proteins to become an early peroxisomal precursor. Also called late preperoxisome.
- HSP
-
(Heat-shock protein). Protein induced in response to elevated temperature, classified according to its size. Functions as a molecular chaperone.
- TPR
-
The tetratricopeptide repeat motif consists of a highly degenerate 34-amino-acid repeat believed to form an interlocking series of α-helices. The TPR is thought to mediate protein–protein interactions, preferably with proteins that have WD-40 repeats.
- WD-40
-
Repeat of ∼40 amino acids with a characteristic central Trp–Asp motif. WD-40 proteins are often associated with TPR- repeat proteins.
- RING
-
A cysteine-rich 'RING' finger domain of 40 to 60 amino acids, also called the C3HC4 Zn finger, which binds two atoms of Zn and might mediate protein–protein interactions. Most RING-finger proteins have been shown to bind DNA.
- AAA ATPASES
-
ATPases associated with different cellular activities that contain one or two AAA motifs of 230 to 250 amino acids, including the Walker homology sequences of P-loop ATPases and regions of similarity unique to AAA proteins.
- SH3
-
60-amino-acid Src homology 3 domain that mediates the assembly of specific protein complexes through binding to proline-rich peptides.
- MATURE PEROXISOMES
-
Metabolically active, functional peroxisomes that contain the complete complement of membrane and matrix proteins and membrane lipids.
Rights and permissions
About this article
Cite this article
Titorenko, V., Rachubinski, R. The life cycle of the peroxisome. Nat Rev Mol Cell Biol 2, 357–368 (2001). https://doi.org/10.1038/35073063
Issue Date:
DOI: https://doi.org/10.1038/35073063
This article is cited by
-
Genomic features of the polyphagous cotton leafworm Spodoptera littoralis
BMC Genomics (2022)
-
Proteomics analysis reveals the effect of 1α,25(OH)2VD3-glycosides on development of early testes in piglets
Scientific Reports (2021)
-
MrPEX33 is involved in infection-related morphogenesis and pathogenicity of Metarhizium robertsii
Applied Microbiology and Biotechnology (2021)
-
A defect in the peroxisomal biogenesis in germ cells induces a spermatogenic arrest at the round spermatid stage in mice
Scientific Reports (2019)
-
FgPEX4 is involved in development, pathogenicity, and cell wall integrity in Fusarium graminearum
Current Genetics (2019)