Abstract
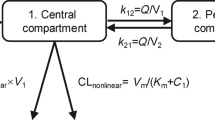
We have studied the pharmacokinetics of mitoxantrone in cancer patients. Two regimens were used: eight women (10 kinetics) received a 10 min i.v. infusion of 12 mg m-2 of mitoxantrone; seven women (seven kinetics) received high-dose mitoxantrone associated to high-dose alkylating agents and underwent autologous bone marrow transplantation (BMT). High-dose mitoxantrone was administered according to two different protocols. The drug was quantified in plasma with an HPLC assay and pharmacokinetic analysis was performed with the APIS software. Mitoxantrone pharmacokinetics were best described by an open two- (six kinetics) or an open three compartment model (11 kinetics). A large interindivual variability was observed in pharmacokinetic parameters. In the first group of patients, mean +/- s.d. values of clearance, half-life and total distribution volume were 21.41 +/- 14.59 1 h-1, 19.83 +/- 23.95 h, 165.89 +/- 134.75 1 respectively. In the high-dose group, these values were 21.68 +/- 7.30 1 h-1, 50.26 +/- 20.62 h, 413.70 +/- 194.81 1 respectively. Results showed that identification through the open 2-compartment model is certainly related to the small number of late time-points. We therefore think that mitoxantrone pharmacokinetics is generally best described by an open 3-compartment model. Clearance values showed that there was no saturation in mitoxantrone elimination, even at the highest doses. Terminal elimination half-life was probably underestimated because of the lack of late time-points in some kinetics. The half-life is long for patients receiving high-dose mitoxantrone (mean value was 50 h) and it would be hazardous to perform BMT too early after mitoxantrone infusion. Mitoxantrone metabolites were detected in the plasma of five patients receiving high-dose mitoxantrone and in one with hepatic impairment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Richard, B., Launay-Iliadis, M., Iliadis, A. et al. Pharmacokinetics of mitoxantrone in cancer patients treated by high-dose chemotherapy and autologous bone marrow transplantation. Br J Cancer 65, 399–404 (1992). https://doi.org/10.1038/bjc.1992.81
Issue Date:
DOI: https://doi.org/10.1038/bjc.1992.81
This article is cited by
-
Impact of anticancer chemotherapy on the extension of beta-lactamase spectrum: an example with KPC-type carbapenemase activity towards ceftazidime-avibactam
Scientific Reports (2020)
-
Overexpression of erb B2 remains a major risk factor in non-metastatic breast cancers treated with high-dose alkylating agents and autologous stem cell transplantation
Bone Marrow Transplantation (2002)
-
Assessment of the therapeutic potential of cytokines, cytotoxic drugs and effector cell populations for the treatment of multiple myeloma using the 5T33 murine myeloma model
Immunology & Cell Biology (1995)