Abstract
It has been reported that the population of regulatory T cells (T regs) is increased in tumour-infiltrating lymphocytes in cancer-bearing hosts. Recently, forkhead/winged helix transcription factor p3, Foxp3, is thought to be the most reliable marker of T regs. In the present study, we investigated the prevalence and localisation pattern of Foxp3+ cells in gastric cancer (n=80) by immunohistochemistry, in relation to the clinical outcome of gastric cancer patients. Immunohistochemical staining was performed with anti-Foxp3 mAb, and Foxp3+ cells were semiquantified. We divided all cases into two groups: Foxp3+-high (n=40) and Foxp3+-low (n=40) groups, by the median size of the population of Foxp3+ cells. Furthermore, in terms of the localisation pattern of accumulating Foxp3+ cells in tumours, we classified all cases into three groups: a peri-tumour group (n=30), a diffuse group (n=40), and a follicular group (n=10). As a result, although the populations of Foxp3+ cells in stage IV were significantly larger than those in stage I (P<0.05), there was no significant difference in survival between the patients with high and low population levels of Foxp3+ cells. However, survival in patients with a diffuse pattern of Foxp3+ cells was significantly poorer than in those with a peri-tumoral pattern. In conclusion, the localisation pattern, but not the population size, of Foxp3+ cells was significantly related to patient survival.
Similar content being viewed by others
Main
Regulatory T cells (T regs) are one of the T-cell subsets, which play important roles in immunological self-tolerance (Sakaguchi et al, 1995; Jonuleit et al, 2001; Ng et al, 2001; Beyer and Schultze, 2006). They have a functionally immunosuppressive property that inhibits effector cells from acting against self in autoimmune diseases or a tumour (Sakaguchi et al, 1995; Jonuleit et al, 2001; Ng et al, 2001; Beyer and Schultze, 2006). They constitutively express CD25 (IL-2 receptor α-chain), CD45RO, glucocorticoid-induced tumour-necrosis factor receptor-related protein, and cytotoxic T-lymphocyte-associated antigen-4 (Sakaguchi et al, 1995; Dieckmann et al, 2001; Beyer and Schultze, 2006). Recently, it has been reported that Foxp3, forkhead/winged helix transcription factor, is a reliable marker of T regs (Hori and Sakaguchi, 2004; Yagi et al, 2004). Therefore, it is possible to define T regs more strictly as CD4+CD25+Foxp3+ cells.
In mice, it is known that autoimmune diseases, such as ulcerative colitis or Crohn's disease, occur due to the depletion of T regs (Hori and Sakaguchi, 2004; Sakaguchi et al, 2006). Also in humans, immune dysregulation polyendocrinopathy enteropathy X-linked syndrome is an autoimmune disease due to a deficiency of T regs (Kelsen et al, 2005; Dejaco et al, 2006; Sakaguchi et al, 2006; Takahashi et al, 2006). These observations indicated that T regs play important roles in immunological homeostasis. Although the mechanisms of suppression by T regs are still unclear, it has been reported that T regs can inhibit the function of effector T cells directly through cell-to-cell contact or indirectly via the secretion of immune-suppressive cytokines, and also suppress the Ag-presenting function of dendritic or NK cells (Dieckmann et al, 2002; Misra et al, 2004; Earle et al, 2005; Longhi et al, 2006; Smyth et al, 2006).
Recently, many studies in murine models have shown that the depletion of T regs can amplify antitumour immunity (Viehl et al, 2006; Rudge et al, 2007). Moreover, in humans, many studies have revealed that the population of T regs in tumour-infiltrating lymphocytes (TILs) is significantly larger than in normal tissues in several malignancies (Woo et al, 2001; Liyanage et al, 2002; Ormandy et al, 2005; Badoual et al, 2006; Petersen et al, 2006). We have reported that the frequency of T regs among TILs, tumour-draining regional lymph nodes, and peripheral blood lymphocytes is higher in gastric and oesophageal cancer patients than that in their normal counterparts (Ichihara et al, 2003). Importantly, after patients received curative resections of gastric cancers, the increased proportion of T regs was significantly reduced, and the levels were almost equal to those in normal healthy donors (Kono et al, 2006). These results strongly suggest that tumour-related factors induce and expand T regs. There is, however, still limited information describing the mechanisms behind T reg accumulation within cancer microenvironments and their expansion locoregionally. Thus, it is important to evaluate the localisation of infiltrating T regs in relation to the clinical outcome.
In the current study, we investigated the population and localisation pattern of Foxp3+ T regs in gastric cancer by immunohistochemistry, and evaluated the relationship between the findings and clinical outcome.
Materials and methods
Patients and samples
Eighty patients with gastric cancer, who were operated on in the University of Yamanashi Hospital from 1997 to 1998, were enrolled in the present study. The median follow-up time of the patients was 87.7 months. The characteristics of the study subjects are summarised in Table 1. None of the patients received radiotherapy, chemotherapy, or other medical interventions before the study. This study was approved by the Ethical Committee of the University of Yamanashi, and written informed consent was obtained from all individuals.
Immunohistochemical analysis
Foxp3 staining was conducted using the avidin–biotin–peroxidase complex method with paraffin-embedded, 4-μm-thick sections of gastric cancer. Briefly, each paraffin section was dewaxed, followed by antigen retrieval with Epitope Retrieval Solution (10 mmol citrate buffer (pH 6.0), Dakocytomation, Glostrup, Denmark) in a preheated water bath (98°C, 40 min), and endogenous peroxidase was blocked by ChemMate Peroxidase Blocking Solution (Dako). Then, biotinylated anti-human Foxp3 antibody (diluted by PBS, 1 : 20; eBioscience, San Diego, CA, USA) was applied for 40 min at room temperature. Thereafter, the sections were incubated with streptavidin-conjugated horseradish peroxidase (Dako) for 10 min, followed by development with 3,3′-diaminobenzidine (Dako) for 5 min and counter-staining with haematoxylin. Negative control staining was performed with isotype control, mouse IgG2a (Dako), instead of the primary antibody.
Quantitative evaluation of Foxp3+ cells was analysed in five randomly selected areas at a magnification of × 400 by two observers (YM and KK) in a blinded manner. Then, all cases with gastric cancer were divided into two groups: Foxp3+-high and Foxp3+-low groups, classified by the median value of the total population of Foxp3+ cells (median=34.5, range=2–525).
Furthermore, localisation patterns of infiltrating Foxp3+ cells in the tumour were divided into three groups: a peri-tumour group, a diffuse group, and a follicular group. The peri-tumour group was defined as the population of Foxp3+ cells in the peri-tumoral region that had increased more than five-fold compared to that in the central region of the tumour at a magnification of × 400, while the diffuse group was defined as the difference in the Foxp3+ cells between the peri-tumoral region and central region of the tumour that was less than five-fold. Moreover, the follicular group was defined as the population of Foxp3+ cells that mainly occupied the lymphoid follicles of the submucosal layer compared to any other region of the tumour.
Statistical analysis
Actuarial overall survival rates were analysed by the Kaplan–Meier method, and survival was measured in days from the operation to death or the last review. Differences between survival curves were analysed by the log-rank test. Deviation in immunohistochemical patterns was evaluated by the χ2 test.
To assess the correlation between survival time and multiple clinicopathologic variables, univariate and multivariate analyses were conducted using Cox's proportional hazards model. Differences were considered significant at P<0.05. All statistical analyses were performed with StatView-J 5.0 software (Abacus Concepts, Berkeley, CA, USA).
Results
Immunohistochemical analysis of Foxp3+ cells in gastric cancer
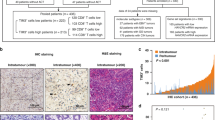
Foxp3+ cells were specifically identified and semiquantified by immunohistochemistry (Figure 1A–C). Then, localisation patterns of infiltrating Foxp3+ cells were divided into three groups: a peri-tumour group (Figure 1A, n=30), diffuse group (Figure 1B, n=40), and follicular group (Figure 1C, n=10), in terms of where Foxp3+ cells dominantly occupied the lesion by immunohistochemistry as described in Materials and Methods. There was a very small population of Foxp3+ cells in normal gastric mucosa in the same specimens (Table 2). The population of Foxp3+ cells in stage IV was significantly larger than those in stage I (Table 2, P<0.05). There were no significant differences in the prevalence of Foxp3+ cells among T factors, N factors, or between histological classifications (Table 2).
Localisation pattern of accumulating Foxp3+ cells in gastric cancer by immunohistochemistry. Localisation patterns of infiltrating Foxp3+ cells in the tumour were divided into three groups: a peri-tumour group (A, n=30), a diffuse group (B, n=40), and a follicular group (C, n=10). Representative staining is demonstrated. The peri-tumour group was defined as the population of Foxp3+ cells in the peri-tumoral region that had increased more than five-fold compared to that in the central region of the tumour at a magnification of × 400, while the diffuse group was defined as the difference in the Foxp3+ cells between the peri-tumoral region and central region of the tumour that was less than five-fold. The follicular group was defined as the population of Foxp3+ cells that mainly occupied lymphoid follicles of the submucosal layer compared to any other region of the tumour (original magnification: × 4, × 40).
Interestingly, tumours with a diffuse distribution pattern of Foxp3+ cells were more frequent in stages II+III+IV, while tumours with a peri-tumoral distribution pattern of Foxp3+ cells were more frequent in stage I (P<0.05 by the χ2 test, Table 2). There were no significant differences in the localisation pattern of Foxp3+ cells among T factors or between histological classifications, although there was significant difference in the localisation pattern of Foxp3+ cells among N factors (Table 2).
The frequency and distribution pattern of Foxp3+ cells relating to survival in gastric cancer
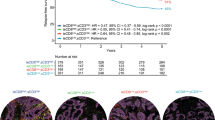
While the grade of the T factor (Figure 2A), N factor (Figure 2B), and stage classification (Figure 2C) was significantly correlated with the survival of patients, there was no significant difference in survival between patients with large and small populations of Foxp3+ cells (Figure 2D). However, the survival rate in patients with a diffuse pattern of Foxp3+ cells was significantly poorer than in those with a peri-tumoral pattern (Figure 2E). These results indicated that the localisation pattern, but not the population size, of Foxp3+ cells was significantly related to patient survival.
The localisation pattern, but not the population size, of Foxp3+ cells was significantly related to patient survival. The median survival time in early disease was significantly longer than that in advanced disease in terms of the pathologic tumour status (A), pathologic node status (B), and stage (C) (n=80, P<0.05, by the log-rank test). (D) Foxp3+-high and Foxp3+-low groups were classified by the median value of the population of Foxp3+ cells (median value=34.5, range=2–525). NS: not significant. (E) Localisation patterns of infiltrating Foxp3+ cells in the tumour were divided into three groups: a peri-tumour group (n=30), a diffuse group (n=40), and a follicular group (n=10).
To further assess whether the localisation pattern of Foxp3+ cells represented a prognostic parameter, we used Cox's proportional hazards model. The covariate parameters included several clinicopathologic factors, as shown in Table 3. On univariate analysis, a diffuse group in terms of the localisation of Foxp3+ cells showed a significantly higher hazard ratio for a poor prognosis (vs the peri-tumoral group, hazard ratio=4.65 (1.35–15.96), P<0.02), although multivariate analysis revealed that the localisation pattern of Foxp3+ cells was not an independent prognostic factor (P=0.39, Table 3).
Discussion
In general, T regs have functionally suppressive actions on other effector T cells (Sakaguchi et al, 1995; Dieckmann et al, 2001; Jonuleit et al, 2001; Ng et al, 2001; Earle et al, 2005; Beyer and Schultze, 2006). They have been characterised as a CD4+CD25high population among CD4+ T cells (Sakaguchi et al, 1995; Dieckmann et al, 2001; Jonuleit et al, 2001; Ng et al, 2001; Earle et al, 2005; Beyer and Schultze, 2006). However, it was difficult to discriminate T regs from conventional effector T cells that express CD25 intermediately, because effector T cells continuously express CD25 on their cell surface (Zheng et al, 2004; Fontenot et al, 2005; Allan et al, 2007; Pillai et al, 2007). Recently, it has been reported that Foxp3, forkhead/winged helix transcription factor, is the most reliable marker of T regs (Hori and Sakaguchi, 2004; Yagi et al, 2004; Fontenot et al, 2005). Therefore, it is possible to define T regs more strictly as CD4+CD25+Foxp3+ cells. Moreover, Foxp3 expression is crucial regarding whether the cells have a virtually suppressive function. Thus, Foxp3+ cells were analysed by immunohistochemistry in order to evaluate T regs in gastric cancer.
Previous studies in human malignancies reported that the prevalence of T regs defined as a CD4+CD25+ population was significantly increased in TILs compared to the normal counterparts (Woo et al, 2001; Liyanage et al, 2002; Ormandy et al, 2005; Badoual et al, 2006; Petersen et al, 2006). Furthermore, the high prevalence of CD4+CD25+ T regs is closely associated with a poor prognosis in ovarian (Curiel et al, 2004) or pancreatic (Liyanage et al, 2002) cancer. Recently, intratumoral Foxp3+ T regs in ovarian (Wolf et al, 2005) or pancreatic (Hiraoka et al, 2006) cancer were correlated with a poor prognosis (Wolf et al, 2005; Hiraoka et al, 2006). In particular, multivariate analysis in pancreatic cancer (Hiraoka et al, 2006) showed that the prevalence of Foxp3+ T regs was an independent prognostic factor. On the contrary, it has been reported that the clinical outcome was not dependent on the prevalence of Foxp3+ T regs in TILs in renal cell carcinoma (Siddiqui et al, 2007). Thus, in human malignancies, it remains controversial as to whether infiltrating T regs, in particular Foxp3+ T regs, are related to the clinical outcome.
In the current study, we analysed the prevalence of Foxp3+ cells in gastric cancer by immunohistochemistry. The prevalence of Foxp3+ cells was significantly increased in the tumour lesion compared to normal gastric tissue. Moreover, the population of Foxp3+ cells in stage IV was significantly larger than those in stage I. However, the prevalence of Foxp3+ cells is not significantly associated with the overall survival of patients with gastric cancer, in contrast with previous reports on ovarian carcinoma (Wolf et al, 2005) and pancreatic cancer (Hiraoka et al, 2006). The present study is, to our knowledge, the first report describing the prevalence of Foxp3+ T regs related to the prognosis in gastric cancer.
With regards to gastric mucosa, there are several reports describing the presence of Helicobacter pylori infection-associated Foxp3+ T regs (Lundgren et al, 2005; Enarsson et al, 2006). Since, in the present cohort with gastric cancer, there was no information on H. pylori infection, it is impossible to evaluate the influence of this infection. Thus, the presence of H. pylori infection may lead to complications in the evaluation of Foxp3+ T regs in tumoral immunity of gastric cancer.
Of note, we found that the patients with a predominant localisation of Foxp3+ cells in the peri-tumoral region had a better prognosis than those showing a diffuse localisation of Foxp3+ cells. Since the precise mechanisms inducing and expanding T regs remain unclear, the biological difference between the peri-tumoral and diffuse localisation of Foxp3+ cells is under investigation. It is possible to presume that a tumour-related factor may induce an accumulation of Foxp3+ T regs in the peri-tumoral region at the early stage of gastric cancer, since stage I patients with a peri-tumoral localisation pattern were significantly more frequent. For example, the cancer–stromal reaction including the migration factors for Foxp3+ T regs may differ within tumours, leading to the difference in the localisation pattern of Foxp3+ T regs. That is, the migration factors for Foxp3+ T regs were mainly produced in the peri-tumoral region as a result of the tumour–stromal reaction at the early stage of gastric cancer.
In conclusion, the prevalence of Foxp3+ cells is not significantly associated with the overall survival of patients with gastric cancer. However, the patients with a predominant localisation of Foxp3+ cells in the peri-tumoral region had a better prognosis than those showing the diffuse localisation of Foxp3+ cells. A better understanding of the underlying mechanism of T reg regulation or a strategy for controlling T regs may lead to a novel therapeutic strategy for gastric cancer.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Allan SE, Crome SQ, Crellin NK, Passerini L, Steiner TS, Bacchetta R, Roncarolo MG, Levings MK (2007) Activation-induced FOXP3 in human T effector cells does not suppress proliferation or cytokine production. Int Immunol 19: 345–354
Badoual C, Hans S, Rodriguez J, Peyrard S, Klein C, Agueznay Nel H, Mosseri V, Laccourreye O, Bruneval P, Fridman WH, Brasnu DF, Tartour E (2006) Prognostic value of tumor-infiltrating CD4+ T-cell subpopulations in head and neck cancers. Clin Cancer Res 12: 465–472
Beyer M, Schultze JL (2006) Regulatory T cells in cancer. Blood 108: 804–811
Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, Evdemon-Hogan M, Conejo-Garcia JR, Zhang L, Burow M, Zhu Y, Wei S, Kryczek I, Daniel B, Gordon A, Myers L, Lackner A, Disis ML, Knutson KL, Chen L, Zou W (2004) Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 10: 942–949
Dejaco C, Duftner C, Grubeck-Loebenstein B, Schirmer M (2006) Imbalance of regulatory T cells in human autoimmune diseases. Immunology 117: 289–300
Dieckmann D, Bruett CH, Ploettner H, Lutz MB, Schuler G (2002) Human CD4+CD25+ regulatory, contact-dependent T cells, induce interleukin 10-producing, contact-independent type 1-like regulatory T cells. J Exp Med 196: 247–253
Dieckmann D, Plottner H, Berchtold S, Berger T, Schuler G (2001) Ex vivo isolation and characterization of CD4(+)CD25(+) T cells with regulatory properties from human blood. J Exp Med 193: 1303–1310
Earle KE, Tang Q, Zhou X, Liu W, Zhu S, Bonyhadi ML, Bluestone JA (2005) In vitro expanded human CD4+CD25+ regulatory T cells suppress effector T cell proliferation. Clin Immunol 115: 3–9
Enarsson K, Lundgren A, Kindlund B, Hermansson M, Roncador G, Banham AH, Lundun BS, Quiding-Järbrink M (2006) Function and recruitment of mucosal regulatory T cells in human chronic Helicobacter pylori infection and gastric adenocarcinoma. Clin Immunol 121: 358–368
Fontenot JD, Rasmussen JP, Williams LM, Dooley JL, Farr AG, Rudensky AY (2005) Regulatory T cell lineage specification by the forkhead transcription factor Foxp3. Immunity 22: 329–341
Hiraoka N, Onozato K, Kosuge T, Hirohashi S (2006) Prevalence of FOXP3+ regulatory T cells increased during the progression of pancreatic ductal adenocarcinoma and its premalignant lesions. Clin Cancer Res 12: 5423–5434
Hori S, Sakaguchi S (2004) Foxp3: a critical regulator of the development and function of regulatory T cells. Microbes Infect 6: 745–751
Ichihara F, Kono K, Takahashi A, Kawaida H, Sugai H, Fujii H (2003) Increased populations of regulatory T cells in peripheral blood and tumor-infiltrating lymphocytes in patient with gastric and esophageal cancers. Clin Cancer Res 9: 4404–4408
Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma, 2nd (English edition). Gastric Cancer 1: 10–24
Jonuleit H, Schmitt E, Stassen M, Tuettenberg A, Knop J, Enk AH (2001) Identification and functional characterization of human CD4(+)CD25(+) T cells with regulatory properties isolated from peripheral blood. J Exp Med 193: 1285–1294
Kelsen J, Agnholt J, Hoffmann HJ, Romer JL, Hvas CL, Dahlerup JF (2005) Foxp3+CD4+CD25+ T cells with regulatory properties can be cultured from colonic mucosa of patients with Crohn's disease. Clin Exp Immunol 141: 549–557
Kono K, Kawaida H, Takahashi A, Sugai H, Mimura K, Miyagawa N, Omata H, Fujii H (2006) CD4(+)CD25high regulatory T cells increase with tumor stage in patients with gastric and esophageal cancers. Cancer Immunol Immunother 55: 1064–1071
Liyanage UK, Moore TT, Joo HG, Tanaka Y, Herrmann V, Doherty G, Drebin JA, Strasberg SM, Eberlein TJ, Goedegebuure PS, Linehan DC (2002) Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol 169: 2756–2761
Longhi MS, Hussain MJ, Mitry RR, Arora SK, Mieli-Vergani G, Vergani D, Ma Y (2006) Functional study of CD4+CD25+ regulatory T cells in health and autoimmune hepatitis. J Immunol 176: 4484–4491
Lundgren A, Strömberg E, Sjöling A, Lindholm C, Enarsson K, Edebo A, Johnsson E, Suri-Payer E, Larsson P, Rudin A, Svennerholm AM, Lundin BS (2005) Mucosal FOXP3-expressing CD4+ CD25high regulatory T cells in Helicobacter pylori-infected patients. Infect Immun 73: 523–531
Misra N, Bayry J, Lacroix-Desmazes S, Kazatchkine MD, Kaveri SV (2004) Cutting edge: human CD4+CD25+ T cells restrain the maturation and antigen-presenting function of dendritic cells. J Immunol 172: 4676–4680
Ng WF, Duggan PJ, Ponchel F, Matarese G, Lombardi G, Edwards AD, Issacs JD, Lechler RI (2001) Human CD4(+)CD25(+) cells: a naturally occurring population of regulatory T cells. Blood 98: 2736–2744
Ormandy LA, Hillemann T, Wedemeyer H, Manns MP, Greten TF, Korangy F (2005) Increased populations of regulatory T cells in peripheral blood of patients with hepatocellular carcinoma. Cancer Res 65: 2457–2464
Petersen RP, Campa MJ, Sperlazza J, Conlon D, Joshi MB, Harpole Jr DH, Patz Jr EF (2006) Tumor infiltrating FOXP3+ regulatory T-cells are associated with recurrence in pathologic stage I NSCLC patients. Cancer 107: 2866–2872
Pillai V, Ortega SB, Wang CK, Karandikar NJ (2007) Transient regulatory T-cells: a state attained by all activated human T-cells. Clin Immunol 123: 18–29
Rudge G, Barrett SP, Scott B, van Driel IR (2007) Infiltration of a mesothelioma by IFN-gamma-producing cells and tumor rejection after depletion of regulatory T cells. J Immunol 178: 4089–4096
Sakaguchi S, Ono M, Setoguchi R, Yagi H, Hori S, Fehervari Z, Shimizu J, Takahashi T, Nomura T (2006) Foxp3+CD25+CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol Rev 212: 8–27
Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M (1995) Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 155: 1151–1164
Siddiqui SA, Frigola X, Bonne-Annee S, Mercader M, Kuntz SM, Krambeck AE, Sengupta S, Dong H, Cheville JC, Lohse CM, Krco CJ, Webster WS, Leibovich BC, Blute ML, Knutson KL, Kwon ED (2007) Tumor-infiltrating Foxp3−CD4+CD25+ T cells predict poor survival in renal cell carcinoma. Clin Cancer Res 13: 2075–2081
Smyth MJ, Teng MW, Swann J, Kyparissoudis K, Godfrey DI, Hayakawa Y (2006) CD4+CD25+ T regulatory cells suppress NK cell-mediated immunotherapy of cancer. J Immunol 176: 1582–1587
Takahashi M, Nakamura K, Honda K, Kitamura Y, Mizutani T, Araki Y, Kabemura T, Chijiiwa Y, Harada N, Nawata H (2006) An inverse correlation of human peripheral blood regulatory T cell frequency with the disease activity of ulcerative colitis. Dig Dis Sci 51: 677–686
Viehl CT, Moore TT, Liyanage UK, Frey DM, Ehlers JP, Eberlein TJ, Goedegebuure PS, Linehan DC (2006) Depletion of CD4+CD25+ regulatory T cells promotes a tumor-specific immune response in pancreas cancer-bearing mice. Ann Surg Oncol 13: 1252–1258
Wolf D, Wolf AM, Rumpold H, Fiegl H, Zeimet AG, Muller-Holzner E, Deibl M, Gastl G, Gunsilius E, Marth C (2005) The expression of the regulatory T cells-specific forkhead box transcription factor FoxP3 is associated with poor prognosis in ovarian cancer. Clin Cancer Res 11: 8326–8331
Woo EY, Chu CS, Goletz TJ, Schlienger K, Yeh H, Coukos G, Rubin SC, Kaiser LR, June CH (2001) Regulatory CD4(+)CD25(+) T cells in tumors from patients with early-stage non-small cell lung cancer and late-stage ovarian cancer. Cancer Res 61: 4766–4772
Yagi H, Nomura T, Nakamura K, Yamazaki S, Kitawaki T, Hori S, Maeda M, Onodera M, Uchiyama T, Fujii S, Sakaguchi S (2004) Crucial role of FOXP3 in the development and function of human CD25+CD4+ regulatory T cells. Int Immunol 16: 1643–1656
Zheng SG, Wang JH, Gray JD, Soucier H, Horwitz DA (2004) Natural and induced CD4+CD25+ cells educate CD4+CD25− cells to develop suppressive activity: the role of IL-2, TGF-β, and IL-10. J Immunol 172: 5213–5221
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Mizukami, Y., Kono, K., Kawaguchi, Y. et al. Localisation pattern of Foxp3+ regulatory T cells is associated with clinical behaviour in gastric cancer. Br J Cancer 98, 148–153 (2008). https://doi.org/10.1038/sj.bjc.6604149
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6604149
Keywords
This article is cited by
-
Regulatory T cell-mediated immunosuppression orchestrated by cancer: towards an immuno-genomic paradigm for precision medicine
Nature Reviews Clinical Oncology (2024)
-
The immune contexture and Immunoscore in cancer prognosis and therapeutic efficacy
Nature Reviews Cancer (2020)
-
From inflammation to gastric cancer – the importance of Hedgehog/GLI signaling in Helicobacter pylori-induced chronic inflammatory and neoplastic diseases
Cell Communication and Signaling (2017)
-
Increased frequency of CD4+ CD25+ FOXP3+ cells correlates with the progression of 4-nitroquinoline1-oxide-induced rat tongue carcinogenesis
Clinical Oral Investigations (2014)
-
Critical role of spatial interaction between CD8+ and Foxp3+ cells in human gastric cancer: the distance matters
Cancer Immunology, Immunotherapy (2014)